ZINACEF- cefuroxime sodium injection, solution
ZINACEF- cefuroxime sodium injection, powder, for solution
GlaxoSmithKline LLC
----------
ZINACEF®
(cefuroxime for injection)
ZINACEF®
(cefuroxime injection)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZINACEF and other antibacterial drugs, ZINACEF should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
DESCRIPTION
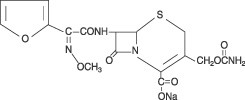
Cefuroxime is a semisynthetic, broad-spectrum, cephalosporin antibiotic for parenteral administration. It is the sodium salt of (6R,7R)-3-carbamoyloxymethyl-7-[Z-2-methoxyimino-2-(fur-2-yl)acetamido]ceph-3-em-4-carboxylate, and it has the following chemical structure:
The empirical formula is C16H15N4NaO8S, representing a molecular weight of 446.4.
ZINACEF contains approximately 54.2 mg (2.4 mEq) of sodium per gram of cefuroxime activity.
ZINACEF in sterile crystalline form is supplied in vials equivalent to 750 mg, 1.5 g, or 7.5 g of cefuroxime as cefuroxime sodium and in ADD-Vantage® vials equivalent to 750 mg or 1.5 g of cefuroxime as cefuroxime sodium. Solutions of ZINACEF range in color from light yellow to amber, depending on the concentration and diluent used. The pH of freshly constituted solutions usually ranges from 6 to 8.5.
ZINACEF is available as a frozen, iso-osmotic, sterile, nonpyrogenic solution with 750 mg or 1.5 g of cefuroxime as cefuroxime sodium. Approximately 1.4 g of Dextrose Hydrous, USP has been added to the 750-mg dose to adjust the osmolality. Sodium Citrate Hydrous, USP has been added as a buffer (300 mg and 600 mg to the 750-mg and 1.5-g doses, respectively). ZINACEF contains approximately 111 mg (4.8 mEq) and 222 mg (9.7 mEq) of sodium in the 750-mg and 1.5-g doses, respectively. The pH has been adjusted with hydrochloric acid and may have been adjusted with sodium hydroxide. Solutions of premixed ZINACEF range in color from light yellow to amber. The solution is intended for intravenous (IV) use after thawing to room temperature. The osmolality of the solution is approximately 300 mOsmol/kg, and the pH of thawed solutions ranges from 5 to 7.5.
The plastic container for the frozen solution is fabricated from a specially designed multilayer plastic, PL 2040. Solutions are in contact with the polyethylene layer of this container and can leach out certain chemical components of the plastic in very small amounts within the expiration period. The suitability of the plastic has been confirmed in tests in animals according to USP biological tests for plastic containers as well as by tissue culture toxicity studies.
CLINICAL PHARMACOLOGY
After intramuscular (IM) injection of a 750-mg dose of cefuroxime to normal volunteers, the mean peak serum concentration was 27 mcg/mL. The peak occurred at approximately 45 minutes (range, 15 to 60 minutes). Following IV doses of 750 mg and 1.5 g, serum concentrations were approximately 50 and 100 mcg/mL, respectively, at 15 minutes. Therapeutic serum concentrations of approximately 2 mcg/mL or more were maintained for 5.3 hours and 8 hours or more, respectively. There was no evidence of accumulation of cefuroxime in the serum following IV administration of 1.5-g doses every 8 hours to normal volunteers. The serum half-life after either IM or IV injections is approximately 80 minutes.
Approximately 89% of a dose of cefuroxime is excreted by the kidneys over an 8-hour period, resulting in high urinary concentrations.
Following the IM administration of a 750-mg single dose, urinary concentrations averaged 1,300 mcg/mL during the first 8 hours. Intravenous doses of 750 mg and 1.5 g produced urinary levels averaging 1,150 and 2,500 mcg/mL, respectively, during the first 8-hour period.
The concomitant oral administration of probenecid with cefuroxime slows tubular secretion, decreases renal clearance by approximately 40%, increases the peak serum level by approximately 30%, and increases the serum half-life by approximately 30%. Cefuroxime is detectable in therapeutic concentrations in pleural fluid, joint fluid, bile, sputum, bone, and aqueous humor.
Cefuroxime is detectable in therapeutic concentrations in cerebrospinal fluid (CSF) of adults and pediatric patients with meningitis. The following table shows the concentrations of cefuroxime achieved in cerebrospinal fluid during multiple dosing of patients with meningitis.
|
Patients |
Dose |
Number of Patients |
Mean (Range) CSF Cefuroxime Concentrations (mcg/mL) Achieved Within 8 Hours Post Dose |
|
Pediatric patients (4 weeks to 6.5 years) |
200 mg/kg/day, divided q 6 hours |
5 |
6.6 (0.9-17.3) |
|
Pediatric patients (7 months to 9 years) |
200 to 230 mg/kg/day, divided q 8 hours |
6 |
8.3 (<2-22.5) |
|
Adults |
1.5 grams q 8 hours |
2 |
5.2 (2.7-8.9) |
|
Adults |
1.5 grams q 6 hours |
10 |
6.0 (1.5-13.5) |
Cefuroxime is approximately 50% bound to serum protein.
Microbiology
Cefuroxime has in vitro activity against a wide range of gram-positive and gram-negative organisms, and it is highly stable in the presence of beta-lactamases of certain gram-negative bacteria. The bactericidal action of cefuroxime results from inhibition of cell-wall synthesis.
Cefuroxime is usually active against the following organisms in vitro.
Aerobes, Gram-positive
Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, and Streptococcus pyogenes (and other streptococci).
NOTE: Most strains of enterococci, e.g., Enterococcusfaecalis (formerly Streptococcus faecalis), are resistant to cefuroxime. Methicillin-resistant staphylococci and Listeriamonocytogenes are resistant to cefuroxime.
Aerobes, Gram-negative
Citrobacter spp., Enterobacter spp., Escherichia coli, Haemophilusinfluenzae (including ampicillin-resistant strains), Haemophilusparainfluenzae, Klebsiella spp. (including Klebsiellapneumoniae), Moraxella (Branhamella) catarrhalis (including ampicillin- and cephalothin-resistant strains), Morganellamorganii (formerly Proteus morganii), Neisseriagonorrhoeae (including penicillinase- and non–penicillinase-producing strains), Neisseriameningitidis, Proteus mirabilis, Providenciarettgeri(formerly Proteus rettgeri), Salmonella spp., and Shigella spp.
NOTE: Some strains of Morganellamorganii, Enterobacter cloacae, and Citrobacter spp. have been shown by in vitro tests to be resistant to cefuroxime and other cephalosporins. Pseudomonas and Campylobacter spp., Legionellaspp.,Acinetobactercalcoaceticus, and most strains of Serratia spp. and Proteus vulgarisare resistant to most first- and second-generation cephalosporins.
Anaerobes
Gram-positive and gram-negative cocci (including Peptococcus and Peptostreptococcus spp.), gram-positive bacilli (including Clostridium spp.), and gram-negative bacilli (including Bacteroides and Fusobacterium spp.).
NOTE: Clostridium difficile and most strains of Bacteroidesfragilis are resistant to cefuroxime.
Susceptibility Tests
Diffusion Techniques
Quantitative methods that require measurement of zone diameters give an estimate of antibiotic susceptibility. One such standard procedure1 that has been recommended for use with disks to test susceptibility of organisms to cefuroxime uses the 30-mcg cefuroxime disk. Interpretation involves the correlation of the diameters obtained in the disk test with the minimum inhibitory concentration (MIC) for cefuroxime.
A report of “Susceptible” indicates that the pathogen is likely to be inhibited by generally achievable blood levels. A report of “Moderately Susceptible” suggests that the organism would be susceptible if high dosage is used or if the infection is confined to tissues and fluids in which high antibiotic levels are attained. A report of “Intermediate” suggests an equivocable or indeterminate result. A report of “Resistant” indicates that achievable concentrations of the antibiotic are unlikely to be inhibitory and other therapy should be selected.
Reports from the laboratory giving results of the standard single-disk susceptibility test for organisms other than Haemophilus spp. and Neisseriagonorrhoeae with a 30-mcg cefuroxime disk should be interpreted according to the following criteria:
|
Zone Diameter (mm) |
Interpretation |
|
≥18 |
(S) Susceptible |
|
15-17 |
(MS) Moderately Susceptible |
|
≤14 |
(R) Resistant |
Results for Haemophilus spp. should be interpreted according to the following criteria:
|
Zone Diameter (mm) |
Interpretation |
|
≥24 |
(S) Susceptible |
|
21-23 |
(I) Intermediate |
|
≤20 |
(R) Resistant |
Results for Neisseriagonorrhoeae should be interpreted according to the following criteria:
|
Zone Diameter (mm) |
Interpretation |
|
≥31 |
(S) Susceptible |
|
26-30 |
(MS) Moderately Susceptible |
|
≤25 |
(R) Resistant |
Organisms should be tested with the cefuroxime disk since cefuroxime has been shown by in vitro tests to be active against certain strains found resistant when other beta-lactam disks are used. The cefuroxime disk should not be used for testing susceptibility to other cephalosporins.
Standardized procedures require the use of laboratory control organisms. The 30-mcg cefuroxime disk should give the following zone diameters.
1. Testing for organisms other than Haemophilus spp. and Neisseriagonorrhoeae:
|
Organism |
Zone Diameter (mm) |
|
Staphylococcus aureus ATCC 25923 |
27-35 |
|
Escherichia coli ATCC 25922 |
20-26 |
2. Testing for Haemophilus spp.:
|
Organism |
Zone Diameter (mm) |
|
Haemophilusinfluenzae ATCC 49766 |
28-36 |
3. Testing for Neisseriagonorrhoeae:
|
Organism |
Zone Diameter (mm) |
|
Neisseriagonorrhoeae ATCC 49226 |
33-41 |
|
Staphylococcus aureus ATCC 25923 |
29-33 |
Dilution Techniques
Use a standardized dilution method1 (broth, agar, microdilution) or equivalent with cefuroxime powder. The MIC values obtained for bacterial isolates other than Haemophilus spp. and Neisseriagonorrhoeae should be interpreted according to the following criteria:
|
MIC (mcg/mL) |
Interpretation |
|
≤8 |
(S) Susceptible |
|
16 |
(MS) Moderately Susceptible |
|
≥32 |
(R) Resistant |
MIC values obtained for Haemophilus spp. should be interpreted according to the following criteria:
|
MIC (mcg/mL) |
Interpretation |
|
≤4 |
(S) Susceptible |
|
8 |
(I) Intermediate |
|
≥16 |
(R) Resistant |
MIC values obtained for Neisseriagonorrhoeae should be interpreted according to the following criteria:
|
MIC (mcg/mL) |
Interpretation |
|
≤1 |
(S) Susceptible |
|
2 |
(MS) Moderately Susceptible |
|
≥4 |
(R) Resistant |
As with standard diffusion techniques, dilution methods require the use of laboratory control organisms. Standard cefuroxime powder should provide the following MIC values.
1. For organisms other than Haemophilus spp. and Neisseriagonorrhoeae:
|
Organism |
MIC (mcg/mL) |
|
Staphylococcus aureus ATCC 29213 |
0.5-2.0 |
|
Escherichia coli ATCC 25922 |
2.0-8.0 |
2. For Haemophilus spp.:
|
Organism |
MIC (mcg/mL) |
|
Haemophilusinfluenzae ATCC 49766 |
0.25-1.0 |
3. For Neisseriagonorrhoeae:
|
Organism |
MIC (mcg/mL) |
|
Neisseriagonorrhoeae ATCC 49226 |
0.25-1.0 |
|
Staphylococcus aureus ATCC 29213 |
0.25-1.0 |
INDICATIONS AND USAGE
ZINACEF is indicated for the treatment of patients with infections caused by susceptible strains of the designated organisms in the following diseases:
- 1.
- Lower Respiratory Tract Infections, including pneumonia, caused by Streptococcus pneumoniae, Haemophilusinfluenzae (including ampicillin-resistant strains), Klebsiella spp., Staphylococcus aureus (penicillinase- and non–penicillinase-producing strains), Streptococcus pyogenes, and Escherichia coli.
- 2.
- Urinary Tract Infections caused by Escherichia coli and Klebsiella spp.
- 3.
- Skin and SkinStructure Infections caused by Staphylococcus aureus (penicillinase- and non–penicillinase-producing strains), Streptococcus pyogenes, Escherichia coli, Klebsiella spp., and Enterobacter spp.
- 4.
- Septicemia caused by Staphylococcus aureus (penicillinase- and non–penicillinase-producing strains), Streptococcus pneumoniae, Escherichia coli, Haemophilusinfluenzae (including ampicillin-resistant strains), and Klebsiella spp.
- 5.
- Meningitis caused by Streptococcus pneumoniae, Haemophilusinfluenzae (including ampicillin-resistant strains),Neisseriameningitidis, and Staphylococcus aureus (penicillinase- and non–penicillinase-producing strains).
- 6.
- Gonorrhea: Uncomplicated and disseminated gonococcal infections due to Neisseriagonorrhoeae(penicillinase- and non–penicillinase-producing strains) in both males and females.
- 7.
- Bone and Joint Infections caused by Staphylococcus aureus (penicillinase- and non–penicillinase-producing strains).
Clinical microbiological studies in skin and skinstructure infections frequently reveal the growth of susceptible strains of both aerobic and anaerobic organisms. ZINACEF has been used successfully in these mixed infections in which several organisms have been isolated.
In certain cases of confirmed or suspected gram-positive or gram-negative sepsis or in patients with other serious infections in which the causative organism has not been identified, ZINACEF may be used concomitantly with an aminoglycoside (see PRECAUTIONS). The recommended doses of both antibiotics may be given depending on the severity of the infection and the patient's condition.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZINACEF and other antibacterial drugs, ZINACEF should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Prevention
The preoperative prophylactic administration of ZINACEF may prevent the growth of susceptible disease-causing bacteria and thereby may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures (e.g., vaginal hysterectomy) that are classified as clean-contaminated or potentially contaminated procedures. Effective prophylactic use of antibiotics in surgery depends on the time of administration. ZINACEF should usually be given one-half to 1 hour before the operation to allow sufficient time to achieve effective antibiotic concentrations in the wound tissues during the procedure. The dose should be repeated intraoperatively if the surgical procedure is lengthy.
Prophylactic administration is usually not required after the surgical procedure ends and should be stopped within 24 hours. In the majority of surgical procedures, continuing prophylactic administration of any antibiotic does not reduce the incidence of subsequent infections but will increase the possibility of adverse reactions and the development of bacterial resistance.
The perioperative use of ZINACEF has also been effective during open heart surgery for surgical patients in whom infections at the operative site would present a serious risk. For these patients it is recommended that therapy with ZINACEF be continued for at least 48 hours after the surgical procedure ends. If an infection is present, specimens for culture should be obtained for the identification of the causative organism, and appropriate antimicrobial therapy should be instituted.
CONTRAINDICATIONS
ZINACEF is contraindicated in patients with known allergy to the cephalosporin group of antibiotics.
WARNINGS
BEFORE THERAPY WITH ZINACEF IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. THIS PRODUCT SHOULD BE GIVEN CAUTIOUSLY TO PENICILLIN-SENSITIVE PATIENTS. ANTIBIOTICS SHOULD BE ADMINISTERED WITH CAUTION TO ANY PATIENT WHO HAS DEMONSTRATED SOME FORM OF ALLERGY, PARTICULARLY TO DRUGS. IF AN ALLERGIC REACTION TO ZINACEF OCCURS, DISCONTINUE THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE EPINEPHRINE AND OTHER EMERGENCY MEASURES.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including ZINACEF, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficileproduces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
When the colitis is not relieved by drug discontinuation or when it is severe, oral vancomycin is the treatment of choice for antibiotic-associated pseudomembranous colitis produced by Clostridium difficile. Other causes of colitis should also be considered.
PRECAUTIONS
General
Although ZINACEF rarely produces alterations in kidney function, evaluation of renal status during therapy is recommended, especially in seriously ill patients receiving the maximum doses. Cephalosporins should be given with caution to patients receiving concurrent treatment with potent diuretics as these regimens are suspected of adversely affecting renal function.
The total daily dose of ZINACEF should be reduced in patients with transient or persistent renal insufficiency (see DOSAGE AND ADMINISTRATION), because high and prolonged serum antibiotic concentrations can occur in such individuals from usual doses.
As with other antibiotics, prolonged use of ZINACEF may result in overgrowth of nonsusceptible organisms. Careful observation of the patient is essential. If superinfection occurs during therapy, appropriate measures should be taken.
Broad-spectrum antibiotics should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
Nephrotoxicity has been reported following concomitant administration of aminoglycoside antibiotics and cephalosporins.
As with other therapeutic regimens used in the treatment of meningitis, mild-to-moderate hearing loss has been reported in a few pediatric patients treated with cefuroxime. Persistence of positive CSF (cerebrospinal fluid) cultures at 18 to 36 hours has also been noted with cefuroxime injection, as well as with other antibiotic therapies; however, the clinical relevance of this is unknown.
Cephalosporins may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment, or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous Vitamin K administered as indicated.
Prescribing ZINACEF in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Information for Patients
Patients should be counseled that antibacterial drugs, including ZINACEF, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When ZINACEF is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may: (1) decrease the effectiveness of the immediate treatment, and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by ZINACEF or other antibacterial drugs in the future.
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as 2 or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Drug Interactions
In common with other antibiotics, cefuroxime may affect the gut flora, leading to lower estrogen reabsorption and reduced efficacy of combined estrogen/progesterone oral contraceptives.
Drug/Laboratory Test Interactions
A false-positive reaction for glucose in the urine may occur with copper reduction tests (Benedict's or Fehling's solution or with CLINITEST® tablets) but not with enzyme-based tests for glycosuria. As a false-negative result may occur in the ferricyanide test, it is recommended that either the glucose oxidase or hexokinase method be used to determine blood plasma glucose levels in patients receiving ZINACEF.
Cefuroxime does not interfere with the assay of serum and urine creatinine by the alkaline picrate method.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Although lifetime studies in animals have not been performed to evaluate carcinogenic potential, no mutagenic activity was found for cefuroxime in the mouse lymphoma assay and a battery of bacterial mutation tests. Positive results were obtained in an in vitro chromosome aberration assay, however, negative results were found in an in vivo micronucleus test at doses up to 10 g/kg. Reproduction studies in mice at doses up to 3,200 mg/kg/day (3.1 times the recommended maximum human dose based on mg/m2) have revealed no impairment of fertility.
Reproductive studies revealed no impairment of fertility in animals.
Pregnancy
Teratogenic Effects
Pregnancy Category B. Reproduction studies have been performed in mice at doses up to 6,400 mg/kg/day (6.3 times the recommended maximum human dose based on mg/m2) and rabbits at doses up to 400 mg/kg/day (2.1 times the recommended maximum human dose based on mg/m2) and have revealed no evidence of impaired fertility or harm to the fetus due to cefuroxime. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
Since cefuroxime is excreted in human milk, caution should be exercised when ZINACEF is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in pediatric patients below 3 months of age have not been established. Accumulation of other members of the cephalosporin class in newborn infants (with resulting prolongation of drug half-life) has been reported.
Geriatric Use
Of the 1,914 subjects who received cefuroxime in 24 clinical studies of ZINACEF, 901 (47%) were 65 years and older while 421 (22%) were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater susceptibility of some older individuals to drug effects cannot be ruled out. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see DOSAGE AND ADMINISTRATION).
ADVERSE REACTIONS
ZINACEF is generally well tolerated. The most common adverse effects have been local reactions following IV administration. Other adverse reactions have been encountered only rarely.
Gastrointestinal
Gastrointestinal symptoms occurred in 1 in 150 patients and included diarrhea (1 in 220 patients) and nausea (1 in 440 patients). The onset of pseudomembranous colitis may occur during or after antibacterial treatment (see WARNINGS).
Hypersensitivity Reactions
Hypersensitivity reactions have been reported in fewer than 1% of the patients treated with ZINACEF and include rash (1 in 125). Pruritus, urticaria, and positive Coombs' test each occurred in fewer than 1 in 250 patients, and, as with other cephalosporins, rare cases of anaphylaxis, drug fever, erythema multiforme, interstitial nephritis, toxic epidermal necrolysis, and Stevens-Johnson syndrome have occurred.
Blood
A decrease in hemoglobin and hematocrit has been observed in 1 in 10 patients and transient eosinophilia in 1 in 14 patients. Less common reactions seen were transient neutropenia (fewer than 1 in 100 patients) and leukopenia (1 in 750 patients). A similar pattern and incidence were seen with other cephalosporins used in controlled studies. As with other cephalosporins, there have been rare reports of thrombocytopenia.
Hepatic
Transient rise in SGOT and SGPT (1 in 25 patients), alkaline phosphatase (1 in 50 patients), LDH (1 in 75 patients), and bilirubin (1 in 500 patients) levels has been noted.
Kidney
Elevations in serum creatinine and/or blood urea nitrogen and a decreased creatinine clearance have been observed, but their relationship to cefuroxime is unknown.
Postmarketing Experience with ZINACEF
In addition to the adverse events reported during clinical trials, the following events have been observed during clinical practice in patients treated with ZINACEF and were reported spontaneously. Data are generally insufficient to allow an estimate of incidence or to establish causation.
Cephalosporin-class Adverse Reactions
In addition to the adverse reactions listed above that have been observed in patients treated with cefuroxime, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics:
Adverse Reactions
Vomiting, abdominal pain, colitis, vaginitis including vaginal candidiasis, toxic nephropathy, hepatic dysfunction including cholestasis, aplastic anemia, hemolytic anemia, hemorrhage.
Several cephalosporins, including ZINACEF, have been implicated in triggering seizures, particularly in patients with renal impairment when the dosage was not reduced (see DOSAGE AND ADMINISTRATION). If seizures associated with drug therapy should occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
OVERDOSAGE
Overdosage of cephalosporins can cause cerebral irritation leading to convulsions. Serum levels of cefuroxime can be reduced by hemodialysis and peritoneal dialysis.
DOSAGE AND ADMINISTRATION
Dosage
Adults
The usual adult dosage range for ZINACEF is 750 mg to 1.5 grams every 8 hours, usually for 5 to 10 days. In uncomplicated urinary tract infections, skin and skinstructure infections, disseminated gonococcal infections, and uncomplicated pneumonia, a 750-mg dose every 8 hours is recommended. In severe or complicated infections, a 1.5-gram dose every 8 hours is recommended.
In bone and joint infections, a 1.5-gram dose every 8 hours is recommended. In clinical trials, surgical intervention was performed when indicated as an adjunct to therapy with ZINACEF. A course of oral antibiotics was administered when appropriate following the completion of parenteral administration of ZINACEF.
In life-threatening infections or infections due to less susceptible organisms, 1.5 grams every 6 hours may be required. In bacterial meningitis, the dosage should not exceed 3 grams every 8 hours. The recommended dosage for uncomplicated gonococcal infection is 1.5 grams given intramuscularly as a single dose at 2 different sites together with 1 gram of oral probenecid. For preventive use for clean-contaminated or potentially contaminated surgical procedures, a 1.5-gram dose administered intravenously just before surgery (approximately one-half to 1 hour before the initial incision) is recommended. Thereafter, give 750 mg intravenously or intramuscularly every 8 hours when the procedure is prolonged.
For preventive use during open heart surgery, a 1.5-gram dose administered intravenously at the induction of anesthesia and every 12 hours thereafter for a total of 6 grams is recommended.
Impaired Renal Function
A reduced dosage must be employed when renal function is impaired. Dosage should be determined by the degree of renal impairment and the susceptibility of the causative organism (see Table 2).
| a Since ZINACEF is dialyzable, patients on hemodialysis should be given a further dose at the end of the dialysis. | ||
|
Creatinine Clearance (mL/min) |
Dose |
Frequency |
|
>20 |
750 mg-1.5 grams |
q8h |
|
10-20 |
750 mg |
q12h |
|
<10 |
750 mg |
q24ha |
When only serum creatinine is available, the following formula2 (based on sex, weight, and age of the patient) may be used to convert this value into creatinine clearance. The serum creatinine should represent a steady state of renal function.
|
Males: Creatinine clearance (mL/min) = |
Weight (kg) x (140 - age) 72 x serum creatinine (mg/dL) |
Females: 0.85 x male value
Note: As with antibiotic therapy in general, administration of ZINACEF should be continued for a minimum of 48 to 72 hours after the patient becomes asymptomatic or after evidence of bacterial eradication has been obtained; a minimum of 10 days of treatment is recommended in infections caused by Streptococcus pyogenes in order to guard against the risk of rheumatic fever or glomerulonephritis; frequent bacteriologic and clinical appraisal is necessary during therapy of chronic urinary tract infection and may be required for several months after therapy has been completed; persistent infections may require treatment for several weeks; and doses smaller than those indicated above should not be used. In staphylococcal and other infections involving a collection of pus, surgical drainage should be carried out where indicated.
Pediatric Patients Above 3 Months of Age
Administration of 50 to 100 mg/kg/day in equally divided doses every 6 to 8 hours has been successful for most infections susceptible to cefuroxime. The higher dosage of 100 mg/kg/day (not to exceed the maximum adult dosage) should be used for the more severe or serious infections.
In bone and joint infections, 150 mg/kg/day (not to exceed the maximum adult dosage) is recommended in equally divided doses every 8 hours. In clinical trials, a course of oral antibiotics was administered to pediatric patients following the completion of parenteral administration of ZINACEF.
In cases of bacterial meningitis, a larger dosage of ZINACEF is recommended, 200 to 240 mg/kg/day intravenously in divided doses every 6 to 8 hours.
In pediatric patients with renal insufficiency, the frequency of dosing should be modified consistent with the recommendations for adults.
Preparation of Solution and Suspension
The directions for preparing ZINACEF for both IV and IM use are summarized in Table 3.
For Intramuscular Use
Each 750-mg vial of ZINACEF should be constituted with 3.0 mL of Sterile Water for Injection. Shake gently to disperse and withdraw completely the resulting suspension for injection.
For Intravenous Use
Each 750-mg vial should be constituted with 8.3 mL of Sterile Water for Injection. Withdraw completely the resulting solution for injection.
Each 1.5-gram vial should be constituted with 16.0 mL of Sterile Water for Injection, and the solution should be completely withdrawn for injection.
The 7.5-gram pharmacy bulk vial should be constituted with 77 mL of Sterile Water for Injection; each 8 mL of the resulting solution contains 750 mg of cefuroxime.
|
Strength |
Amount of Diluent to Be Added (mL) |
Volume to Be Withdrawn |
Approximate Cefuroxime Concentration (mg/mL) |
|
750-mg Vial |
3.0 (IM) |
Totala |
225 |
|
750-mg Vial |
8.3 (IV) |
Total |
90 |
|
1.5-gram Vial |
16.0 (IV) |
Total |
90 |
|
7.5-gram Pharmacy bulk package |
77 (IV) |
Amount Neededb |
95 |
a NOTE: ZINACEF is a suspension at IM concentrations.
b 8 mL of solution contains 750 mg of cefuroxime; 16 mL contains 1.5 grams of cefuroxime.
Administration
After constitution, ZINACEF may be given intravenously or by deep IM injection into a large muscle mass (such as the gluteus or lateral part of the thigh). Before injecting intramuscularly, aspiration is necessary to avoid inadvertent injection into a blood vessel.
Intravenous Administration
The IV route may be preferable for patients with bacterial septicemia or other severe or life-threatening infections or for patients who may be poor risks because of lowered resistance, particularly if shock is present or impending.
For direct intermittent IV administration, slowly inject the solution into a vein over a period of 3 to 5 minutes or give it through the tubing system by which the patient is also receiving other IV solutions.
For intermittent IV infusion with a Y-type administration set, dosing can be accomplished through the tubing system by which the patient may be receiving other IV solutions. However, during infusion of the solution containing ZINACEF, it is advisable to temporarily discontinue administration of any other solutions at the same site.
ADD-Vantage vials are to be constituted only with 50 or 100 mL of 5% Dextrose Injection, 0.9% Sodium Chloride Injection, or 0.45% Sodium Chloride Injection in Abbott ADD-Vantage flexible diluent containers (see Instructions for Constitution). ADD-Vantage vials that have been joined to Abbott ADD-Vantage diluent containers and activated to dissolve the drug are stable for 24 hours at room temperature or for 7 days under refrigeration. Joined vials that have not been activated may be used within a 14-day period; this period corresponds to that for use of Abbott ADD-Vantage containers following removal of the outer packaging (overwrap).
Freezing solutions of ZINACEF in the ADD-Vantage system is not recommended.
For continuous IV infusion, a solution of ZINACEF may be added to an IV infusion pack containing one of the following fluids: 0.9% Sodium Chloride Injection; 5% Dextrose Injection; 10% Dextrose Injection; 5% Dextrose and 0.9% Sodium Chloride Injection; 5% Dextrose and 0.45% Sodium Chloride Injection; or 1/6 M Sodium Lactate Injection.
Solutions of ZINACEF, like those of most beta-lactam antibiotics, should not be added to solutions of aminoglycoside antibiotics because of potential interaction.
However, if concurrent therapy with ZINACEF and an aminoglycoside is indicated, each of these antibiotics can be administered separately to the same patient.
Directions for Use of ZINACEF Frozen in Galaxy® Plastic Containers
ZINACEF supplied as a frozen, sterile, iso-osmotic, nonpyrogenic solution in plastic containers is to be administered after thawing either as a continuous or intermittent IV infusion. The thawed solution of the premixed product is stable for 28 days if stored under refrigeration (5°C) or for 24 hours if stored at room temperature (25°C). Do not refreeze.
Thaw container at room temperature (25°C) or under refrigeration (5°C). Do not force thaw by immersion in water baths or by microwave irradiation. Components of the solution may precipitate in the frozen state and will dissolve upon reaching room temperature with little or no agitation. Potency is not affected. Mix after solution has reached room temperature. Check for minute leaks by squeezing bag firmly. Discard bag if leaks are found as sterility may be impaired. Do not add supplementary medication. Do not use unless solution is clear and seal is intact.
Use sterile equipment.
COMPATIBILITY AND STABILITY
Intramuscular
When constituted as directed with Sterile Water for Injection, suspensions of ZINACEF for IM injection maintain satisfactory potency for 24 hours at room temperature and for 48 hours under refrigeration (5°C).
After the periods mentioned above any unused suspensions should be discarded.
Intravenous
When the 750-mg, 1.5-g, and 7.5-g pharmacy bulk vials are constituted as directed with Sterile Water for Injection, the solutions of ZINACEF for IV administration maintain satisfactory potency for 24 hours at room temperature and for 48 hours (750-mg and 1.5-g vials) or for 7 days (7.5-g pharmacy bulk vial) under refrigeration (5°C). More dilute solutions, such as 750 mg or 1.5 g plus 100 mL of Sterile Water for Injection, 5% Dextrose Injection, or 0.9% Sodium Chloride Injection, also maintain satisfactory potency for 24 hours at room temperature and for 7 days under refrigeration.
These solutions may be further diluted to concentrations of between 1 and 30 mg/mL in the following solutions and will lose not more than 10% activity for 24 hours at room temperature or for at least 7 days under refrigeration: 0.9% Sodium Chloride Injection; 1/6 M Sodium Lactate Injection; Ringer's Injection, USP; Lactated Ringer's Injection, USP; 5% Dextrose and 0.9% Sodium Chloride Injection; 5% Dextrose Injection; 5% Dextrose and 0.45% Sodium Chloride Injection; 5% Dextrose and 0.225% Sodium Chloride Injection; 10% Dextrose Injection; and 10% Invert Sugar in Water for Injection.
Unused solutions should be discarded after the time periods mentioned above.
ZINACEF has also been found compatible for 24 hours at room temperature when admixed in IV infusion with heparin (10 and 50 U/mL) in 0.9% Sodium Chloride Injection and Potassium Chloride (10 and 40 mEq/L) in 0.9% Sodium Chloride Injection. Sodium Bicarbonate Injection, USP is not recommended for the dilution of ZINACEF.
The 750-mg and 1.5-g ZINACEF ADD-Vantage vials, when diluted in 50 or 100 mL of 5% Dextrose Injection, 0.9% Sodium Chloride Injection, or 0.45% Sodium Chloride Injection, may be stored for up to 24 hours at room temperature or for 7 days under refrigeration.
Frozen Stability
Constitute the 750-mg, 1.5-g, or 7.5-g vial as directed for IV administration in Table 3. Immediately withdraw the total contents of the 750-mg or 1.5-g vial or 8 or 16 mL from the 7.5-g bulk vial and add to a Baxter VIAFLEX®MINI-BAG™ containing 50 or 100 mL of 0.9% Sodium Chloride Injection or 5% Dextrose Injection and freeze. Frozen solutions are stable for 6 months when stored at -20°C. Frozen solutions should be thawed at room temperature and not refrozen. Do not force thaw by immersion in water baths or by microwave irradiation. Thawed solutions may be stored for up to 24 hours at room temperature or for 7 days in a refrigerator.
Note: Parenteral drug products should be inspected visually for particulate matter and discoloration before administration whenever solution and container permit.
As with other cephalosporins, ZINACEF powder as well as solutions and suspensions tend to darken, depending on storage conditions, without adversely affecting product potency.
Directions for Dispensing
Pharmacy Bulk Package—Not for Direct Infusion
The pharmacy bulk package is for use in a pharmacy admixture service only under a laminar flow hood. Entry into the vial must be made with a sterile transfer set or other sterile dispensing device, and the contents dispensed in aliquots using aseptic technique. The use of syringe and needle is not recommended as it may cause leakage (see DOSAGE AND ADMINISTRATION). AFTER INITIAL WITHDRAWAL USE ENTIRE CONTENTS OF VIAL PROMPTLY. ANY UNUSED PORTION MUST BE DISCARDED WITHIN 24 HOURS.
HOW SUPPLIED
ZINACEF in the dry state should be stored between 15° and 30°C (59° and 86°F) and protected from light. ZINACEF is a dry, white to off-white powder supplied in vials as follows:
NDC 0173-0352-10 750-mg* Vial (Tray of 10)
NDC 0173-0354-10 1.5-g* Vial (Tray of 10)
NDC 0173-0400-00 7.5-g* Pharmacy Bulk Package (Tray of 6)
NDC 0173-0436-00 750-mg ADD-Vantage Vial (Tray of 25)
NDC 0173-0437-00 1.5-g ADD-Vantage Vial (Tray of 10)
(The above ADD-Vantage vials are to be used only with Abbott ADD-Vantage diluent containers.)
ZINACEF frozen as a premixed solution of cefuroxime injection should not be stored above -20°C. ZINACEF is supplied frozen in 50-mL, single-dose, plastic containers as follows:
NDC 0173-0424-00 750-mg* Plastic Container (Carton of 24)
NDC 0173-0425-00 1.5-g* Plastic Container (Carton of 24)
*Equivalent to cefuroxime.
REFERENCES
- 1.
- National Committee for Clinical Laboratory Standards. Performance Standards for Antimicrobial Susceptibility Testing. Third Informational Supplement. NCCLS Document M100-S3, Vol. 11, No. 17. Villanova, Pa: NCCLS; 1991.
- 2.
- Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16:31-41.
ZINACEF® (cefuroxime for injection):
GlaxoSmithKline
Research Triangle Park, NC 27709
ZINACEF® (cefuroxime injection):
Manufactured for GlaxoSmithKline
Research Triangle Park, NC 27709
by Baxter Healthcare Corporation, Deerfield, IL 60015
ZINACEF is a registered trademark of GlaxoSmithKline.
ADD-Vantage is a registered trademark of Hospira, Inc.
CLINITEST is a registered trademark of Ames Division, Miles Laboratories, Inc.
GALAXY and VIAFLEX are registered trademarks of Baxter International Inc.
MINI-BAG is a trademark of Baxter International Inc.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2010, GlaxoSmithKline. All rights reserved.
August 2010 ZNF:2PI
-----------------------------------------------------------------------------------------------------------------
Tear Away
ZINACEF®
(cefuroxime for injection)
Instructions for Constitution of ADD-Vantage® Vials
To Open Diluent Container:
Peel the corner of the ADD-Vantage diluent overwrap and remove flexible diluent container. Some opacity of the plastic flexible container due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
To Assemble Vial and Flexible DiluentContainer(Use Aseptic Technique):
1. Remove the protective covers from the top of the vial and the vial port on the diluent container as follows:
a. To remove the breakaway vial cap, swing the pull ring over the top of the vial and pull down far enough to start the opening (see Figure 1), then pull straight up to remove the cap (see Figure 2). Note: Once the breakaway cap has been removed, do not access vial with syringe.
 Figure 1 | 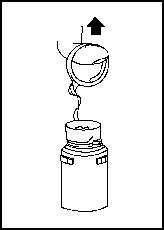 Figure 2 |
b. To remove the vial port cover, grasp the tab on the pull ring, pull up to break the 3 tie strings, then pull back to remove the cover (see Figure 3).
2. Screw the vial into the vial port until it will go no further. THE VIAL MUST BE SCREWED IN TIGHTLY TO ASSURE A SEAL. This occurs approximately one-half turn (180°) after the first audible click (see Figure 4). The clicking sound does not assure a seal; the vial must be turned as far as it will go. Note: Once vial is seated, do not attempt to remove (see Figure 4).
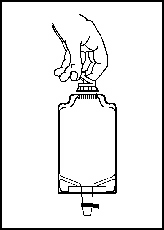 Figure 3 | 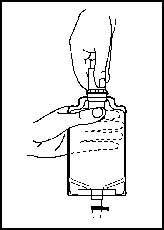 Figure 4 |
3. Recheck the vial to assure that it is tight by trying to turn it further in the direction of assembly.
4. Label appropriately.
To Prepare Admixture:
1. Squeeze the bottom of the diluent container gently to inflate the portion of the container surrounding the end of the drug vial.
2. With the other hand, push the drug vial down into the container, telescoping the walls of the container. Grasp the inner cap of the vial through the walls of the container (see Figure 5).
3. Pull the inner cap from the drug vial (see Figure 6). Verify that the rubber stopper has been pulled out, allowing the drug and diluent to mix.
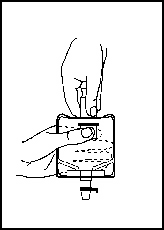 Figure 5 | 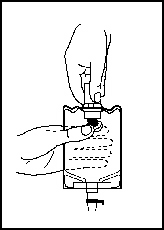 Figure 6 |
4. Mix container contents thoroughly and use within the specified time.
Preparation for Administration (Use Aseptic Technique):
1. Confirm the activation and admixture of vial contents.
2. Check for leaks by squeezing container firmly. If leaks are found, discard unit as sterility may be impaired.
3. Close flow control clamp of administration set.
4. Remove cover from outlet port at bottom of container.
5. Insert piercing pin of administration set into port with a twisting motion until the pin is firmly seated. Note: See full directions on administration set carton.
6. Lift the free end of the hanger loop on the bottom of the vial, breaking the 2 tie strings. Bend the loop outward to lock it in the upright position, then suspend container from hanger.
7. Squeeze and release drip chamber to establish proper fluid level in chamber.
8. Open flow control clamp and clear air from set. Close clamp.
9. Attach set to venipuncture device. If device is not indwelling, prime and make venipuncture.
10. Regulate rate of administration with flow control clamp.
WARNING: Do not use flexible container in series connections.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2010, GlaxoSmithKline. All rights reserved.
August 2010 ZNF:2DIR
Principal Display Panel
NDC 0173-0352-10
Zinacef®
(cefuroxime for injection)
750 mg
Equivalent to 750 mg of Cefuroxime Activity
For IM or IV use.
Rx only
STORE UPRIGHT.
See accompanying prescribing information for Dosage and Administration.
Store between 15o and 30oC (59o and 86oF). Protect from light.
To prepare IM suspension, add 3.0 mL of Sterile Water for Injection. Shake gently to disperse and withdraw completely the resulting suspension for injection. The resulting concentration is approximately 225 mg/mL
To prepare IV solution, add 8.3 mL of Sterile Water for Injection. Shake until dissolved and withdraw completely the resulting solution for injection.
After constitution, the suspension and solution maintain potency for 24 hours at room temperature or 48 hours under refrigeration (5oC). Color changes do not affect potency.
GlaxoSmithKline, RTP, NC 27709
Made in England
Rev. 12/09
10000000076134
Principal Display Panel
NDC 0173-0354-10
Zinacef®
(cefuroxime for injection)
1.5 g
Equivalent to 1.5 g of Cefuroxime Activity.
For IV use.
Rx only
See prescribing information for Dosage and Administration.
Store between 15o and 30oC (59o and 86oF). Protect from light.
To prepare IV solution, add 16.0 mL of Sterile Water for Injection. Shake until dissolved and withdraw completely for injection.
After constitution, the solution maintains potency for 24 hours at room temperature or 48 hours under refrigeration (5oC). Color changes in solution do not affect potency.
GlaxoSmithKline, RTP, NC 27709
Made in England
Rev. 12/09
10000000076137
Principal Display Panel
NDC 0173-0400-00
Zinacef®
(cefuroxime for injection)
7.5 g
Equivalent to 7.5 g of Cefuroxime Activity.
Pharmacy Bulk Package – Not for Direct Infusion
Rx only
See accompanying prescribing information for Dosage and Administration.
Store between 15o and 30oC (59o and 86oF). Protect from light.
To prepare solution, add 77 mL of Sterile Water for Injection. Shake well to dissolve. The constituted solution occupies a volume of about 82.5 mL and contains approximately 750 mg of cefuroxime activity per 8 mL.
After constitution, the solution maintains potency for 24 hours at room temperature or 7 days under refrigeration (5°C). Dispense aliquots from the vial via a suitable dispensing device into infusion fluids under a laminar flow hood using aseptic technique.
DISCARD VIAL WITHIN 24 HOURS AFTER INITIAL WITHDRAWAL.
GlaxoSmithKline
Research Triangle Park, NC 27709
Made in England
Rev. 12/09
10000000076190
Principal Display Panel
NDC 0173-0436-00
Zinacef®
(cefuroxime for injection)
ADD-Vantage® Vial
750 mg
Equivalent to 750 mg of Cefuroxime Activity.
For IV use.
Rx only
See accompanying prescribing information for Dosage and Administration and Instructions for Constitution.
Before constitution, store between 15o and 30oC (59o and 86oF) and protect from light.
Color changes do not affect potency.
GlaxoSmithKline
RTP, NC 27709
Made in England
*ADD-Vantage® is a trademark of Hospira, Inc.
Rev. 9/10
10000000084188
Principal Display Panel
NDC 0173-0437-00
Zinacef®
(cefuroxime for injection)
ADD-Vantage® Vial
1.5 g
Equivalent to 1.5 g of Cefuroxime Activity.
For IV use.
Rx only
See accompanying prescribing information for Dosage and Administration and Instructions for Constitution.
Before constitution, store between 15o and 30oC (59o and 86oF) and protect from light.
Color changes do not affect potency.
GlaxoSmithKline
RTP, NC 27709
Made in England
*ADD-Vantage® is a trademark of Hospira, Inc.
Rev. 9/10
10000000084262
Principal Display Panel
NDC 0173-0424-00
Zinacef®
(cefuroxime injection)
750 mg
Galaxy
Single-Dose Container
50 mL
Iso-osmotic
Code 2G3546
Sterile, Nonpyrogenic
Each 50 mL contains: cefuroxime sodium equivalent to 750 mg of cefuroxime with approx. 1.4 g of dextrose hydrous, USP added to adjust osmolality and 300 mg of sodium citrate hydrous, USP added as a buffer. pH adjusted with hydrochloric acid and may have been adjusted with sodium hydroxide. pH 5.0 to 7.5.
Dosage: Intravenously as directed by a physician. See accompanying prescribing information.
Cautions: Do not add supplementary medication. Must not be used in series connections. Check for minute leaks and solution clarity.
Rx only
Do not store above -20oC. Thaw at room temperature (25oC) or under refrigeration (5oC). DO NOT FORCE THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION. Thawed solution is stable for 28 days under refrigeration or 24 hours at room temperature. Do not refreeze.
Zinacef is a registered trademark of GlaxoSmithKline
Galaxy is a registered trademark of Baxter International Inc.
Manufactured for GlaxoSmithKline
Research Triangle Park, NC 27709
By Baxter Healthcare Corporation, Deerfield, IL 60015 USA
Made in England
PL 2040 Plastic
7-34-61-893
Rev. 1/10
10000000076245
Principal Display Panel
NDC 0173-0425-00
Zinacef®
(cefuroxime injection)
1.5 g
Galaxy
Single-Dose Container
50 mL
Iso-osmotic
Code 2G3547
Sterile, Nonpyrogenic
Each 50 mL contains: cefuroxime sodium equivalent to 1.5 g of cefuroxime with 600 mg of sodium citrate hydrous, USP added as a buffer. pH adjusted with hydrochloric acid and may have been adjusted with sodium hydroxide. pH 5.0 to 7.5.
Dosage: Intravenously as directed by a physician. See accompanying prescribing information.
Cautions: Do not add supplementary medication. Must not be used in series connections. Check for minute leaks and solution clarity.
Rx only
Do not store above -20oC. Thaw at room temperature (25oC) or under refrigeration (5oC). DO NOT FORCE THAW BY IMMERSION IN WATER BATHS OR BY MICROWAVE IRRADIATION. Thawed solution is stable for 28 days under refrigeration or 24 hours at room temperature. Do not refreeze.
Zinacef is a registered trademark of GlaxoSmithKline
Galaxy is a registered trademark of Baxter International Inc.
Manufactured for GlaxoSmithKline
Research Triangle Park, NC 27709
By Baxter Healthcare Corporation, Deerfield, IL 60015 USA
Made in England
PL 2040 Plastic
7-34-61-894
Rev. 1/10
10000000076247
| ZINACEF
cefuroxime injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, powder, for solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| ZINACEF
cefuroxime injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - GlaxoSmithKline LLC (167380711) |