Label: ALLERGY RELIEF- diphenhydramine hcl tablet, film coated
- NDC Code(s): 46122-441-62
- Packager: Amerisource Bergen
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated September 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
- to make a child sleepy
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
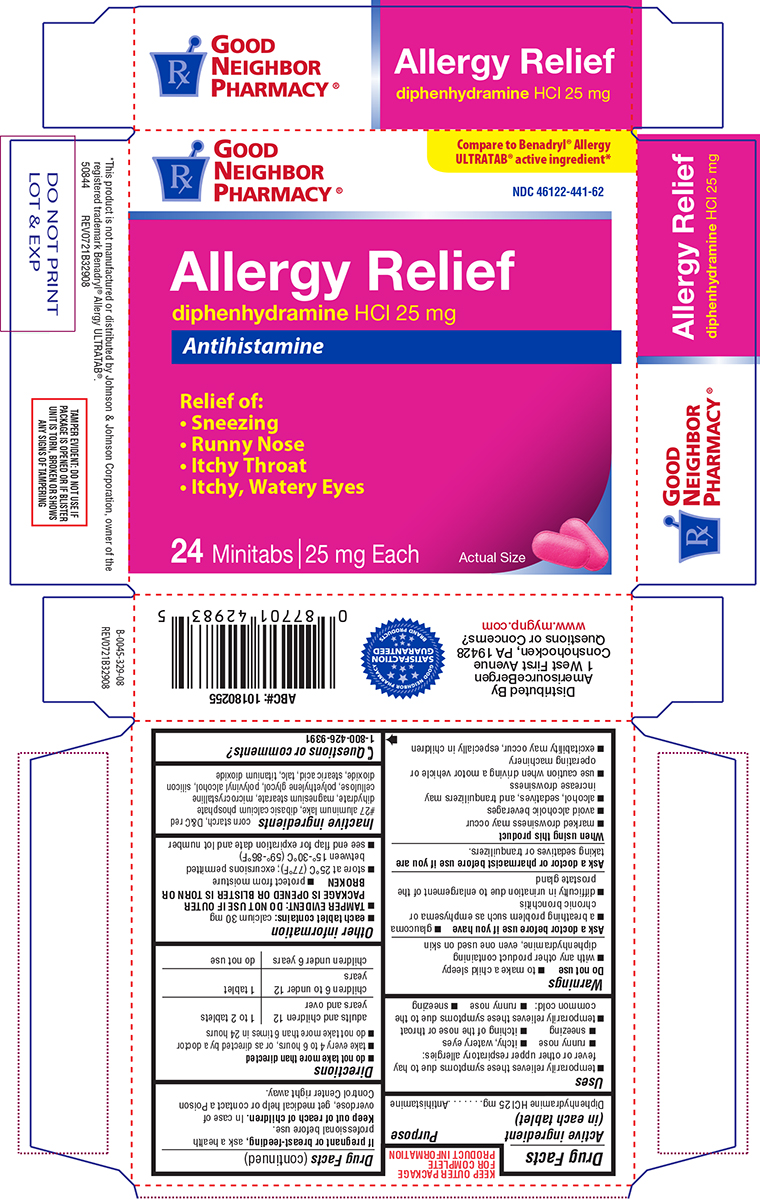
Principal display panel
GOOD
NEIGHBOR
PHARMACY®Compare to Benadryl® Allergy
ULTRATAB® active ingredient*NDC 46122-441-62
Allergy Relief
diphenhydramine HCl 25 mg
AntihistamineRelief of:
• Sneezing
• Runny Nose
• Itchy Throat
• Itchy, Watery Eyes24 Minitabs | 25 mg Each
Actual Size
*This product is not manufactured or distributed by Johnson & Johnson Corporation, owner of the
registered trademark Benadryl® Allergy ULTRATAB®.
50844 REV0721B32908TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERINGDistributed By
AmerisourceBergen
1 West First Avenue
Conshohocken, PA 19428
Questions or Concerns?
www.mygnp.com
GOOD NEIGHBOR PHARMACY 44-329
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
diphenhydramine hcl tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46122-441 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) D&C RED NO. 27 ALUMINUM LAKE (UNII: ZK64F7XSTX) DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color pink Score no score Shape OVAL Size 11mm Flavor Imprint Code 44;329 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46122-441-62 2 in 1 CARTON 03/02/1990 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 03/02/1990 Labeler - Amerisource Bergen (007914906) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(46122-441) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(46122-441) , pack(46122-441) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(46122-441) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 manufacture(46122-441) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(46122-441)