Label: ANTI-AGING COMPLEX EYE TREATMENT MERLE NORMAN- octinoxate, zinc oxide lotion
- NDC Code(s): 57627-150-01, 57627-150-02
- Packager: Merle Norman Cosmetics
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 27, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
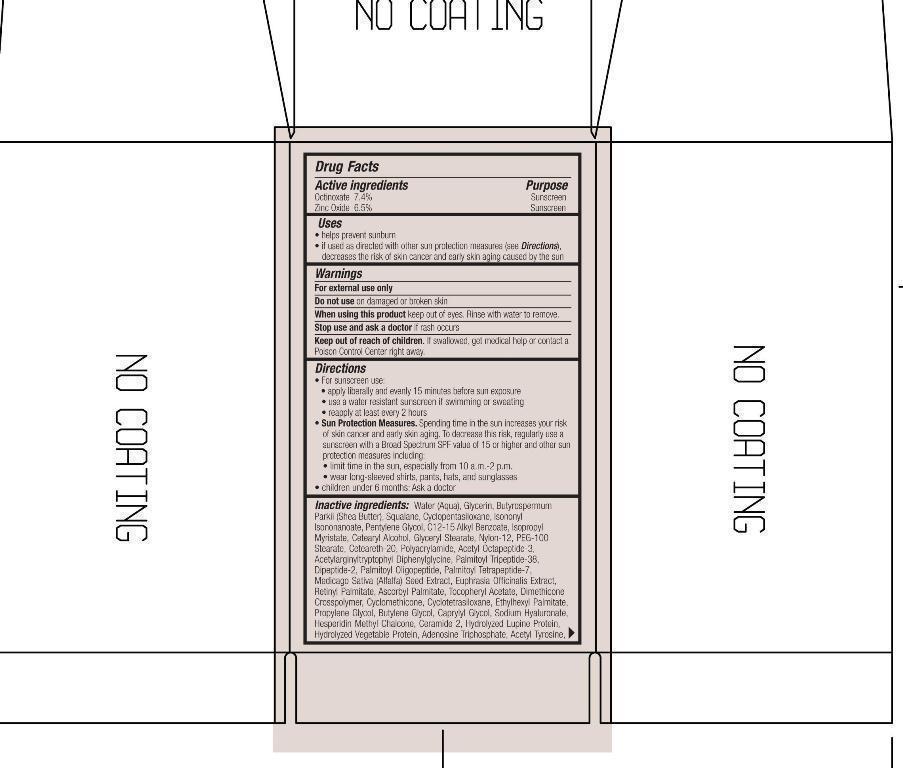
ACTIVE INGREDIENT
Active ingredients Purpose
Octinoxate 7.4% Sunscreen
Zinc Oxide 6.5% Sunscreen
Uses
helps prevent sunburn
If used as directed with other sun protection measures (See Directions) decreases the risk of skin cancer and early skin aging caused by the sun.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Stop use and ask a doctor if rash occurs
Warnings
For External use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Directions
For sunscreen
- apply liberally and evenly 15 minutes before sun exposure
- use a water resistant sunscreen if swimming
- reapply at least every 2 hours
Sun Protection Measures. Spending time in the sun increase your risk of skin cancer and early skin aging. To decrease this risk of skin cancer, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including"
- limit time in the sun, especially from 10 a.m.-2p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
children under six months: Ask a doctor
Inactive ingredients
Water (Aqua), Glycerin, Butyrospermum Parkii (Shea Butter), Squalane, Cyclopentasiloxane, Isononyl Myristate, Pentylene Glycol,
C12-15 Alkyl Benzoate, Isopropyl Myristate, Cetearyl Alcohol, Glyceryl Stearate, Nylon-12, PEG-100 Stearate, Ceteareth-20, Polyacrylamide,
Acetyl Octapeptide-3, Acetylarginyltryptophyl Diphenylglycine, Palmitoyl Tetrapeptide-7, Medicago Sativa (Alfafa) Seed Extract,
Euphrasia Officinalis Extract, Retinyl Palmitate, Ascorbyl Palmitate, Tocopheryl Acetate, Dimethicone Crosspolymer, Cyclomethicone,
Cyclotetrasiloxane, Ethylhexyl palmitate, Propylene Glycol, Butylene Glycol, Caprylyl Glycol, Sodium Hyaluronate, Hesperidin Methyl Chalcone, Ceramide-2, Hydrolyzed Lupine Protein, Hydrolyzed Vegetable Protein, Adenosine Triphosphate, Acetyl Tyrosine, Phospholipids, C13-14 Isoparaffin, Proline, Tribehenin, Triethoxycaprylylsilane, PEG-30 Dipolyhydroxypropyl, Laureth-7, Steareth-20 PEG-10 Rapeseed Sterol, Xanthan Gum, Hydroxypropyl Cyclodextrin, PVP, BHT Silica, Silica Dimethyl Silylate, Trisodium EDTA, Benzyl Alcohol, Tropolone Chlorphenesin, Phenoxyethanol, Titanium Dioxide, Mica. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ANTI-AGING COMPLEX EYE TREATMENT MERLE NORMAN
octinoxate, zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57627-150 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 1.18 g in 16 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 1.04 g in 16 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) SHEA BUTTER (UNII: K49155WL9Y) SQUALANE (UNII: GW89575KF9) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) ISONONYL ISONONANOATE (UNII: S4V5BS6GCX) PENTYLENE GLYCOL (UNII: 50C1307PZG) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) NYLON-12 (UNII: 446U8J075B) PEG-100 STEARATE (UNII: YD01N1999R) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) POLYACRYLAMIDE (1500 MW) (UNII: 5D6TC4BRWV) ACETYL OCTAPEPTIDE-3 (UNII: 8K14HJF88S) VALYLTRYPTOPHAN (UNII: 3G64B4AFQN) PALMITOYL OLIGOPEPTIDE (UNII: HO4ZT5S86C) PALMITOYL TETRAPEPTIDE-7 (UNII: Q41S464P1R) ALFALFA SEED (UNII: 67PHZ58858) EUPHRASIA STRICTA (UNII: C9642I91WL) VITAMIN A PALMITATE (UNII: 1D1K0N0VVC) ASCORBYL PALMITATE (UNII: QN83US2B0N) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CYCLOMETHICONE (UNII: NMQ347994Z) CYCLOMETHICONE 4 (UNII: CZ227117JE) ETHYLHEXYL PALMITATE (UNII: 2865993309) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CAPRYLYL GLYCOL (UNII: 00YIU5438U) HYALURONATE SODIUM (UNII: YSE9PPT4TH) HESPERIDIN METHYLCHALCONE (UNII: 4T2GVA922X) CERAMIDE 2 (UNII: C04977SRJ5) ADENOSINE TRIPHOSPHATE (UNII: 8L70Q75FXE) N-ACETYLTYROSINE (UNII: DA8G610ZO5) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) C13-14 ISOPARAFFIN (UNII: E4F12ROE70) PROLINE (UNII: 9DLQ4CIU6V) TRIBEHENIN (UNII: 8OC9U7TQZ0) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) PEG-30 DIPOLYHYDROXYSTEARATE (UNII: 9713Q0S7FO) LAURETH-7 (UNII: Z95S6G8201) STEARETH-20 (UNII: L0Q8IK9E08) PEG-10 RAPESEED STEROL (UNII: 258O76T85M) XANTHAN GUM (UNII: TTV12P4NEE) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SILICA DIMETHYL SILYLATE (UNII: EU2PSP0G0W) EDETATE TRISODIUM (UNII: 420IP921MB) BENZYL ALCOHOL (UNII: LKG8494WBH) TROPOLONE (UNII: 7L6DL16P1T) CHLORPHENESIN (UNII: I670DAL4SZ) PHENOXYETHANOL (UNII: HIE492ZZ3T) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) MICA (UNII: V8A1AW0880) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57627-150-02 1 in 1 CARTON 11/09/2012 1 NDC:57627-150-01 16 g in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 11/09/2012 Labeler - Merle Norman Cosmetics (008479388) Registrant - Merle Norman Cosmetics (008479388) Establishment Name Address ID/FEI Business Operations Merle Norman Cosmetics 008479388 manufacture(57627-150)