NIGHTTIME COUGH- dextromethorphan hydrobromide, doxylamine succinate liquid
HyVee Inc
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Hy-Vee, Inc. Night Time Cough Drug Facts
Warnings
Do not use
- •
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- •
- to make a child sleepy
Ask a doctor before use if you have
- •
- glaucoma
- •
- cough that occurs with too much phlegm (mucus)
- •
- a breathing problem such as emphysema or chronic bronchitis
- •
- persistent or chronic cough as occurs with smoking, asthma, or emphysema
- •
- trouble urinating due to an enlarged prostate gland
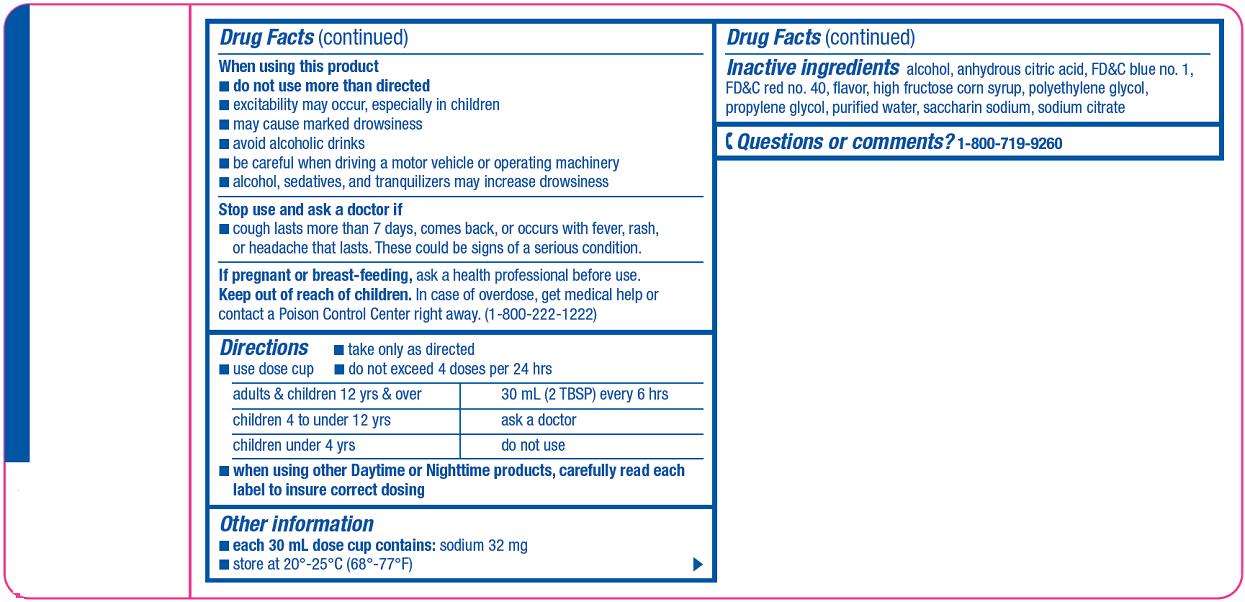
When using this product
- •
- do not use more than directed
- •
- excitability may occur, especially in children
- •
- may cause marked drowsiness
- •
- avoid alcoholic drinks
- •
- be careful when driving a motor vehicle or operating machinery
- •
- alcohol, sedatives, and tranquilizers may increase drowsiness
Directions
- •
- take only as directed
- •
- use dose cup
- •
- do not exceed 4 doses per 24 hrs
|
adults & children 12 yrs & over |
30 mL (2 TBSP) every 6 hrs |
|
children 4 to under 12 yrs |
ask a doctor |
|
children under 4 yrs |
do not use |
- •
- when using other Daytime or Nighttime products, carefully read each label to insure correct dosing
| NIGHTTIME COUGH
dextromethorphan hydrobromide, doxylamine succinate liquid |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - HyVee Inc (006925671) |
Revised: 11/2020
Document Id: 104ad645-65b1-44e7-bf6f-4f4f9612d48f
Set id: 264b6d1d-5e11-4862-b1c4-7b9dea812901
Version: 8
Effective Time: 20201124
HyVee Inc