DEPO-PROVERA- medroxyprogesterone acetate injection, suspension
Pharmacia and Upjohn Company LLC
----------
Depo-Provera®
medroxyprogesterone
acetate injectable
suspension, USP
DESCRIPTION
DEPO-PROVERA Sterile Aqueous Suspension contains medroxyprogesterone acetate, which is a derivative of progesterone and is active by the parenteral and oral routes of administration. It is a white to off-white, odorless crystalline powder, stable in air, melting between 200° and 210° C. It is freely soluble in chloroform, soluble in acetone and in dioxane, sparingly soluble in alcohol and methanol, slightly soluble in ether and insoluble in water.
The chemical name for medroxyprogesterone acetate is Pregn-4-ene-3, 20-dione, 17-(acetyloxy)-6-methyl-, (6α)-. The structural formula is:
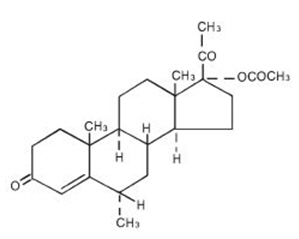
medroxyprogesterone acetate
DEPO-PROVERA for intramuscular injection is available as 400 mg/mL medroxyprogesterone acetate. Each mL of the 400 mg/mL suspension contains:
| Medroxyprogesterone acetate | 400 mg |
| Polyethylene glycol 3350 | 20.3 mg |
| Sodium sulfate anhydrous | 11 mg |
| with | |
| Myristyl-gamma-picolinium chloride | 1.69 mg |
| added as preservative | |
When necessary, pH was adjusted with sodium hydroxide and/or hydrochloric acid.
ACTIONS
Medroxyprogesterone acetate, administered parenterally in the recommended doses to women with adequate endogenous estrogen, transforms proliferative endometrium into secretory endometrium.
Medroxyprogesterone acetate inhibits (in the usual dose range) the secretion of pituitary gonadotropin which, in turn, prevents follicular maturation and ovulation.
Because of its prolonged action and the resulting difficulty in predicting the time of withdrawal bleeding following injection, medroxyprogesterone acetate is not recommended in secondary amenorrhea or dysfunctional uterine bleeding. In these conditions oral therapy is recommended.
INDICATIONS AND USES
Adjunctive therapy and palliative treatment of inoperable, recurrent, and metastatic endometrial or renal carcinoma.
CONTRAINDICATIONS
- Active thrombophlebitis, or current or past history of thromboembolic disorders, or cerebral vascular disease
- Known sensitivity to DEPO-PROVERA (medroxyprogesterone acetate or any of its other ingredients).
WARNINGS
1. Thromboembolic Disorders
The physician should be alert to the earliest manifestations of thrombotic disorder (thrombophlebitis, cerebrovascular disorder, pulmonary embolism, and retinal thrombosis). Should any of these occur or be suspected, the drug should be discontinued immediately.
PRECAUTIONS
1. Physical Examination
It is good medical practice for all women to have annual history and physical examinations, including women using DEPO-PROVERA Sterile Aqueous Suspension. The physical examination, however, may be deferred until after initiation of DEPO-PROVERA if requested by the woman and judged appropriate by the clinician. The physical examination should include special reference to blood pressure, breasts, abdomen and pelvic organs, including cervical cytology and relevant laboratory tests. In case of undiagnosed, persistent or recurrent abnormal vaginal bleeding, appropriate measures should be conducted to rule out malignancy.
2. Breast Cancer
Women who have or have had a history of breast cancer should be advised against the use of DEPO-PROVERA, as breast cancer may be hormonally sensitive. Women with a strong family history of breast cancer should be monitored with particular care.
3. Fluid Retention
Because progestational drugs may cause some degree of fluid retention, conditions which might be influenced by this condition, such as epilepsy, migraine, asthma, cardiac or renal dysfunction, require careful observation.
4.Vaginal Bleeding
In cases of breakthrough bleeding, as in all cases of irregular bleeding per vaginum, nonfunctional causes should be borne in mind and adequate diagnostic measures undertaken.
5. Depression
Patients who have a history of psychic depression should be carefully observed and the drug discontinued if the depression recurs to a serious degree.
6.Masking of Climacteric
The age of the patient constitutes no absolute limiting factor although treatment with progestin may mask the onset of the climacteric.
7. Use with Estrogen
Studies of the addition of a progestin product to an estrogen replacement regimen for seven or more days of a cycle of estrogen administration have reported a lowered incidence of endometrial hyperplasia. Morphological and biochemical studies of endometrial suggest that 10–13 days of a progestin are needed to provide maximal maturation of the endometrium and to eliminate any hyperplastic changes. Whether this will provide protection from endometrial carcinoma has not been clearly established.
There are possible risks which may be associated with the inclusion of progestin in estrogen replacement regimen, including adverse effects on carbohydrate and lipid metabolism. The dosage used may be important in minimizing these adverse effects.
A decrease in glucose tolerance has been observed in a small percentage of patients on estrogen-progestin combination treatment. The mechanism of this decrease is obscure. For this reason, diabetic patients should be carefully observed while receiving such therapy.
8. Hepatic Dysfunction
Monitor patients for hepatic dysfunction periodically and temporarily interrupt DEPO-PROVERA Sterile Aqueous Suspension use if the patient develops hepatic dysfunction. Do not resume use until markers of liver function return to normal.
9. Decrease in Bone Mineral Density
Studies in pre-menopausal women show that medroxyprogesterone acetate given as 150 mg intramuscularly every three months reduces serum estrogen levels and is associated with loss of bone mineral density (BMD). It is unknown if use of Depo-Provera during adolescence and early adulthood, a critical period of bone accretion, will reduce peak bone mass. An evaluation of BMD may be appropriate in some patients who use higher doses of medroxyprogesterone acetate for long-term treatment of endometrial or renal carcinoma.
10. Effects on the Hypothalmic-Pituitary-Adrenal Axis
Some patients receiving medroxyprogesterone acetate may exhibit suppressed adrenal function. Medroxyprogesterone acetate may have cortisol-like glucocorticoid activity and provide negative feedback to the hypothalamus or pituitary. This may result in decreased plasma cortisol levels, decreased cortisol secretion, and low plasma ACTH levels.
The use of DEPO-PROVERA Sterile Aqueous Suspension may, due to its cortisol-like glucocorticoid activity, also produce Cushingoid symptoms such as weight gain, edema/fluid retention, and facial swelling.
11. Prolonged Use
The effect of prolonged use of DEPO-PROVERA Sterile Aqueous Suspension at the recommended doses on pituitary, ovarian, adrenal, hepatic, and uterine function is not known.
12. Interference with Laboratory Tests
The use of DEPO-PROVERA Sterile Aqueous Suspension may change the results of some laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins. [See LABORATORY TEST INTERACTIONS].
13. Multi-dose Use
When multi-dose vials are used, special care to prevent contamination of the contents is essential. There is some evidence that benzalkonium chloride is not an adequate antiseptic for sterilizing DEPO-PROVERA Sterile Aqueous Suspension multi-dose vials. A povidone-iodine solution or similar product is recommended to cleanse the vial top prior to aspiration of contents. [See WARNINGS].
14. Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term intramuscular administration of Medroxyprogesterone acetate (MPA) has been shown to produce mammary tumors in beagle dogs. There is no evidence of a carcinogenic effect associated with the oral administration of MPA to rats and mice. Medroxyprogesterone acetate was not mutagenic in a battery of in vitro or in vivo genetic toxicity assays.
Medroxyprogesterone acetate at high doses is an anti-fertility drug and return to ovulation and fertility may be delayed after stopping treatment.
16. Nursing Mothers
Published studies report the presence of medroxyprogesterone acetate in human milk. Caution should be exercised when medroxyprogesterone acetate is administered to a nursing woman.
17. Pediatric Use
Safety and efficacy of DEPO- PROVERA for endometrial and renal carcinoma have not been established in pediatric patients.
Studies in pre-menopausal women show that Depo-Provera is associated with loss of BMD. It is unknown if use of Depo-Provera during adolescence and early adulthood, a critical period of bone accretion, will reduce peak bone mass. (See PRECAUTIONS: Decrease in Bone Mineral Density)
DRUG INTERACTIONS
Aminoglutethimide administered concomitantly with DEPO-PROVERA Sterile Aqueous Suspension may significantly depress the serum concentrations of medroxyprogesterone acetate. DEPO-PROVERA users should be warned of the possibility of decreased efficacy with the use of this or any related drugs.
In vitro
Medroxyprogesterone acetate is metabolized primarily by hydroxylation via the CYP3A4. Though no formal drug interaction trials have been conducted, concomitant administration of strong CYP3A inhibitors is expected to increase concentrations of medroxyprogesterone acetate, whereas the concomitant administration of strong CYP3A inducers is expected to decrease medroxyprogesterone acetate concentrations. Therefore, coadministration with strong CYP3A inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole) or strong CYP3A inducers (e.g., phenytoin, carbamazepine, rifampin, rifabutin, rifapentin, phenobarbital, St. John's Wort) should be avoided.
LABORATORY TEST INTERACTIONS
The pathologist should be advised of progestin therapy when relevant specimens are submitted. The following laboratory tests may be affected by progestins including DEPO-PROVERA Sterile Aqueous Suspension:
- a)
- Plasma and urinary steroid levels are decreased (e.g. progesterone, estradiol, pregnanediol, testosterone, cortisol).
- b)
- Gonadotropin levels are decreased.
- c)
- Sex-hormone binding globulin concentrations are decreased.
- d)
- Protein bound iodine and butanol extractable protein bound iodine may increase. T3 uptake values may decrease.
- e)
- Coagulation test values for prothrombin (Factor II), and Factors VII, VIII, IX, and X may increase.
- f)
- Sulfobromophthalein and other liver function test values may be increased.
- g)
- The effects of medroxyprogesterone acetate on lipid metabolism are inconsistent. Both increases and decreases in total cholesterol, triglycerides, low-density lipoprotein (LDL) cholesterol, and high-density lipoprotein (HDL) cholesterol have been observed in studies.
ADVERSE REACTIONS
See PRECAUTIONS for possible adverse effects on the fetus
Reproductive System and Breast Disorders
- –
- breakthrough bleeding
- –
- spotting
- –
- change in menstrual flow
- –
- amenorrhea
- –
- changes in cervical erosion and cervical secretions
- –
- breast tenderness and galactorrhea
- –
- erectile dysfunction
General Disorders and Administration Site Conditions
- –
- edema
- –
- pyrexia
- –
- fatigue
- –
- malaise
- –
- injection site reaction, injection site pain/tenderness, injection site persistent atrophy/indentation/dimpling, lipodystrophy acquired, injection site nodule/lump
In a few instances there have been undesirable sequelae at the site of injection, such as residual lump, change in color of skin, or sterile abscess.
Skin and Subcutaneous Tissue Disorders
- –
- skin sensitivity reactions consisting of urticaria, pruritus, edema and generalized rash
- –
- acne, alopecia and hirsutism
- –
- rash (allergic) with and without pruritis
A statistically significant association has been demonstrated between use of estrogen-pro-gestin combination drugs and pulmonary embolism and cerebral thrombosis and embolism. For this reason patients on progestin therapy should be carefully observed. There is also evidence suggestive of an association with neuro-ocular lesions, e.g. retinal thrombosis and optic neuritis.
The following adverse reactions have been observed in patients receiving estrogen-progestin combination drugs:
- –
- rise in blood pressure in susceptible individuals
- –
- premenstrual syndrome
- –
- changes in libido
- –
- changes in appetite
- –
- cystitis-like syndrome
- –
- headache
- –
- nervousness
- –
- fatigue
- –
- backache
- –
- hirsutism
- –
- loss of scalp hair
- –
- erythema multiforma
- –
- erythema nodosum
- –
- hemorrhagic eruption
- –
- itching
- –
- dizziness
The following laboratory results may be altered by the use of estrogen-progestin combination drugs:
- –
- increased sulfobromophthalein retention and other hepatic function tests
- –
- coagulation tests: increase in prothrombin factors VII, VIII, IX, and X
- –
- metyrapone test
- –
- pregnanediol determinations
- –
- thyroid function: increase in PBI, and butanol extractable protein bound iodine and decrease in T3 uptake values
DOSAGE AND ADMINISTRATION
The suspension is intended for intramuscular administration only, rotating the sites with every injection. As with any intramuscular injection, to avoid an inadvertent subcutaneous injection, body habitus should be assessed prior to each injection to determine if a longer needle is necessary particularly for gluteal intramuscular injection.
When multi-dose vials are used, special care to prevent contamination of the contents is essential [see WARNINGS and PRECAUTIONS].
Endometrial or Renal Carcinoma
Doses of 400 mg to 1000 mg of DEPO-PROVERA Sterile Aqueous Suspension per week are recommended initially. If improvement is noted within a few weeks or months and the disease appears stabilized, it may be possible to maintain improvement with as little as 400 mg per month. Medroxyprogesterone acetate is not recommended as primary therapy, but as adjunctive and palliative treatment in advanced inoperable cases including those with recurrent or metastatic disease.
Geriatric Use
Renal Carcinoma
Of the 349 subjects in a clinical study of Depo Provera in renal carcinoma, 30 percent were 65 and over, while 5 percent were 75 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Hepatic Impairment
DEPO-PROVERA Sterile Aqueous Suspension should not be used by women with significant liver disease and should be discontinued if jaundice or disturbances of liver function occur [see PRECAUTIONS].
HOW SUPPLIED
DEPO-PROVERA Sterile Aqueous Suspension is available as 400 mg/mL in 2.5 mL vials.
NDC 0009-0626-01 2.5ml vial
| DEPO-PROVERA
medroxyprogesterone acetate injection, suspension |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Pharmacia and Upjohn Company LLC (618054084) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Pharmacia and Upjohn Company LLC | 618054084 | ANALYSIS(0009-0626) , API MANUFACTURE(0009-0626) , LABEL(0009-0626) , MANUFACTURE(0009-0626) , PACK(0009-0626) | |