Label: DANTROLENE SODIUM capsule
- NDC Code(s): 49884-362-01, 49884-363-01, 49884-364-01
- Packager: Par Pharmaceutical, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application Authorized Generic
Drug Label Information
Updated December 5, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
BOXED WARNING
(What is this?)
Dantrolene sodium has a potential for hepatotoxicity, and should not be used in conditions other than those recommended. Symptomatic hepatitis (fatal and non-fatal) has been reported at various dose levels of the drug. The incidence reported in patients taking up to 400 mg/day is much lower than in those taking doses of 800 mg or more per day. Even sporadic short courses of these higher dose levels within a treatment regimen markedly increased the risk of serious hepatic injury. Liver dysfunction as evidenced by blood chemical abnormalities alone (liver enzyme elevations) has been observed in patients exposed to dantrolene sodium for varying periods of time. Overt hepatitis has occurred at varying intervals after initiation of therapy, but has been most frequently observed between the third and twelfth month of therapy. The risk of hepatic injury appears to be greater in females, in patients over 35 years of age, and in patients taking other medication(s) in addition to dantrolene sodium. Spontaneous reports suggest a higher proportion of hepatic events with fatal outcome in elderly patients receiving dantrolene sodium. However, the majority of these cases were complicated with confounding factors such as intercurrent illnesses and/or concomitant potentially hepatotoxic medications (see Geriatric Use subsection). Dantrolene sodium should be used only in conjunction with appropriate monitoring of hepatic function including frequent determination of SGOT or SGPT. If no observable benefit is derived from the administration of dantrolene sodium after a total of 45 days, therapy should be discontinued. The lowest possible effective dose for the individual patient should be prescribed.
-
DESCRIPTION
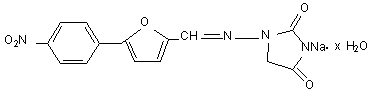
The chemical formula of dantrolene sodium is hydrated 1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]amino]-2, 4-imidazolidinedione sodium salt. It is an orange powder, slightly soluble in water, but due to its slightly acidic nature the solubility increases somewhat in alkaline solution. The anhydrous salt has a molecular weight of 336. The hydrated salt contains approximately 15% water (3-1/2 moles) and has a molecular weight of 399. The structural formula for the hydrated salt is:
Dantrolene sodium is supplied in capsules of 25 mg, 50 mg and 100 mg.
Inactive Ingredients: Each capsule contains Edible black ink, FD&C Yellow No. 6, Gelatin, Lactose, Magnesium stearate, Starch, Synthetic iron oxide red, Synthetic iron oxide yellow, Talc, and Titanium dioxide.
-
CLINICAL PHARMACOLOGY
In isolated nerve-muscle preparation, dantrolene sodium has been shown to produce relaxation by affecting the contractile response of the skeletal muscle at a site beyond the myoneural junction, directly on the muscle itself. In skeletal muscle, dantrolene sodium dissociates the excitation-contraction coupling, probably by interfering with the release of Ca++ from the sarcoplasmic reticulum. This effect appears to be more pronounced in fast muscle fibers as compared to slow ones, but generally affects both. A central nervous system effect occurs, with drowsiness, dizziness, and generalized weakness occasionally present. Although dantrolene sodium does not appear to directly affect the CNS, the extent of its indirect effect is unknown. The absorption of dantrolene sodium after oral administration in humans is incomplete and slow but consistent, and dose-related blood levels are obtained. The duration and intensity of skeletal muscle relaxation is related to the dosage and blood levels. The mean biologic half-life of dantrolene sodium in adults is 8.7 hours after a 100-mg dose. Specific metabolic pathways in the degradation and elimination of dantrolene sodium in human subjects have been established. Metabolic patterns are similar in adults and pediatric patients. In addition to the parent compound, dantrolene, which is found in measurable amounts in blood and urine, the major metabolites noted in body fluids are the 5-hydroxy analog and the acetamido analog. Since dantrolene sodium is probably metabolized by hepatic microsomal enzymes, enhancement of its metabolism by other drugs is possible. However, neither phenobarbital nor diazepam appears to affect dantrolene sodium metabolism.
Clinical experience in the management of fulminant human malignant hyperthermia, as well as experiments conducted in malignant hyperthermia susceptible swine, have revealed that the administration of intravenous dantrolene, combined with indicated supportive measures, is effective in reversing the hypermetabolic process of malignant hyperthermia. Known differences between human and swine malignant hyperthermia are minor. The prophylactic administration of oral or intravenous dantrolene to malignant hyperthermia susceptible swine will attenuate or prevent the development of signs of malignant hyperthermia in a manner dependent upon the dosage of dantrolene administered and the intensity of the malignant hyperthermia triggering stimulus. Limited clinical experience with the administration of oral dantrolene to patients judged malignant hyperthermia susceptible, when combined with clinical experience in the use of intravenous dantrolene for the treatment of malignant hyperthermia and data derived from the above cited animal model experiments, suggests that oral dantrolene will also attenuate or prevent the development of signs of human malignant hyperthermia, provided that currently accepted practices in the management of such patients are adhered to (see INDICATIONS AND USAGE); intravenous dantrolene should also be available for use should the signs of malignant hyperthermia appear.
-
INDICATIONS AND USAGE
In Chronic Spasticity
Dantrolene sodium is indicated in controlling the manifestations of clinical spasticity resulting from upper motor neuron disorders (e.g., spinal cord injury, stroke, cerebral palsy, or multiple sclerosis). It is of particular benefit to the patient whose functional rehabilitation has been retarded by the sequelae of spasticity. Such patients must have presumably reversible spasticity where relief of spasticity will aid in restoring residual function. Dantrolene sodium is not indicated in the treatment of skeletal muscle spasm resulting from rheumatic disorders.
If improvement occurs, it will ordinarily occur within the dosage titration (see DOSAGE AND ADMINISTRATION), and will be manifested by a decrease in the severity of spasticity and the ability to resume a daily function not quite attainable without dantrolene sodium.
Occasionally, subtle but meaningful improvement in spasticity may occur with dantrolene sodium therapy. In such instances, information regarding improvement should be solicited from the patient and those who are in constant daily contact and attendance with him. Brief withdrawal of dantrolene sodium for a period of 2 to 4 days will frequently demonstrate exacerbation of the manifestations of spasticity and may serve to confirm a clinical impression.
A decision to continue the administration of dantrolene sodium on a long-term basis is justified if introduction of the drug into the patient's regimen:
- produces a significant reduction in painful and/or disabling spasticity such as clonus, or
- permits a significant reduction in the intensity and/or degree of nursing care required, or
- rids the patient of any annoying manifestation of spasticity considered important by the patient himself.
In Malignant Hyperthermia
Oral dantrolene sodium is also indicated preoperatively to prevent or attenuate the development of signs of malignant hyperthermia in known, or strongly suspect, malignant hyperthermia susceptible patients who require anesthesia and/or surgery. Currently accepted clinical practices in the management of such patients must still be adhered to (careful monitoring for early signs of malignant hyperthermia, minimizing exposure to triggering mechanisms and prompt use of intravenous Dantrolene Sodium and indicated supportive measures should signs of malignant hyperthermia appear); see also the package insert for Dantrium® (dantrolene sodium) Intravenous.
Oral dantrolene sodium should be administered following a malignant hyperthermic crisis to prevent recurrence of the signs of malignant hyperthermia.
-
CONTRAINDICATIONS
Active hepatic disease, such as hepatitis and cirrhosis, is a contraindication for use of dantrolene sodium. Dantrolene sodium is contraindicated where spasticity is utilized to sustain upright posture and balance in locomotion or whenever spasticity is utilized to obtain or maintain increased function.
-
WARNINGS
It is important to recognize that fatal and non-fatal liver disorders of an idiosyncratic or hypersensitivity type may occur with dantrolene sodium therapy.
At the start of dantrolene sodium therapy, it is desirable to do liver function studies (SGOT, SGPT, alkaline phosphatase, total bilirubin) for a baseline or to establish whether there is pre-existing liver disease. If baseline liver abnormalities exist and are confirmed, there is a clear possibility that the potential for dantrolene sodium hepatotoxicity could be enhanced, although such a possibility has not yet been established.
Liver function studies (e.g., SGOT or SGPT) should be performed at appropriate intervals during dantrolene sodium therapy. If such studies reveal abnormal values, therapy should generally be discontinued. Only where benefits of the drug have been of major importance to the patient, should reinitiation or continuation of therapy be considered. Some patients have revealed a return to normal laboratory values in the face of continued therapy while others have not.
If symptoms compatible with hepatitis, accompanied by abnormalities in liver function tests or jaundice appear, dantrolene sodium should be discontinued. If caused by dantrolene sodium and detected early, the abnormalities in liver function characteristically have reverted to normal when the drug was discontinued.
Dantrolene sodium therapy has been reinstituted in a few patients who have developed clinical and/or laboratory evidence of hepatocellular injury. If such reinstitution of therapy is done, it should be attempted only in patients who clearly need dantrolene sodium and only after previous symptoms and laboratory abnormalities have cleared. The patient should be hospitalized and the drug should be restarted in very small and gradually increasing doses. Laboratory monitoring should be frequent and the drug should be withdrawn immediately if there is any indication of recurrent liver involvement. Some patients have reacted with unmistakable signs of liver abnormality upon administration of a challenge dose, while others have not.
Dantrolene sodium should be used with particular caution in females and in patients over 35 years of age in view of apparent greater likelihood of drug-induced, potentially fatal, hepatocellular disease in these groups. Spontaneous reports suggest a higher proportion of hepatic events with fatal outcome in elderly patients receiving dantrolene sodium. However, the majority of these cases were complicated with confounding factors such as intercurrent illnesses and/or concomitant potentially hepatotoxic medications (see Geriatric Use subsection).
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term safety of dantrolene sodium in humans has not been established. Chronic studies in rats, dogs, and monkeys at dosages greater than 30 mg/kg/day showed growth or weight depression and signs of hepatopathy and possible occlusion nephropathy, all of which were reversible upon cessation of treatment. Sprague-Dawley female rats fed dantrolene sodium for 18 months at dosage levels of 15, 30, and 60 mg/kg/day showed an increased incidence of benign and malignant mammary tumors compared with concurrent controls. At the highest dose level, there was an increase in the incidence of benign lymphatic neoplasms. In a 30-month study at the same dose levels also in Sprague-Dawley rats, Dantrolene Sodium produced a decrease in the time of onset of mammary neoplasms. Female rats at the highest dose level showed an increased incidence of hepatic lymphangiomas and hepatic angiosarcomas.
The only drug-related effect seen in a 30-month study in Fischer-344 rats was a dose-related reduction in the time of onset of mammary and testicular tumors. A 24-month study in HaM/ICR mice revealed no evidence of carcinogenic activity. Carcinogenicity in humans cannot be fully excluded, so that this possible risk of chronic administration must be weighed against the benefits of the drug (i.e., after a brief trial) for the individual patient.
Dantrolene Sodium has produced positive results in the Ames S. Typhimurium bacterial mutagenesis assay in the presence and absence of a liver activating system.
Pregnancy
Pregnancy Category C
Adequate animal reproduction studies have not been conducted with dantrolene sodium. It is also not known whether dantrolene sodium can cause fatal harm when administered to a pregnant woman or can affect reproduction capacity. Dantronlene sodium should be given to a pregnant woman only if clearly needed.
Labor and Delivery
In one non-randomized open-label study, 21 term pregnant patients received prophylactic oral dantrolene sodium 100 mg per day for 2 to 10 days prior to delivery. Dantrolene readily crossed the placenta with maternal and fetal whole blood levels approximately equal at delivery; neonatal levels then fell approximately 50% per day for 2 days before declining sharply. No neonatal respiratory and neuromuscular side effects were detected at low dose. More data, at higher doses, are needed before more definitive conclusions can be made.
Usage in Pediatric Patients
The long-term safety of dantrolene sodium in pediatric patients under the age of 5 years has not been established. Because of the possibility that adverse effects of the drug could become apparent only after many years, a benefit-risk consideration of the long-term use of dantrolene sodium is particularly important in pediatric patients.
Geriatric Use
Clinical studies of dantrolene sodium did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience in the literature has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. As with all patients receiving dantrolene sodium, it is recommended that elderly patients receive the lowest dose compatible with the optimal response. Spontaneous reports suggest a higher proportion of hepatic events with fatal outcome in elderly patients receiving dantrolene sodium. However, the majority of these cases were complicated with confounding factors such as intercurrent illnesses and/or concomitant potentially hepatotoxic medications (for hepatotoxicity details and its management see Black Box and Warnings Sections).
Drug Interactions
Drowsiness may occur with dantrolene sodium therapy, and the concomitant administration of CNS depressants such as sedatives and tranquilizing agents may result in further drowsiness.
While a definite drug interaction with estrogen therapy has not yet been established, caution should be observed if the two drugs are to be given concomitantly. Hepatotoxicity has occurred more often in women over 35 years of age receiving concomitant estrogen therapy.
Cardiovascular collapse in patients treated simultaneously with verapamil and Dantrolene Sodium is rare. The combination of therapeutic doses of intravenous Dantrolene Sodium and verapamil in halothane/α-chloralose anesthetized swine has resulted in ventricular fibrillation and cardiovascular collapse in association with marked hyperkalemia. Until the relevance of these findings to humans is established, the combination of Dantrolene Sodium and calcium channel blockers is not recommended during the management of malignant hyperthermia.
Administration of dantrolene sodium may potentiate vecuronium-induced neuromuscular block.
-
PRECAUTIONS
Dantrolene sodium should be used with caution in patients with impaired pulmonary function, particularly those with obstructive pulmonary disease, and in patients with severely impaired cardiac function due to myocardial disease. Datrolene sodium is associated with pleural effusion with associated eosinophilia. It should be used with caution in patients with a history of previous liver disease or dysfunction (see WARNINGS).
Information for Patients
Patients should be cautioned against driving a motor vehicle or participating in hazardous occupations while taking dantrolene sodium. Caution should be exercised in the concomitant administration of tranquilizing agents.
Dantrolene sodium might possibly evoke a photosensitivity reaction; patients should be cautioned about exposure to sunlight while taking it.
-
ADVERSE REACTIONS
The most frequently occurring side effects of dantrolene sodium have been drowsiness, dizziness, weakness, general malaise, fatigue, and diarrhea. These are generally transient, occurring early in treatment, and can often be obviated by beginning with a low dose and increasing dosage gradually until an optimal regimen is established. Diarrhea may be severe and may necessitate temporary withdrawal of dantrolene sodium therapy. If diarrhea recurs upon readministration of dantrolene sodium, therapy should probably be withdrawn permanently.
Other less frequent side effects, listed according to system, are:
Gastrointestinal: Constipation, rarely progressing to signs of intestinal obstruction, GI bleeding, anorexia, swallowing difficulty, gastric irritation, abdominal cramps, nausea and/or vomiting.
Hepatobiliary: Hepatitis (see WARNINGS).
Neurologic: Speech disturbance, seizure, headache, light-headedness, visual disturbance, diplopia, alteration of taste, insomnia, drooling.
Cardiovascular: Tachycardia, erratic blood pressure, phlebitis, heart failure.
Hematologic: Aplastic anemia, anemia, leukopenia, lymphocytic lymphoma, thrombocytopenia.
Psychiatric: Mental depression, mental confusion, increased nervousness.
Urogenital: Increased urinary frequency, crystalluria, hematuria, difficult erection, urinary incontinence and/or nocturia, difficult urination and/or urinary retention.
Integumentary: Abnormal hair growth, acne-like rash, pruritus, urticaria, eczematoid eruption, sweating.
Musculoskeletal: Myalgia, backache.
Respiratory: Feeling of suffocation, respiratory depression.
Special Senses: Excessive tearing.
Hypersensitivity: Pleural effusion with pericarditis, pleural effusion with associated eosinophilia, anaphylaxis.
Other: Chills and fever.
The published literature has included some reports of dantrolene sodium use in patients with Neuroleptic Malignant Syndrome (NMS). Dantrolene sodium capsules are not indicated for the treatment of NMS and patients may expire despite treatment with dantrolene sodium capsules.
For medical advice about adverse reactions contact your medical professional. To report SUSPECTED ADVERSE REACTIONS, contact Par Pharmaceutical at 1-800-828-9393 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- DRUG ABUSE AND DEPENDENCE
-
OVERDOSAGE
Symptoms which may occur in case of overdose include, but are not limited to, muscular weakness and alterations in the state of consciousness (e.g. lethargy, coma), vomiting, diarrhea, and crystalluria. For acute overdose, general supportive measures should be employed along with immediate gastric lavage.
Intravenous fluids should be administered in fairly large quantities to avert the possibility of crystalluria. An adequate airway should be maintained and artificial resuscitation equipment should be at hand. Electrocardiographic monitoring should be instituted, and the patient carefully observed. To date, no experience has been reported with dialysis and its value in dantrolene sodium overdose is not known.
-
DOSAGE AND ADMINISTRATION
For Use in Chronic Spasticity
Prior to the administration of dantrolene sodium, consideration should be given to the potential response to treatment. A decrease in spasticity sufficient to allow a daily function not otherwise attainable should be the therapeutic goal of treatment with dantrolene sodium. Refer to INDICATIONS AND USAGE section for description of response to be anticipated.
It is important to establish a therapeutic goal (regain and maintain a specific function such as therapeutic exercise program, utilization of braces, transfer maneuvers, etc.) before beginning dantrolene sodium therapy. Dosage should be increased until the maximum performance compatible with the dysfunction due to underlying disease is achieved. No further increase in dosage is then indicated.
Usual Dosage
It is important that the dosage be titrated and individualized for maximum effect. The lowest dose compatible with optimal response is recommended.
In view of the potential for liver damage in long-term dantrolene sodium use, therapy should be stopped if benefits are not evident within 45 days.
Adults
The following gradual titration schedule is suggested. Some patients will not respond until higher daily dosage is achieved. Each dosage level should be maintained for seven days to determine the patient's response. If no further benefit is observed at the next higher dose, dosage should be decreased to the previous lower dose.
- 25 mg once daily for seven days, then
- 25 mg t.i.d. for seven days
- 50 mg t.i.d. for seven days
- 100 mg t.i.d.
Therapy with a dose four times daily may be necessary for some individuals. Doses higher than 100 mg four times daily should not be used. (See Box Warning.)
Pediatric Patients
The following gradual titration schedule is suggested. Some patients will not respond until higher daily dosage is achieved. Each dosage level should be maintained for seven days to determine the patient's response. If no further benefit is observed at the next higher dose, dosage should be decreased to the previous lower dose.
- 0.5 mg/kg once daily for seven days, then
- 0.5 mg/kg t.i.d. for seven days
- 1 mg/kg t.i.d. for seven days
- 2 mg/kg t.i.d.
Therapy with a dose four times daily may be necessary for some individuals. Doses higher than 100 mg four times daily should not be used. (See Box Warning.)
For Malignant Hyperthermia
Preoperatively
Administer 4 to 8 mg/kg/day of oral dantrolene sodium in 3 or 4 divided doses for one or two days prior to surgery, with the last dose being given approximately 3 to 4 hours before scheduled surgery with a minimum of water.
This dosage will usually be associated with skeletal muscle weakness and sedation (sleepiness or drowsiness); adjustment can usually be made within the recommended dosage range to avoid incapacitation or excessive gastrointestinal irritation (including nausea and/or vomiting).
-
HOW SUPPLIED
Dantrolene sodium is available in:
25-mg opaque, orange and tan capsules imprinted with DANTRIUM 25 mg on the cap and
0149 0030 with a single bar on the body.
NDC 49884-362-01 bottle of 10050-mg opaque, orange and tan capsules imprinted with DANTRIUM 50 mg on the cap and
0149 0031 with a double bar on the body.
NDC 49884-363-01 bottle of 100100-mg opaque, orange and tan capsules imprinted with DANTRIUM 100 mg on the cap and
0149 0033 with a triple bar on the body.
NDC 49884-364-01 bottle of 100 - SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 25 mg Capsule Bottle Label
- PRINCIPAL DISPLAY PANEL - 50 mg Capsule Bottle Label
- PRINCIPAL DISPLAY PANEL – 100 mg Capsules Bottle Label
-
INGREDIENTS AND APPEARANCE
DANTROLENE SODIUM
dantrolene sodium capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-362 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DANTROLENE SODIUM (UNII: 287M0347EV) (DANTROLENE - UNII:F64QU97QCR) DANTROLENE SODIUM 25 mg Inactive Ingredients Ingredient Name Strength FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color ORANGE, BROWN (tan) Score no score Shape CAPSULE Size 16mm Flavor Imprint Code Dantrium;25;mg;0149;0030 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-362-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/28/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA017443 03/28/2016 DANTROLENE SODIUM
dantrolene sodium capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-363 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DANTROLENE SODIUM (UNII: 287M0347EV) (DANTROLENE - UNII:F64QU97QCR) DANTROLENE SODIUM 50 mg Inactive Ingredients Ingredient Name Strength FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color ORANGE, BROWN (tan) Score no score Shape CAPSULE Size 18mm Flavor Imprint Code Dantrium;50;mg;0149;0031 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-363-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/28/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA017443 03/28/2016 DANTROLENE SODIUM
dantrolene sodium capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:49884-364 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DANTROLENE SODIUM (UNII: 287M0347EV) (DANTROLENE - UNII:F64QU97QCR) DANTROLENE SODIUM 100 mg Inactive Ingredients Ingredient Name Strength FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERRIC OXIDE RED (UNII: 1K09F3G675) Product Characteristics Color ORANGE, BROWN Score no score Shape CAPSULE Size 20mm Flavor Imprint Code Dantirum;100;mg;0149;0033 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49884-364-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 03/28/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA AUTHORIZED GENERIC NDA017443 03/28/2016 Labeler - Par Pharmaceutical, Inc. (092733690) Establishment Name Address ID/FEI Business Operations Par Formulations Private Limited 676159161 ANALYSIS(49884-362, 49884-363, 49884-364) , MANUFACTURE(49884-362, 49884-363, 49884-364) , PACK(49884-362, 49884-363, 49884-364)






