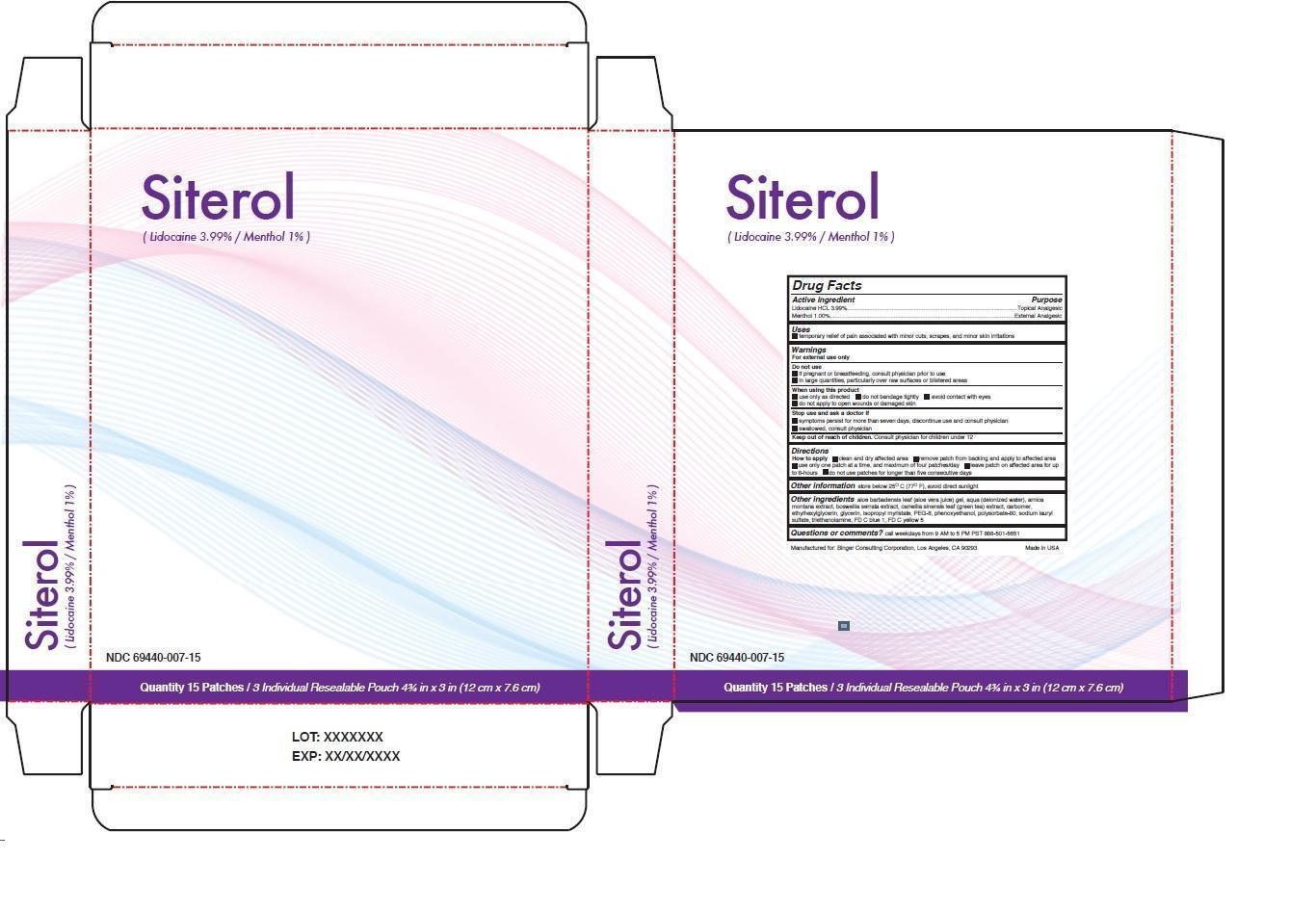
Label: SITEROL- lidocaine hcl, menthol patch
- NDC Code(s): 69440-007-15
- Packager: Binger Consulting Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 11, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- KEEP OUT OF REACH OF CHILDREN
- Uses
-
Warnings
For external use only
Do not use
• if pregnant or breastfeeding, consult physician prior to use
• in large quantities, particularly over raw surfaces or blistered areas
When using this product
• use only as directed • do not bandage tightly • avoid contact with eyes
• do not apply to open wounds or damaged skin
Stop use and ask a doctor if
• symptoms persist for more than seven days, discontinue use and consult physician
• swallowed, consult physician
- Directions
-
INACTIVE INGREDIENT
Other ingredients aloe barbadensis leaf (aloe vera juice) gel, aqua (deionized water), arnica montana extract, boswellia serrata extract, camellia sinensis leaf (green tea) extract, carbomer, ethylhexylglycerin, glycerin, isopropyl myristate, PEG-8, phenoxyethanol, polysorbate-80, sodium lauryl sulfate, triethanolamine, FD C blue 1, FD C yellow 5
- QUESTIONS
- STORAGE AND HANDLING
- Packaging
-
INGREDIENTS AND APPEARANCE
SITEROL
lidocaine hcl, menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69440-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 3.99 g in 100 g MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 1 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) ARNICA MONTANA WHOLE (UNII: O80TY208ZW) POLYSORBATE 80 (UNII: 6OZP39ZG8H) ALOE VERA LEAF (UNII: ZY81Z83H0X) BOSWELLIA SACRA WHOLE (UNII: 8O600AZL0W) GREEN TEA LEAF (UNII: W2ZU1RY8B0) CARBOMER COPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 809Y72KV36) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) PEG-8 GLYCERYL ISOSTEARATE (UNII: 74QQ5X3KL1) PHENOXYETHANOL (UNII: HIE492ZZ3T) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TRIETHANOLAMINE BENZOATE (UNII: M3EN4GC19W) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69440-007-15 15 in 1 BOX 01/01/2015 1 100 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/01/2015 Labeler - Binger Consulting Corporation (079635976) Establishment Name Address ID/FEI Business Operations Active Intelligence, LLC 080416593 manufacture(69440-007)