Label: LORATADINE AND PSEUDOEPHEDRINE SULFATE tablet, film coated, extended release
- NDC Code(s): 30142-724-15, 30142-724-69
- Packager: The Kroger Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 26, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
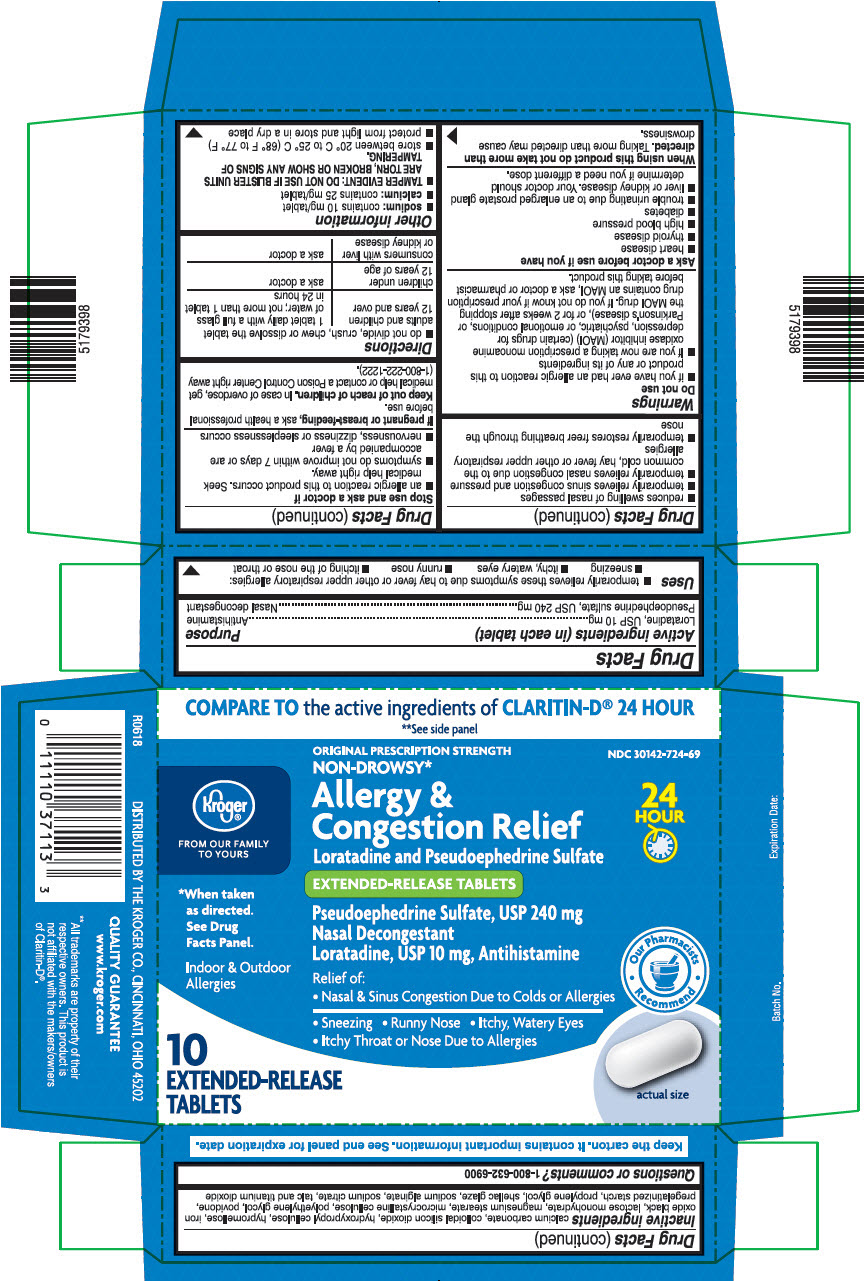
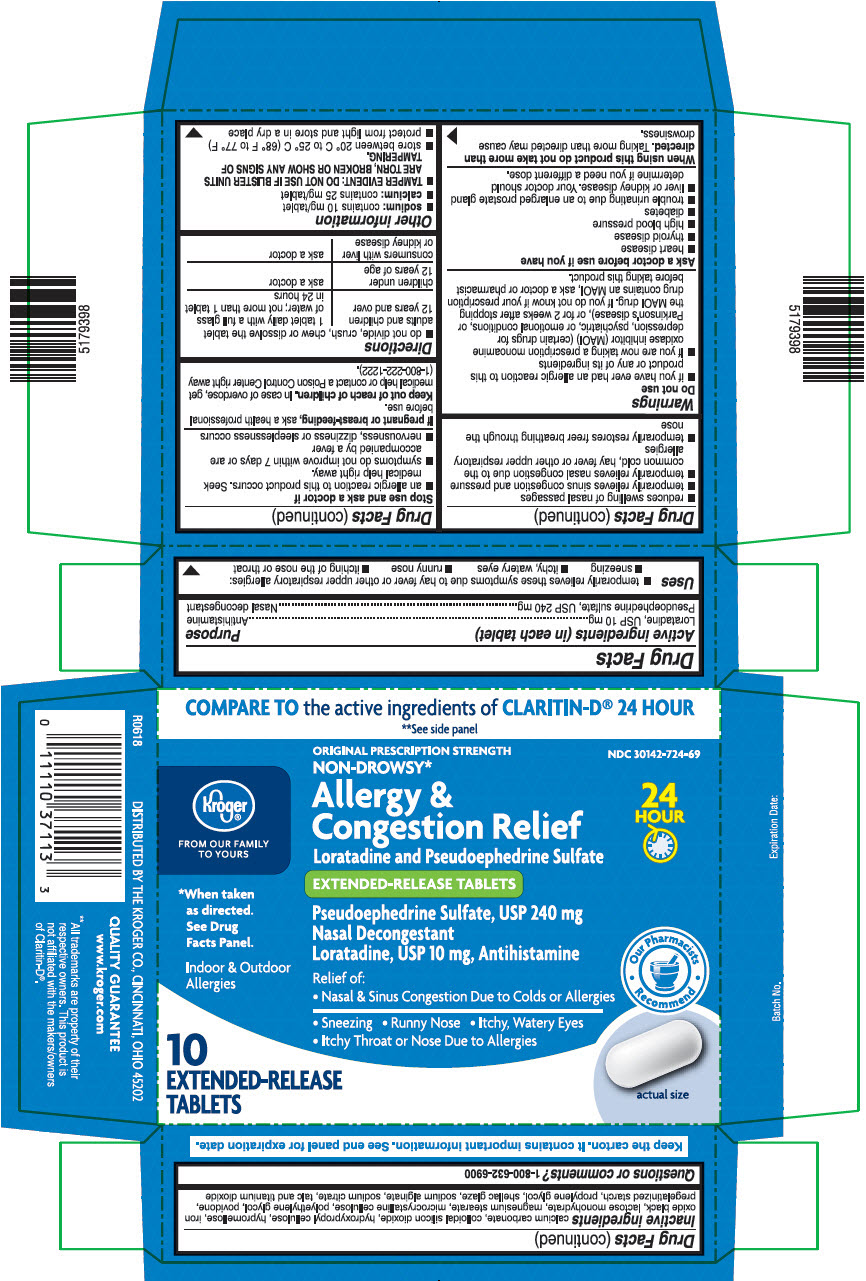
- ACTIVE INGREDIENTS (IN EACH TABLET)
- PURPOSE
-
USES
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- sneezing
- itchy, watery eyes
- runny nose
- itching of the nose or throat
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily restores freer breathing through the nose
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
-
WARNINGS
Do not use
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- heart disease
- thyroid disease
- high blood pressure
- diabetes
- trouble urinating due to an enlarged prostate gland
- liver or kidney disease. Your doctor should determine if you need a different dose.
- DIRECTIONS
- OTHER INFORMATION
-
INACTIVE INGREDIENTS
Calcium carbonate, colloidal silicon dioxide, hydroxypropyl cellulose, hypromellose, iron oxide black, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, pregelatinized starch, propylene glycol, shellac glaze, sodium alginate, sodium citrate, talc and titanium dioxide
- QUESTIONS OR COMMENTS?
-
PRINCIPAL DISPLAY PANEL - 240 mg/10 mg Tablet Blister Pack Carton
COMPARE TO the active ingredients of CLARITIN-D® 24 HOUR
**See side panelKroger®
FROM OUR FAMILY
TO YOURSORIGINAL PRESCRIPTION STRENGTH
NON-DROWSY*NDC 30142-724-69
Allergy &
Congestion Relief
Loratadine and Pseudoephedrine Sulfate24
HOUREXTENDED-RELEASE TABLETS
*When taken
as directed.
See Drug
Facts Panel.Pseudoephedrine Sulfate, USP 240 mg
Nasal Decongestant
Loratadine, USP 10 mg, AntihistamineIndoor & Outdoor
AllergiesRelief of:
• Nasal & Sinus Congestion Due to Colds or Allergies
• Sneezing • Runny Nose • Itchy, Watery Eyes
• Itchy Throat or Nose Due to AllergiesOur Pharmacists
Recommend10
EXTENDED-RELEASE
TABLETSactual size
-
INGREDIENTS AND APPEARANCE
LORATADINE AND PSEUDOEPHEDRINE SULFATE
loratadine and pseudoephedrine sulfate tablet, film coated, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:30142-724 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg PSEUDOEPHEDRINE SULFATE (UNII: Y9DL7QPE6B) (PSEUDOEPHEDRINE - UNII:7CUC9DDI9F) PSEUDOEPHEDRINE SULFATE 240 mg Inactive Ingredients Ingredient Name Strength CALCIUM CARBONATE (UNII: H0G9379FGK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYDROXYPROPYL CELLULOSE (1600000 WAMW) (UNII: RFW2ET671P) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) FERROSOFERRIC OXIDE (UNII: XM0M87F357) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) STARCH, CORN (UNII: O8232NY3SJ) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) SODIUM ALGINATE (UNII: C269C4G2ZQ) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (White to Off-White) Score no score Shape CAPSULE Size 17mm Flavor Imprint Code RX724 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:30142-724-69 10 in 1 CARTON 11/17/2004 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC:30142-724-15 15 in 1 CARTON 11/17/2004 2 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA076557 11/17/2004 Labeler - The Kroger Company (006999528) Registrant - Sun Pharmaceutical Industries Inc. (146974886) Establishment Name Address ID/FEI Business Operations Ohm Laboratories Inc. 051565745 manufacture(30142-724)