Label: PHENAZOPYRIDINE HYDROCHLORIDE- phenazopyridine tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 65162-517-10, 65162-520-10 - Packager: Amneal Pharmaceuticals LLC
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated May 17, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Phenazopyridine Hydrochloride is light or dark red to dark violet, odorless, slightly bitter, crystalline powder. It has a specific local analgesic effect in the urinary tract, promptly relieving burning and pain. It has the following structural formula:
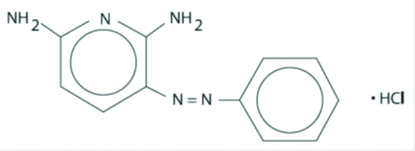
C11H11N5•HCl M.W. 249.70
Phenazopyridine HCl tablets contain the following inactive ingredients: carnauba wax, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, pregelatinized starch.
-
CLINICAL PHARMACOLOGY
Phenazopyridine HCl is excreted in the urine where it exerts a topical analgesic effect on the mucosa of the urinary tract. This action helps to relieve pain, burning, urgency and frequency. The precise mechanism of action is not known.
The pharmacokinetic properties of Phenazopyridine HCl have not been determined. Phenazopyridine HCl is rapidly excreted by the kidneys, with as much as 66% of an oral dose being excreted unchanged in the urine.
-
INDICATIONS AND USAGE
Phenazopyridine HCl is indicated for the symptomatic relief of pain, burning, urgency, frequency, and other discomforts arising from irritation of the lower urinary tract mucosa caused by infection, trauma, surgery, endoscopic procedures, or the passage of sounds or catheters. The use of Phenazopyridine HCl for relief of symptoms should not delay definitive diagnosis and treatment of causative conditions. Because it provides only symptomatic relief, prompt appropriate treatment of the cause of pain must be instituted and Phenazopyridine HCl should be discontinued when symptoms are controlled.
The analgesic action may reduce or eliminate the need for systemic analgesics or narcotics. It is, however, compatible with antibacterial therapy and can help to relieve pain and discomfort during the interval before antibacterial therapy controls the infection. Treatment of a urinary tract infection with Phenazopyridine HCl should not exceed 2 days because there is a lack of evidence that the combined administration of Phenazopyridine HCl and an antibacterial provides greater benefit than administration of the antibacterial alone after 2 days. (See DOSAGE AND ADMINISTRATION section.)
- CONTRAINDICATIONS
-
ADVERSE REACTIONS
Headache, rash, pruritus and occasional gastrointestinal disturbance. An anaphylactoid-like reaction has been described. Methemoglobinemia, hemolytic anemia, renal and hepatic toxicity have been reported, usually at overdosage levels (see Overdosage Section).
-
PRECAUTIONS
General
A yellowish tinge of the skin or sclera may indicate accumulation due to impaired renal excretion and the need to discontinue therapy. The decline in renal function associated with advanced age should be kept in mind.
NOTE: Patients should be informed that Phenazopyridine HCl produces a reddish-orange discoloration of the urine and may stain fabric. Staining of contact lenses has been reported.
Laboratory Test Interaction
Due to its properties as an azo dye, Phenazopyridine HCl may interfere with urinalysis based on spectrometry or color reactions.
Carcinogenicity, Mutagenicity, Impairment of Fertility
Long-term administration of Phenazopyridine HCl has induced neoplasia in rats (large intestine) and mice (liver).
Although no association between Phenazopyridine HCl and human neoplasia has been reported, adequate epidemiological studies along these lines have not been conducted.
Pregnancy Category B
Reproduction studies have been performed in rats at doses up to
50 mg/kg/day and have revealed no evidence of impaired fertility or harm to the fetus due to Phenazopyridine HCl. There are, however, no adequate and well controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
-
DOSAGE AND ADMINISTRATION
100 mg Tablets: Average adult dosage is two tablets 3 times a day after meals.
200 mg Tablets: Average adult dosage is one tablet 3 times a day after meals.
When used concomitantly with an antibacterial agent for the treatment of a urinary tract infection, the administration of Phenazopyridine HCl should not exceed 2 days.
-
OVERDOSAGE
Exceeding the recommended dose in patients with good renal function or administering the usual dose to patients with impaired renal function (common in elderly patients) may lead to increased serum levels and toxic reactions. Methemoglobinemia generally follows a massive, acute overdose. Methylene blue, 1 to 2 mg/kg/body weight intravenously or ascorbic acid 100 to 200 mg given orally should cause prompt reduction of the methemoglobinemia and disappearance of the cyanosis which is an aid in diagnosis. Oxidative Heinz body hemolytic anemia may also occur, and “bite cells” (degmacytes) may be present in a chronic overdosage situation. Red blood cell G-6-PD deficiency may predispose to hemolysis. Renal and hepatic impairment and occasional failure, usually due to hypersensitivity, may also occur.
-
HOW SUPPLIED
100 mg Tablets: Supplied in bottles of 100 (NDC 65162-517-10) counts.
Appearance: Deep brown to maroon colored, round, film coated tablets debossed “AN” above “1” on one side and plain on the other.
200 mg Tablets: Supplied in bottles of 100 (NDC 65162-520-10) counts.
Appearance: Deep brown to maroon colored, round, film coated tablets debossed “AN” above “2” on one side and plain on the other.
DISPENSE contents with a child-resistant closure (as required) and in a tight container as defined in the USP.
STORE at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Distributed by:
Amneal Pharmaceuticals
Bridgewater, NJ 08807Rev. 12-2015-00
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PHENAZOPYRIDINE HYDROCHLORIDE
phenazopyridine tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65162-517 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENAZOPYRIDINE HYDROCHLORIDE (UNII: 0EWG668W17) (PHENAZOPYRIDINE - UNII:K2J09EMJ52) PHENAZOPYRIDINE HYDROCHLORIDE 100 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 1000 (UNII: U076Q6Q621) POVIDONE (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color BROWN (MAROON) Score no score Shape ROUND Size 10mm Flavor Imprint Code AN;1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65162-517-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 02/01/2011 PHENAZOPYRIDINE HYDROCHLORIDE
phenazopyridine tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:65162-520 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENAZOPYRIDINE HYDROCHLORIDE (UNII: 0EWG668W17) (PHENAZOPYRIDINE - UNII:K2J09EMJ52) PHENAZOPYRIDINE HYDROCHLORIDE 200 mg Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL 1000 (UNII: U076Q6Q621) POVIDONE (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color BROWN (MAROON) Score no score Shape ROUND Size 10mm Flavor Imprint Code AN;2 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65162-520-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 02/01/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 02/01/2011 Labeler - Amneal Pharmaceuticals LLC (123797875)




