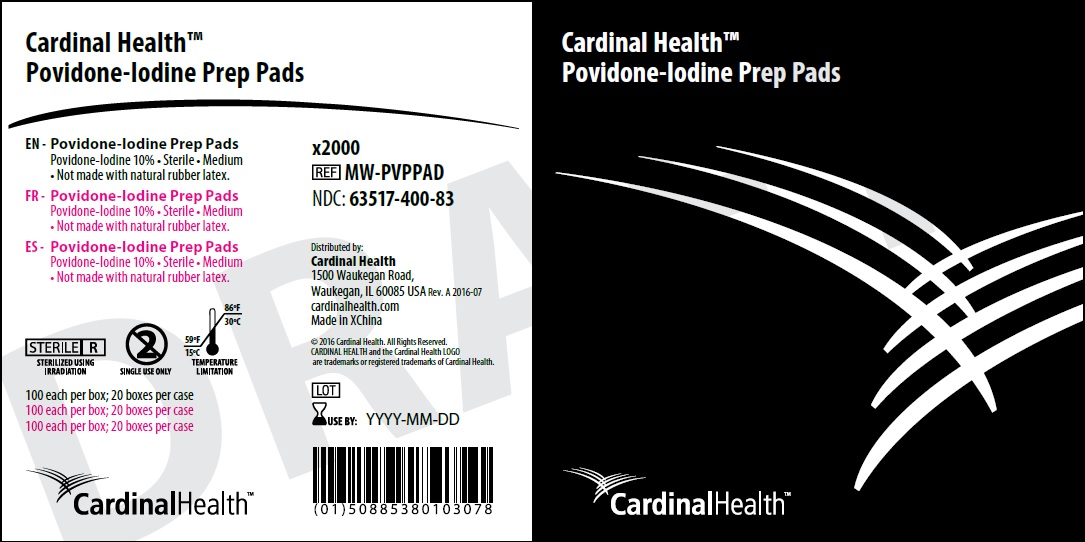
Label: POVIDONE-IODINE PREP PAD- povidone-iodine solution
- NDC Code(s): 63517-400-83
- Packager: Cardinal Health 200, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Use
- Warnings
- Directions
- Other information
- Inactive ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
POVIDONE-IODINE PREP PAD
povidone-iodine solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63517-400 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63517-400-83 10 in 1 CARTON 08/05/2016 1 100 in 1 BOX 1 0.56 g in 1 POUCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 08/05/2016 Labeler - Cardinal Health 200, Inc. (961027315) Establishment Name Address ID/FEI Business Operations Jianerkang Medical Co., Ltd 530968767 manufacture(63517-400)