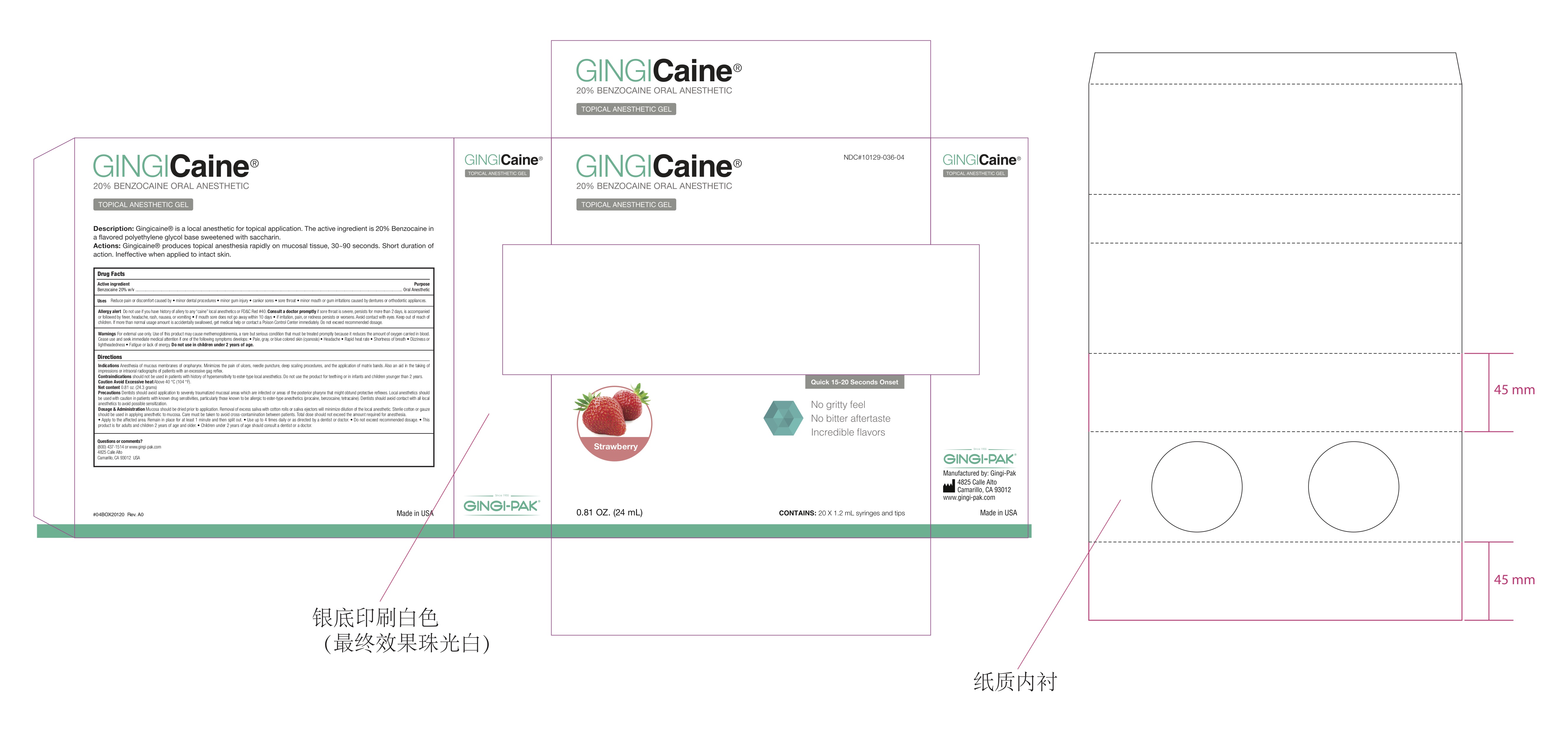
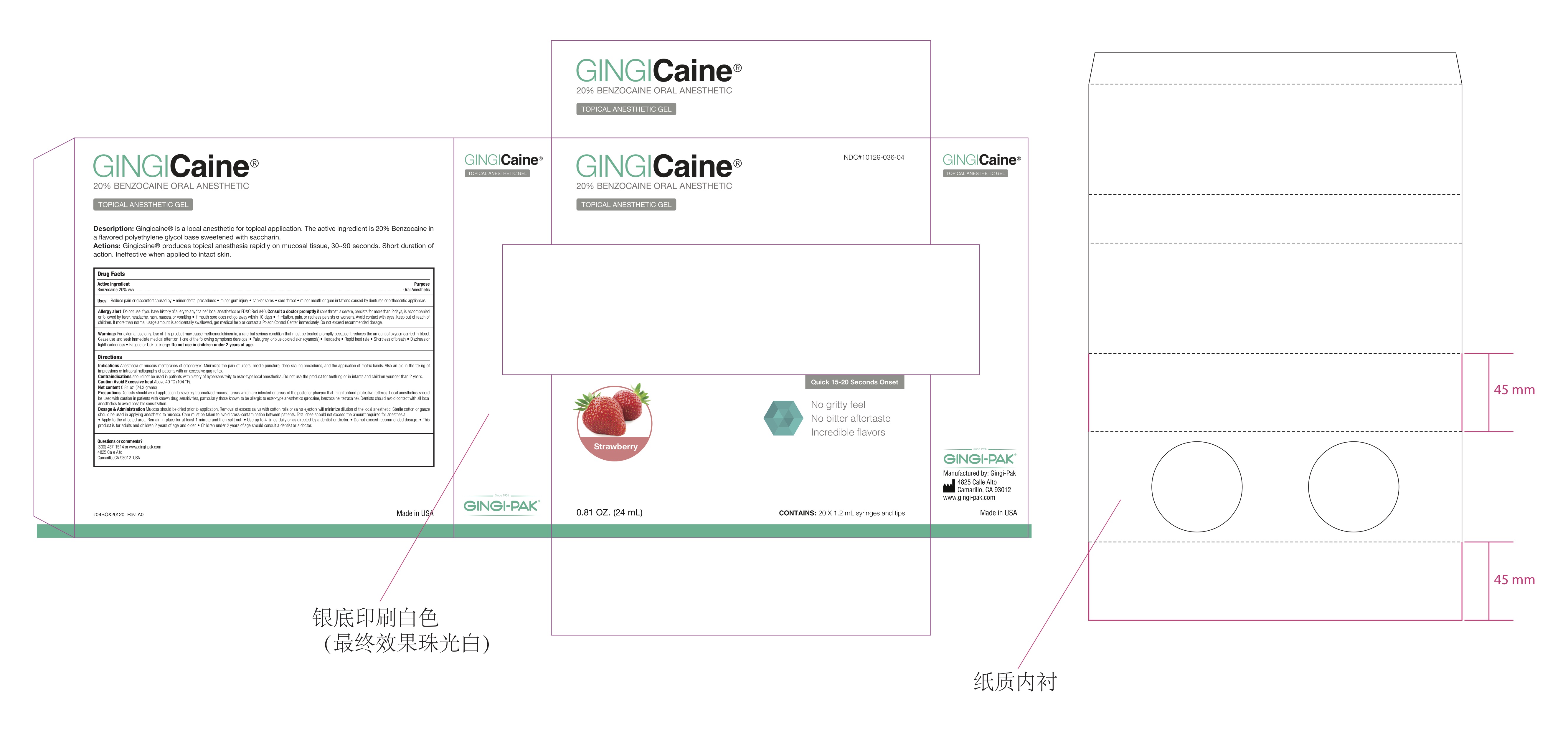
Label: GINGICAINE GEL STRAWBERRY- gingicaine gel strawberry flavor gel
- NDC Code(s): 10129-036-01, 10129-036-04, 10129-036-05
- Packager: Gingi-Pak a Division of the Belport
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Dosage and Administration
- Precaution Section
- Indications and usage section
- Active Ingredient - Benzocaine, 20%
- Warnings
- Consult a doctor promtly
- Do not use
- Avoid excessive heat
- Inactive ingredients
- Purpose
- Keep out of reach of children
- Strawberry Label USA
- GingiCaine Gel Syringe label
- GingiCAINE in Syringe Box
-
INGREDIENTS AND APPEARANCE
GINGICAINE GEL STRAWBERRY
gingicaine gel strawberry flavor gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10129-036 Route of Administration ORAL, DENTAL, PERIODONTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg Inactive Ingredients Ingredient Name Strength POTASSIUM SODIUM SACCHARATE (UNII: 73U34YC90U) 20 mg POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) 520 mg POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) 260 mg Product Characteristics Color red (Strawberry Flavor) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10129-036-01 1 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/06/1993 2 NDC:10129-036-04 20 in 1 SYRINGE, PLASTIC; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) 01/11/2018 3 NDC:10129-036-05 6 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/06/1993 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M022 07/06/1993 Labeler - Gingi-Pak a Division of the Belport (008480121)