PHENYLEPHRINE HYDROCHLORIDE OPHTHALMIC- phenylephrine hydrochloride solution/ drops
HUB Pharmaceuticals, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Sterile Phenylephrine Hydrochloride Ophthalmic Solution 2.5% USP Rx Only
WARNING:
PHYSICIANS SHOULD COMPLETELY FAMILIARIZE THEMSELVES WITH THE COMPLETE CONTENTS OF THIS LEAFLET BEFORE PRESCRIBING PHENYLEPHRINE HYDROCHLORIDE OPHTHALMIC SOLUTION.
DESCRIPTION:
Phenylephrine Hydrochloride Ophthalmic Solution is a sterile solution used as a vasoconstrictor and mydriatic for use in ophthalmology.
Phenylephrine hydrochloride is a synthetic sympathomimetic compound structurally similar to epinephrine and ephedrine. Phenylephrine hydrochloride has the following structural formula:
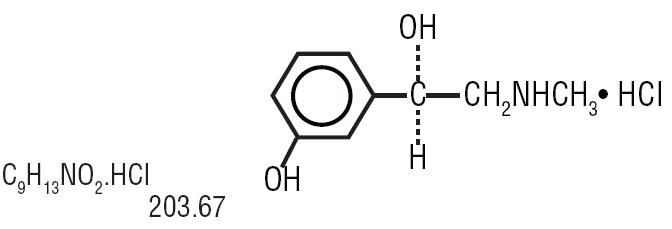
(-)- m-Hydroxy-α-[(methylamino)methyl] benzyl alcohol hydrochloride Phenylephrine Hydrochloride Ophthalmic Solution is available in two strengths, 2.5% and 10%.
Each mL of the 2.5% solution contains: Phenylephrine Hydrochloride 2.5%. Dibasic and Monobasic Sodium Phosphates, Boric Acid, with Benzalkonium Chloride as preservative in Water for Injection q.s.
Each mL of the 10% solution contains: Phenylephrine
Hydrochloride 10%, Dibasic and Monobasic Sodium Phosphates with Benzalkonium
Chloride as preservative in Water for Injection q.s.
Hydrochloric Acid and/or Sodium Hydroxide may have been used to adjust pH of both solutions.
CLINICAL PHARMACOLOGY:
Phenylephrine Hydrochloride Ophthalmic Solution possesses predominantly (α-adrenergic effects. In the eye, phenylephrine acts locally as a potent vasoconstrictor and mydriatic, by constricting ophthalmic blood vessels and radial muscle of the iris.
The ophthalmologic usefulness of phenylephrine hydrochloride is due to its rapid effect and moderately prolonged action, as well as to the fact that it produces no compensatory vasodilatation.
The action of different concentrations of ophthalmic solutions of phenylephrine hydrochloride is shown in the following table:
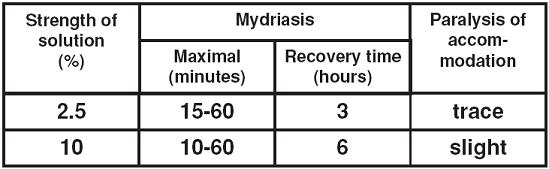
Although rare, systemic absorption of sufficient quantities of phenylephrine may lead to system (α-adrenergic effects, such as rise in blood pressure which may be accompanied by a reflex atropine-sensitive bradycardia.
INDICATIONS AND USAGE:
Phenylephrine hydrochloride is recommended for use as a decongestant and vasoconstrictor and for pupil dilation in uveitis (posterior synechiae). Wide angle glaucoma, prior to surgery, refraction, ophthalmoscopic examination, and diagnostic procedures.
CONTRAINDICATIONS:
Ophthalmic solutions of phenylephrine hydrochloride are contraindicated in persons with narrow angle glaucoma (and in those individuals who are hypersensitive to phenylephrine or to any of the components of the product). Phenylephrine Hydrochloride Ophthalmic Solution in the 10% strength is contraindicated in infants and in patients with aneurysms.
WARNINGS:
There have been rare reports associating the use of Phenylephrine Hydrochloride Ophthalmic Solution 10%, with the development of serious cardiovascular reactions, including ventricular arrhythmias and myocardial infarctions. These episodes, some ending fatally, have usually occurred in elderly patients with pre-existing cardiovascular diseases.
PRECAUTIONS:
General:
Exceeding recommended dosages or applying Phenylephrine Hydrochloride Ophthalmic Solution to the instrumented, traumatized, diseased or post surgical eye or adnexa, or to patients with suppressed lacrimation, as during anesthesia, any result in the absorption of sufficient quantities of phenylephrine to produce a systemic vasopressor response.
A significant elevation in blood pressure is rare but has been reported following conjunctional instillation of recommended doses of Phenylephrine Hydrochloride Ophthalmic Solution,10%. Caution, therefore, should be exercised in administering the 10% solution to children of low body weight, the elderly, and patients with insulin-dependent diabetes, hypertension, hyperthyroidism, generalized arteriosclerosis, or cardiovascular disease. The post-treatment blood pressure of these patients, and any patients who develop symptoms, should be carefully monitored.
Ordinarily, any mydriatic, including phenylephrine hydrochloride, is contraindicated in patients with glaucoma, since it may occasionally raise intraocular pressure. However, when temporary dilation of the pupil may free adhesions or when vasoconstriction of intrinsic vessels may lower intraocular tension, these advantages may temporarily outweigh the danger from coincident dilation of the pupil. Rebound miosis has reported in older persons one day after receiving Phenylephrine Hydrochloride Ophthalmic Solutions, and re-instillation of the drug produced a reduction in mydriasis. This may be of clinical importance in dilating the pupils of older subjects prior to retinal detachment or cataract surgery.
Due to a strong action of the drug on the dilator muscle, older individuals may also develop transient pigment floaters in the aqueous humor 30 to 45 minutes following the administration of Phenylephrine Hydrochloride Ophthalmic Solutions. The appearance may be similar to anterior uveitis or a microscopic hyphema.
To prevent pain, a drop of suitable topical anesthetic may be applied before using the 10% ophthalmic solution.
Drug Interaction:
As with all other adrenergic drugs, when Phenylephrine Hydrochloride Solutions 10% or 2.5% are administered simultaneously with, or up to 21 days after, administration of monoamine oxidase (MAO) inhibitors, careful supervision and adjustment of dosages are required since exaggerated adrenergic effects may occur. The pressor response of adrenergic agents may also be potentiated by tricyclic antidepressants, propranolol, reserpine, guanethidine, methyldopa, and atropine-like drugs.
Carcinogenesis, Mutagenesis, Impairment of Fertility:
No long-term animal studies have been done to evaluate the potential of Phenylephrine Hydrochloride Ophthalmic Solution in these areas.
Pregnancy Category C:
Animal reproduction studies have not been conducted with Phenylephrine Hydrochloride Ophthalmic Solution. It is also not known whether Phenylephrine Hydrochloride Ophthalmic Solution can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Phenylephrine Hydrochloride Ophthalmic Solution should be given to a pregnant woman only if clearly needed.
Nursing Mothers:
It is not known whether this drug is excreted in milk; many are. Caution should be exercised when Phenylephrine Hydrochloride Ophthalmic Solution is administered to a nursing woman.
Pediatric Use:
Phenylephrine Hydrochloride Ophthalmic Solution, 10% is contraindicated in infants. (See CONTRAINDICATIONS.) For use in older children (See DOSAGE AND ADMINISTRATION.)
Exceeding recommended dosages or applying Phenylephrine Hydrochloride Ophthalmic Solution to the instrumented, traumatized, diseased or post surgical eye or adnexa, or to patients with suppressed lacrimation, as during anesthesia, may result in the absorption of sufficient quantities of phenylephrine to produce a systemic vasopressor response.
The hypertensive effects of phenylephrine may be treated with an alpha-adrenergic blocking agent such as phentolamine mesylate, 5 mg to 10 mg intravenously repeated as necessary.
The oral LD 50 of phenylephrine in the rat: 350 mg/kg, in the mouse: 120 mg/kg.
DOSAGE AND ADMINISTRATION:
Prolonged exposure to air or strong light may cause oxidation and discoloration. Do not use if solution is brown or contains a precipitate.
Vasoconstriction and Pupil Dilation:
Phenylephrine Hydrochloride Ophthalmic Solution, 10%, is especially useful when rapid and powerful dilation of the pupil and reduction of congestion in the capillary bed are desired. A drop of suitable topical anesthetic may be applied, followed in a few minutes by 1 drop of the Phenylephrine Hydrochloride Ophthalmic Solution, 10%, on the upper limbus. The anesthetic prevents stinging and consequent dilution of the solution by lacrimation. It may occasionally be necessary to repeat the instillation after one hour, again preceded by the use to the topical anesthetic.
Uveitis: Posterior Synechiae:
Phenylephrine Hydrochloride Ophthalmic Solution, 10%, may be used in patients with uveitis when synechiae are present or may develop. The formulation of synechiae may be prevented by the used of the 10% ophthalmic solution and atropine to produce wide dilation of the pupil. It should be emphasized, however, that the vasoconstrictor effect of the phenylephrine hydrochloride may be antagonistic to the increase of local blood flow in uveal infection.
To free recently formed posterior synechiae, 1 drop of the 10% ophthalmic solution may be applied to the upper surface of the cornea. On the following day, treatment may be continued if necessary. In the interim, hot compresses should be applied for five or ten minutes three times a day, with 1 drop of a 1% or 2% solution of atropine sulfate before and after each series of compresses.
Glaucoma:
In certain patients with glaucoma, temporary reduction of intraocular tension may be attained by producing vasoconstriction of the intraocular vessels; this may be accomplished by placing 1 drop of the 10% ophthalmic solution on the upper surface of the cornea. This treatment may be repeated as often as necessary.
Phenylephrine hydrochloride may be used with miotics in patients with wide angle glaucoma. It reduces the difficulties experienced by the patient because of the small field produced by miosis, and still it permits and often supports the effect of the miotic in lowering the intraocular pressure. Hence, there may be marked improvement in visual acuity after using phenylephrine hydrochloride in conjunction with miotic drugs.
Surgery:
When a short-acting mydriatic is needed for wide dilation of the pupil before intraocular surgery, the 10% or 2.5% ophthalmic solutions may be applied topically from 30-60 minutes before the operation.
Refraction:
Prior to determination of refractive errors, Phenylephrine Hydrochloride Ophthalmic Solution, 2.5% may be used effectively with homatropine hydrobromide, atropine sulfate, or a combination of homatropine and cocaine hydrochloride.
For ADULTS, a drop of the preferred cycloplegic is placed in each eye, followed in five minutes by 1 drop of Phenylephrine Hydrochloride Ophthalmic Solution, 2.5%, and in 10 minutes by another drop of cycloplegic. In 50-60 minutes, the eyes are ready for refraction.
For CHILDREN, a drop of atropine sulfate 1% is placed in each eye, followed in 10-15 minutes by 1 drop of Phenylephrine Hydrochloride Ophthalmic Solution, 2.5%, and in five to ten minutes by a second drop of atropine sulfate 1%. In one to two hours, the eyes are ready for refraction.
For a “one application method”, Phenylephrine Hydrochloride Ophthalmic Solution, 2.5%, may be combined with a cycloplegic to elicit synergistic action. The additive effect varies depending on the patient. Therefore, when using a “one application method”, it may be desirable to increase the concentration of the cycloplegic.
Ophthalmoscopic Examination:
One drop of Phenylephrine Hydrochloride Ophthalmic Solution, 2.5%, is placed in each eye. Sufficient mydriasis to permit examination is produced in 15 to 30 minutes. Dilations lasts from one to three hours.
Diagnostic Procedures:
PROVOCATIVE TEST FOR ANGLE BLOCK IN PATIENTS WITH GLAUCOMA: The 2.5% ophthalmic solution may be used as a provocative test when latent increased intraocular pressure is suspected. Tension is measured before application of the phenylephrine hydrochloride and again after dilation. A 3 to 5 mm of mercury rise in pressure suggest the presence of angle block in patients with glaucoma; however, failure to obtain such a rise does not preclude the presence of glaucoma form other causes.
SHADOW TEST (RETINOSCOPY): When dilation of the pupil without cycloplegic action is desired for the shadow test, the 2.5% ophthalmic solution may be used alone.
BLANCHING TEST: One or two drops of the 2.5% ophthalmic solution should be applied to the injected eye. After five minutes, examine for perilimbal blanching. If blanching occurs, the congestion is superficial and probably does not indicate iritis.
HOW SUPPLIED:
Phenylephrine Hydrochloride Ophthalmic Solution, 2.5%, is available in a sterile 15 mL plastic dropper bottle. Phenylephrine Hydrochloride Ophthalmic Solution, 10%, is available in sterile 5 mL plastic dropper bottle.
Keep container tightly closed.
Store below 40°C ( 104°F), preferably between 15°-30°C (59°-86°F).
| PHENYLEPHRINE HYDROCHLORIDE
OPHTHALMIC
phenylephrine hydrochloride solution/ drops |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - HUB Pharmaceuticals, LLC (611747945) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Conta Care Ophthalmics and Diagnostics | 915821765 | manufacture(17238-193) | |

