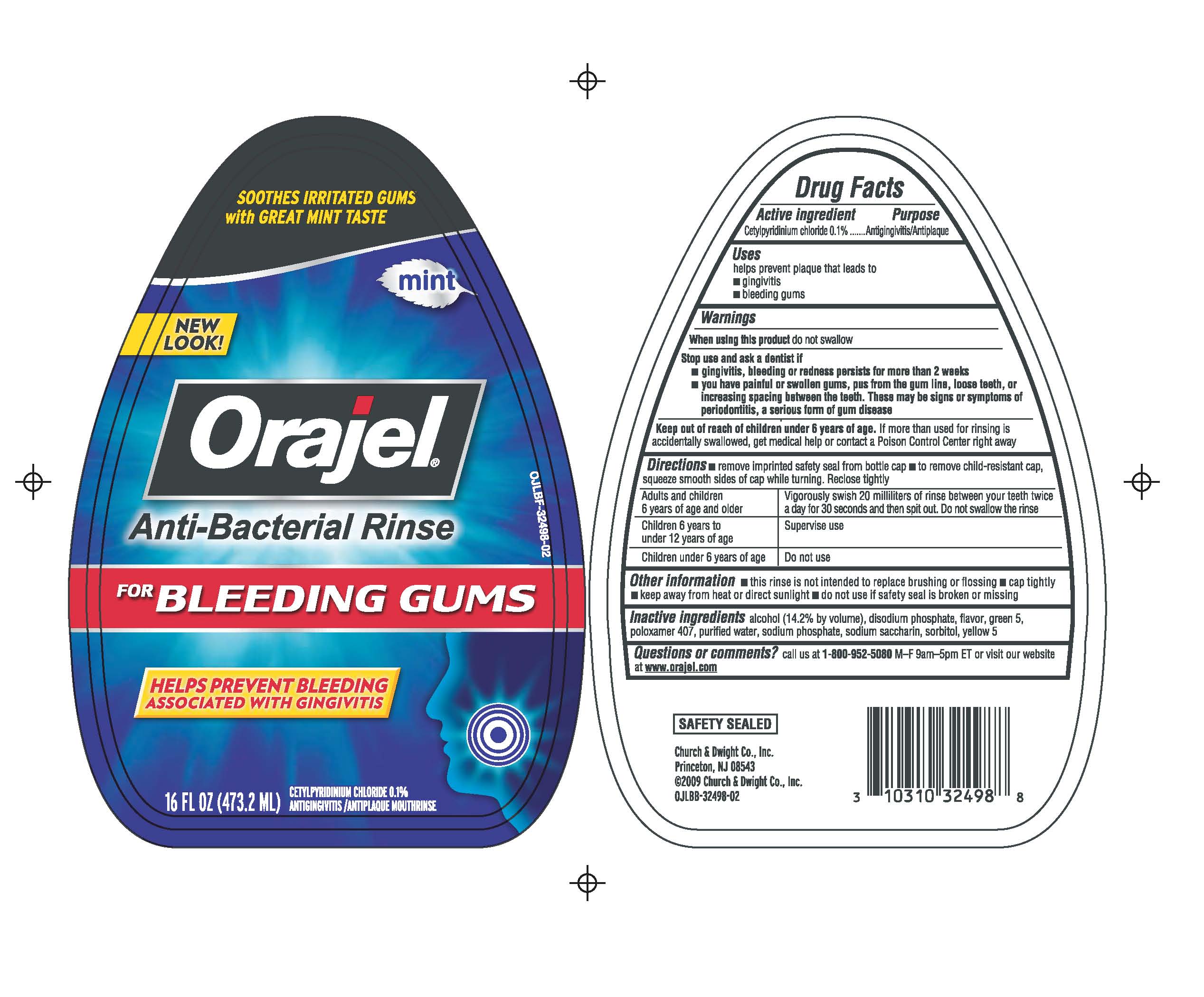
Label: ORAJLE ANTI-BACTERIAL RINSE FOR BLEEDING GUMS- cetylpyridinium chloride liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 10237-721-16 - Packager: Church & Dwight Co., Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 30, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active ingredient Purpose
Cetylpyridinium chloride 0.1% Antigingivitis/Antiplaque
Warnings
When using this product do not swallow
Stop use and ask a dentist if
- gingivitis, bleeding or redness persists for more than 2 weeks
- you have painful or swollen gums, pus from the gum line, loose teeth, or increasing spacing between the teeth. These may be signs or symptoms of periodontitis, a serious form of gum disease.
Directions remove imprinted safety seal from bottle cap; to remove child-resistant cap, squeeze smooth sides of cap while turning. Reclose tightly.
Adults and children Vigorously swish 20 milliliters of rinse between your teeth
6 years of age and older twice a day for 30 seconds and then spit out. Do not swallow the rinseChildren 6 years to under Supervise use
12 years of age
Children under 6 years of age Do not use
Other information this rinse is not intended to replace brushing or flossing ; cap tightly; keep away from heat or direct sunlight; do not use if safety seal is broken or missing.
Inactive ingredients alcohol (14.2% by volume), disodium phosphate, flavor, green 5, poloxamer 407, purified water, sodium phosphate, sodium saccharin, sorbitol, yellow 5
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ORAJLE ANTI-BACTERIAL RINSE FOR BLEEDING GUMS
cetylpyridinium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10237-721 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) (CETYLPYRIDINIUM - UNII:CUB7JI0JV3) CETYLPYRIDINIUM CHLORIDE .001 mL in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) PHOSPHATE ION (UNII: NK08V8K8HR) D&C GREEN NO. 5 (UNII: 8J6RDU8L9X) POLOXAMER 407 (UNII: TUF2IVW3M2) WATER (UNII: 059QF0KO0R) SODIUM PHOSPHATE (UNII: SE337SVY37) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SORBITOL (UNII: 506T60A25R) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10237-721-16 473.2 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 07/30/2010 Labeler - Church & Dwight Co., Inc. (001211952) Establishment Name Address ID/FEI Business Operations Accupac, Inc. 071609663 manufacture