Label: RHINO LONG POWER- menthol, ginkgo biloba spray
- NDC Code(s): 71326-201-11, 71326-201-51
- Packager: HOT PRODUCTIONS AND VERTRIEBS GMBH
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
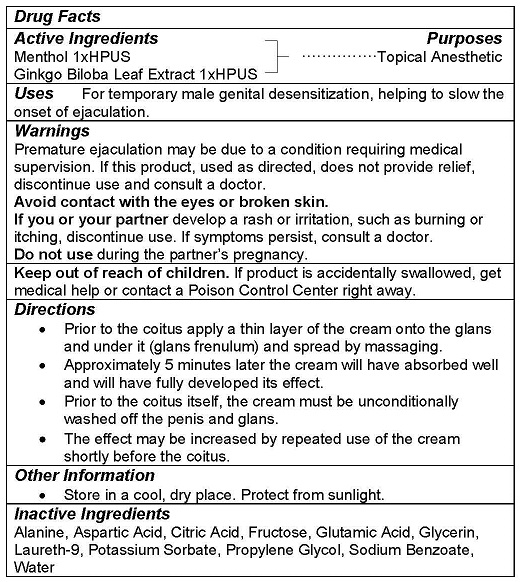
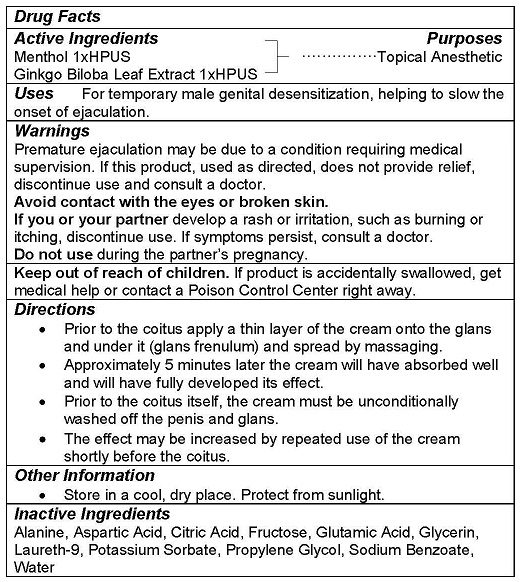
- ACTIVE INGREDIENTS
- PURPOSE
- USE
-
WARNINGS
Premature ejaculation may be due to a condition requiring medical supervision. If this product, used as directed, does not provide relief, discontinue use and consult a doctor.
Avoid contact with the eyes or broken skin.
If you or your partner develop a rash or irritation, such as burning or itching, discontinue use. If symptoms persist, consult a doctor.
Do not use during the partner’s pregnancy. - KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
- Prior to the coitus apply a thin layer of the cream onto the glans and under it (glans frenulum) and spread by massaging.
- Approximately 5 minutes later the cream will have absorbed well and will have fully developed its effect.
- Prior to the coitus itself, the cream must be unconditionally washed off the penis and glans.
- The effect may be increased by repeated use of the cream shortly before the coitus.
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RHINO LONG POWER
menthol, ginkgo biloba sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71326-201 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 1 g in 100 mL GINKGO (UNII: 19FUJ2C58T) (GINKGO - UNII:19FUJ2C58T) GINKGO 1 g in 100 mL Inactive Ingredients Ingredient Name Strength ALANINE (UNII: OF5P57N2ZX) ASPARTIC ACID (UNII: 30KYC7MIAI) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) FRUCTOSE (UNII: 6YSS42VSEV) GLUTAMIC ACID (UNII: 3KX376GY7L) GLYCERIN (UNII: PDC6A3C0OX) POLIDOCANOL (UNII: 0AWH8BFG9A) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM BENZOATE (UNII: OJ245FE5EU) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71326-201-51 1 in 1 BOX 04/06/2017 1 NDC:71326-201-11 10 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/06/2017 Labeler - HOT PRODUCTIONS AND VERTRIEBS GMBH (300011984)