Label: GUAIFENESIN EXTENDED RELEASE- guaifenesin tablet, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 51660-072-54, 51660-072-86 - Packager: OHM LABORATORIES INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 5, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
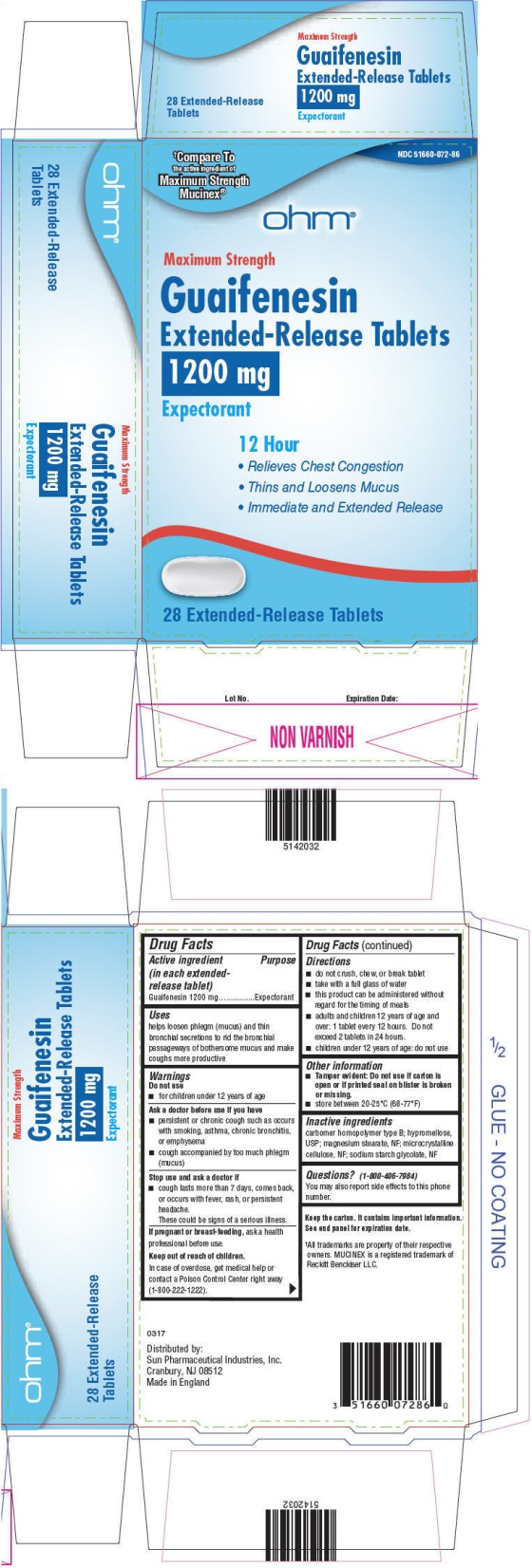
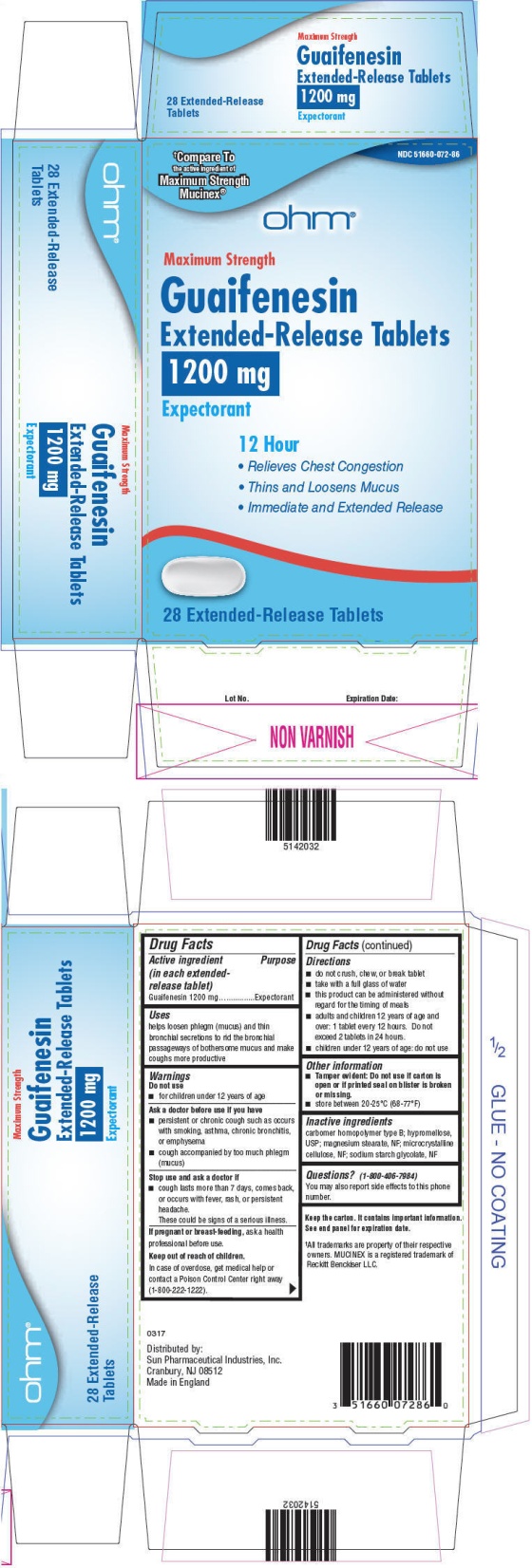
- Active ingredient (in each extended-release tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- •
- cough accompanied by too much phlegm (mucus)
-
Directions
- •
- do not crush, chew, or break tablet
- •
- take with a full glass of water
- •
- this product can be administered without regard for the timing of meals
- •
- adults and children 12 years of age and over: 1 tablet every 12 hours. Do not exceed 2 tablets in 24 hours.
- •
- children under 12 years of age: do not use
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 1200 mg Tablet Blister Pack Carton
-
INGREDIENTS AND APPEARANCE
GUAIFENESIN EXTENDED RELEASE
guaifenesin tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51660-072 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Guaifenesin (UNII: 495W7451VQ) (Guaifenesin - UNII:495W7451VQ) Guaifenesin 1200 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) hypromellose, unspecified (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score no score Shape OVAL Size 22mm Flavor Imprint Code Mxeunic;1200 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51660-072-54 1 in 1 CARTON 12/10/2017 1 14 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:51660-072-86 2 in 1 CARTON 12/10/2017 2 14 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021282 12/10/2017 Labeler - OHM LABORATORIES INC (184769029) Establishment Name Address ID/FEI Business Operations RECKITT BENCKISER HEALTHCARE INTERNATIONAL LTD 230780363 MANUFACTURE(51660-072)