SMART SENSE DAYTIME NIGHTTIME COLD AND FLU- acetaminophen, dextromethorphan hbr, doxylamine succinate, guaifenesin, phenylephrine hcl
Kmart Corporation
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Kmart Corporation Daytime Nighttime Cold & Flu Drug Facts
Nighttime Severe Cold & Flu
Active ingredients (in each 30 mL)
Acetaminophen 650 mg
Dextromethorphan HBr 20 mg
Doxylamine succinate 12.5 mg
Phenylephrine HCl 10 mg
Uses
temporarily relieves common cold/flu symptoms:
- •
- nasal congestion
- •
- sinus congestion and pressure
- •
- minor aches and pains
- •
- headache
- •
- fever
- •
- sore throat
- •
- runny nose and sneezing
- •
- cough due to minor throat and bronchial irritation
- •
- cough to help you sleep
- •
- reduces swelling of nasal passages
- •
- promotes nasal and/or sinus drainage
- •
- temporarily restores freer breathing through the nose
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- •
- more than 4,000 mg of acetaminophen in 24 hours
- •
- with other drugs containing acetaminophen
- •
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- •
- skin reddening
- •
- blisters
- •
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- •
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- •
- liver disease
- •
- heart disease
- •
- high blood pressure
- •
- thyroid disease
- •
- diabetes
- •
- glaucoma
- •
- cough that occurs with too much phlegm (mucus)
- •
- a breathing problem such as emphysema or chronic bronchitis
- •
- trouble urinating due to an enlarged prostate gland
- •
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Ask a doctor or pharmacist before use if you are
- •
- taking sedatives or tranquilizers
- •
- taking the blood thinning drug warfarin
When using this product
- •
- do not use more than directed
- •
- excitability may occur, especially in children
- •
- marked drowsiness may occur
- •
- avoid alcoholic drinks
- •
- be careful when driving a motor vehicle or operating machinery
- •
- alcohol, sedatives, and tranquilizers may increase drowsiness
Stop use and ask a doctor if
- •
- you get nervous, dizzy or sleepless
- •
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- •
- fever gets worse or lasts more than 3 days
- •
- redness or swelling is present
- •
- new symptoms occur
- •
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
- •
- take only as directed - see Overdose warning
- •
- only use the dose cup provided
- •
- do not exceed 4 doses per 24 hrs
|
adults & children 12 yrs & over |
30 mL every 4 hrs |
|
children 4 to under 12 yrs |
ask a doctor |
|
children under 4 yrs |
do not use |
Inactive ingredients
butylated hydroxyanisole, edetate disodium, FD&C blue #1, FD&C red #40, flavor, glycerin, monobasic sodium phosphate, polyethylene glycol, propylene glycol, purified water, saccharin sodium, sucrose, xanthan gum
Daytime Severe Cold & Flu
Active ingredients (in each 15 mL)
Acetaminophen 325 mg
Dextromethorphan HBr 10 mg
Guaifenesin 200 mg
Phenylephrine HCl 5 mg
Uses
temporarily relieves common cold/flu symptoms:
- •
- nasal congestion
- •
- sinus congestion and pressure
- •
- cough due to minor throat and bronchial irritation
- •
- minor aches and pains
- •
- headache
- •
- fever
- •
- sore throat
- •
- reduces swelling of nasal passages
- •
- temporarily restores freer breathing through the nose
- •
- promotes nasal and/or sinus drainage
- •
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if
- •
- adult takes more than 4,000 mg of acetaminophen in 24 hours
- •
- child takes more than 5 doses in 24 hours
- •
- taken with other drugs containing acetaminophen
- •
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- •
- skin reddening
- •
- blisters
- •
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- •
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- •
- liver disease
- •
- heart disease
- •
- thyroid disease
- •
- diabetes
- •
- high blood pressure
- •
- trouble urinating due to an enlarged prostate gland
- •
- cough that occurs with too much phlegm (mucus)
- •
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
Stop use and ask a doctor if
- •
- you get nervous, dizzy or sleepless
- •
- pain, nasal congestion or cough gets worse or lasts more than 5 days (children) or 7 days (adults)
- •
- fever gets worse or lasts more than 3 days
- •
- redness or swelling is present
- •
- new symptoms occur
- •
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
Keep out of reach of children.
Overdose warning: In care of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Directions
- •
- take only as directed - see Overdose warning
- •
- only use the dose cup provided
- •
- do not exceed 4 doses per 24 hrs
|
adults & children 12 yrs & over |
30 mL every 4 hrs |
|
children 6 to under 12 yrs |
15 mL every 4 hrs |
|
children 4 to under 6 yrs |
ask a doctor |
|
children under 4 yrs |
do not use |
Other information
- •
- each 15 mL contains: sodium 6 mg
- •
- store at 20-25°C (68-77°F). Do not refrigerate.
Inactive ingredients
butylated hydroxyanisole, edetate disodium, FD&C yellow #6, flavor, glycerin, menthol, monobasic sodium phosphate, polyethylene glycol, propylene glycol, purified water, saccharin sodium, sucrose, xanthan gum
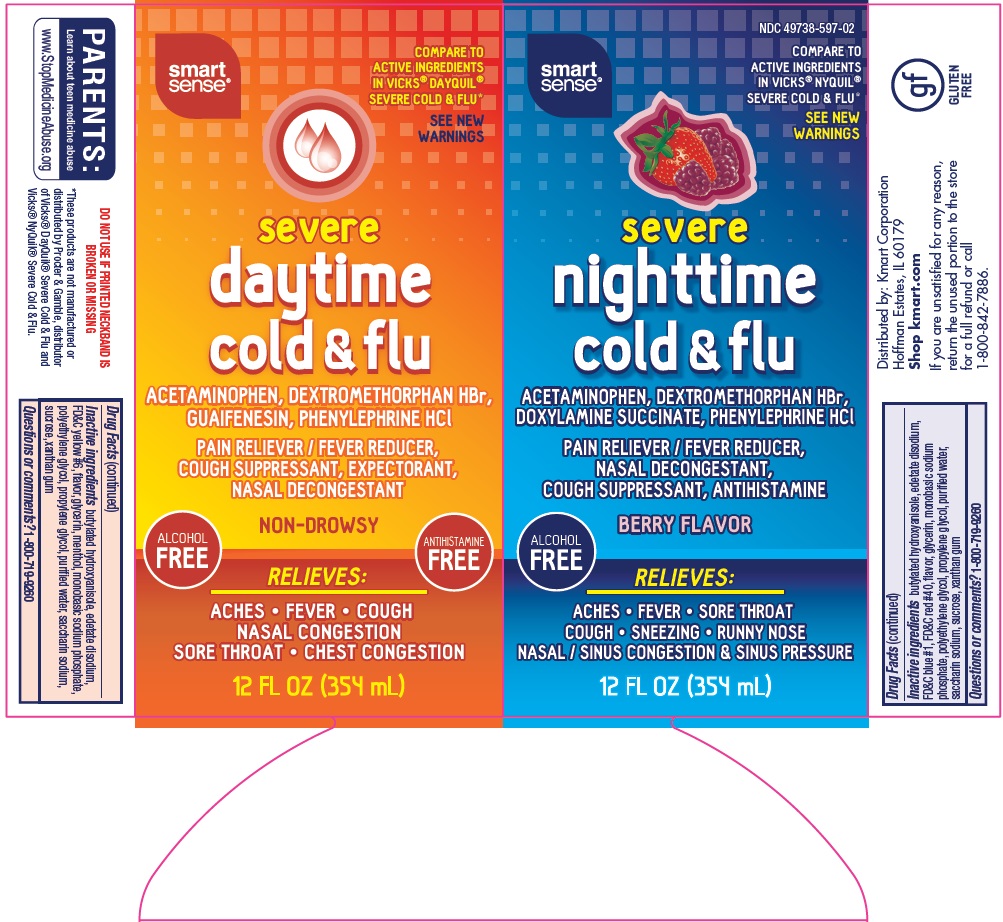
Principal Display Panel
COMPARE TO ACTIVE INGREDIENTS IN VICKS® DAYQUIL® SEVERE COLD & FLU
SEE NEW WARNINGS
severe
daytime
cold & flu
ACETAMINOPHEN, DEXTROMETHORPHAN HBr, GUAIFENESIN, PHENYLEPHRINE HCl
PAIN RELIEVER / FEVER REDUCER, COUGH SUPPRESSANT, EXPECTORANT, NASAL DECONGESTANT
NON-DROWSY
ALCOHOL FREE
ANTIHISTAMINE FREE
RELIEVES:
ACHES – FEVER – COUGH
NASAL CONGESTION
SORE THROAT – CHEST CONGESTION
12 FL OZ (354 mL)
COMPARE TO ACTIVE INGREDIENTS IN VICKS® NYQUIL® SEVERE COLD & FLU
SEE NEW WARNINGS
severe
nighttime
cold & flu
ACETAMINOPHEN, DEXTROMETHORPHAN HBr, DOXYLAMINE SUCCINATE, PHENYLEPHRINE HCl
PAIN RELIEVER / FEVER REDUCER,
NASAL DECONGESTANT, COUGH SUPPRESSANT, ANTIHISTAMINE
BERRY FLAVOR
ALCOHOL FREE
RELIEVES:
ACHES – FEVER – SORE THROAT
COUGH – SNEEZING – RUNNY NOSE
NASAL / SINUS CONGESTION & SINUS PRESSURE
12 FL OZ (354 mL)
| SMART SENSE DAYTIME NIGHTTIME COLD AND FLU
acetaminophen, dextromethorphan hbr, doxylamine succinate, guaifenesin, phenylephrine hcl kit |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Kmart Corporation (008965873) |