METHYLENE BLUE
- methylene blue injection, solution
General Injectables & Vaccines, Inc.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
METHYLENE BLUE 10MG/ML
DESCRIPTION
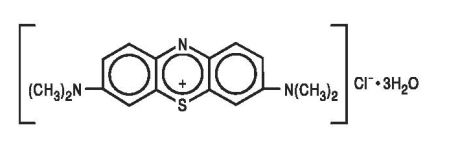
Methylene Blue Injection, USP is a sterile solution of Phenothiazin_5_ium, 3, 7-bis (dimethylamino)- chloride, trihydrate. Each mL contains Methylene Blue, USP, 10 mg in Water for Injection q.s. pH adjusted with Hydrochloric Acid and /or Sodium Hydroxide when necessary.
The structural formula is:
CLINICAL PHARMACOLOGY
Methylene Blue will produce two opposite actions on hemoglobin. Low concentrations will convert methemoglobin to hemoglobin. High concentrations convert the ferrous iron of reduced hemoglobin to ferric iron which results in the formation of methemoglobin.
Methylene blue is metabolized in the body to leukomethylene blue which is excreted primarily in the urine..Some unchaged drug is also excreted in the urine. (1)
CONTRAINDICATION
Methylene blue can cause fetal harm when administered to a pregnant woman. An association exists between the use of methylene blue in amniocentesis and atresia of the ileum and jejunum, ileal occlusions and other adverse effects in the neonate. (2,3) Methylene blue is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Intraspinal and subcutaneous injections are contraindicated.
Methylene blue is contraindicated in patients with a known hypersensitivity to the drug.
WARNINGS
Methylene Blue should not be given by subcutaneous or intrathecal injection.
Methylene blue is a potent monoamine oxidase inhibitor: Methylene blue has been demonstrated to be a potent monoamine oxidase inhibitor (MAOI) and may cause potentially fatal serotonin toxicity (serotonin syndrome) when combined with serotonin reputake inhibitors (SRIs). (4) (See DRUG INTERACTIONS.) Serotonin toxicity is characterized by development of neuromuscular hyperactivity (tremor, clonus, myoclonus and hyperreflexia, and, in the advanced stage, pyramidal rigidity); autonomic hyperactivity (diaphoresis, fever, tachycardia, tachypnoea, and mydraisis); and altered mental status (agitation, excitement, and in the advanced stage, confusion). If methylene blue is judged to be indicated, SRIs must be ceased, prior to treatment/procedure/surgery.
PRECAUTIONS
Drug Interactions: Methylene blue may interact with any drug that acts as a serotonin reuptake inhibitor (SRI) including, amongst others, selective serotonin reuptake inhbitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), norepinephrine-dopamine reuptake inhibitors (NDRIs), triptans and ergot alkaloids; such combinations may have the consequence of potentially fatal serotonin toxicity (serotonin syndrome). Methylene blue should not be co-administered with any drug that acts as an SRI.
Use in Pregnancy: Pregnancy Category X: Epidemiologic evidence exists that Methylene blue is a teratogen. An association exists between the use of methylene blue in amniocentesis and atresia of the ileum and jejunum, ileal occlusions and other adverse effects in the neonate. (2,3) Methylene blue Injection should not be administered to pregnant women during amniocentesis due to the risk of teratogenicity and other newborn adverse effects (see CONTRAINDICATIONS).
Glucose-6-Phosphate Dehydrogenase Deficiency (G6PD): Methylene blue should be avoided in patients with G6PD deficiency due to the risk of paradoxical methemoglobinemia and hemolysis. (5,6)
Renal Failure: Methylene blue should be used with caution in patients with severe renal impairment (see CLINICAL PHARMACOLOGY).
Methylene Blue must be injected intravenously very slowly over a period of several minutes to prevent local high concentration of the compound from producing additional methemoglobin.
Do not exceed recommended dosage.
ADVERSE REACTIONS
Large intravenous doses of Methylene Blue produce nausea, abdominal and precordial pain, dizziness, headache, profuse sweating, mental confusion and the formation of methemoglobin.
USE IN PREGNANCY
Pregnancy Category X: Epidemiologic evidence exists that Methylene blue is a teratogen. An association exists between the use of methylene blue in amniocentesis and atresia of the ileum and jejunum, ileal occlusions and other adverse effects in the neonate. (2,3) Methylene blue Injection should not be administered to pregnant women during amniocentesis due to the risk of teratogenicity and other newborn adverse effects (see CONTRAINDICATIONS).
DOSAGE AND ADMINISTRATION
0.1 to 0.2 mL per kg body weight (0.045 to 0.09 mL per pound body weight). Inject Methylene Blue intravenously very slowly over a period of several minutes.
Methylene blue must be injected intravenously very slowly over a period of several minutes to prevent local high concentration of the compound from producing additional methemoglobin. Do not exceed recommended dosage.
Parenteral drug products should be inspected visually for particulate matter and discoloration, whenever solution and container permit.
Store at 20 degrees-25 degrees C (68 degrees-77 degrees F); excursions permitted to 15 degrees-30 degrees C (59 degrees-86 degrees F) (See USP Controlled Room Temperature).

REFERENCES:
(1) DiSanto AR, Wagner JG. Pharmacokinetics of highly ionized drugs II: methylene blue-absorption, metabolism, and excretion in man and dog after oral administration. J Pharm Sci. 1972;61:1086-1090
(2) Cragan JD. Teratogen update: methylene blue. Teratology. 1999;60:42-48.
(3) Kidd SA, Lancaster PA, Anderson JC, Boogert A, Fisher CC, Robertson R et al. Fetal death after exposure to methylene bluedye during mid-trimester amniocentesis in twin pregnancy. Prenat Diagn. 1996;16:39-47.
(4) Ramsay RR, Dunford C, Gillman PK. Methylene blue and serotonin toxicity: inhibition of monoamine oxidase A (MAOA) confirms a theoretical prediction. Br J Pharmacol. 2007;152:946-51.
(5) Beutler E. G6PD Deficiency. Blood. 1994;84:3613-3636
(6) Youngster I. Arcavi L. Schechmaster R. Akayzen Y, Popliski H, Shimonov J, Beig S, Berkovitch M. Medications and glucose-6-phosphate dehydrogenase deficiency: an evidence-based review. Drug Saf. 2010;33:713-726
IN0372
Rev. 3/11
MG# 14080
AMERICAN REGENT, INC
Shirley, NY 11967
| METHYLENE BLUE
methylene blue injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - General Injectables & Vaccines, Inc. (108250663) |
