FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
HUMULIN N is indicated to improve glycemic control in adults and pediatric patients with diabetes mellitus.
2 DOSAGE AND ADMINISTRATION
2.1 Important Administration Instructions
Inspect HUMULIN N visually before use. It should not contain particulate matter and should appear uniformly cloudy after mixing. Do not use HUMULIN N if particulate matter is seen.
Use HUMULIN N KwikPen with caution in patients with visual impairment that may rely on audible clicks to dial their dose.
2.2 Route of Administration
HUMULIN N should only be administered subcutaneously.
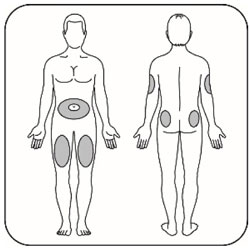
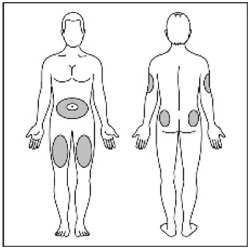
Administer in the subcutaneous tissue of the abdominal wall, thigh, upper arm, or buttocks. Rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy and localized cutaneous amyloidosis. Do not inject into areas of lipodystrophy or localized cutaneous amyloidosis [see Warnings and Precautions (5.2) and Adverse Reactions (6)]. During changes to a patient's insulin regimen, increase the frequency of blood glucose monitoring [see Warnings and Precautions (5.2)].
The HUMULIN N KwikPen dials in 1 unit increments.
Do not administer HUMULIN N intravenously and do not use HUMULIN N in an insulin infusion pump.
2.3 Dosage Information
Individualize and adjust the dosage of HUMULIN N based on the individual's metabolic needs, blood glucose monitoring results and glycemic control goal.
Dosage adjustments may be needed with changes in physical activity, changes in meal patterns (i.e., macronutrient content or timing of food intake), changes in renal or hepatic function or during acute illness [see Warnings and Precautions (5.2, 5.3), and Use in Specific Populations (8.6, 8.7)].
2.4 Dosage Adjustment due to Drug Interactions
Dosage adjustment may be needed when HUMULIN N is coadministered with certain drugs [see Drug Interactions (7)].
Dosage adjustment may be needed when changing from another insulin to HUMULIN N [see Warnings and Precautions (5.2)].
Instructions for Mixing with Other Insulins
HUMULIN N may be used with a prandial insulin if indicated. HUMULIN N may be mixed with HUMULIN R or HUMALOG before injection.
- If HUMULIN N is mixed with HUMULIN R, HUMULIN R should be drawn into the syringe first. Injection should occur immediately after mixing.
- If HUMULIN N is mixed with HUMALOG, HUMALOG should be drawn into the syringe first. Injection should occur immediately after mixing.
3 DOSAGE FORMS AND STRENGTHS
Injectable suspension: 100 units/mL (U-100) white and cloudy suspension available as:
- 10 mL multiple-dose vial
- 3 mL multiple-dose vial
- 3 mL single-patient-use HUMULIN N KwikPen prefilled pen
5 WARNINGS AND PRECAUTIONS
5.1 Never Share a HUMULIN N KwikPen or Syringe Between Patients
HUMULIN N KwikPens must never be shared between patients, even if the needle is changed. Patients using HUMULIN N vials must never share needles or syringes with another person. Sharing poses a risk for transmission of blood-borne pathogens.
5.2 Hyperglycemia or Hypoglycemia with Changes in Insulin Regimen
Changes in an insulin regimen (e.g., insulin strength, manufacturer, type, injection site or method of administration) may affect glycemic control and predispose to hypoglycemia [see Warnings and Precautions (5.3)] or hyperglycemia. Repeated insulin injections into areas of lipodystrophy or localized cutaneous amyloidosis have been reported to result in hyperglycemia; and a sudden change in the injection site (to an unaffected area) has been reported to result in hypoglycemia [see Adverse Reactions (6)].
Make any changes to a patient's insulin regimen under close medical supervision with increased frequency of blood glucose monitoring. Advise patients who have repeatedly injected into areas of lipodystrophy or localized cutaneous amyloidosis to change the injection site to unaffected areas and closely monitor for hypoglycemia. For patients with type 2 diabetes, dosage adjustments of concomitant antidiabetic products may be needed.
5.3 Hypoglycemia
Hypoglycemia is the most common adverse reaction associated with insulins, including HUMULIN N. Severe hypoglycemia can cause seizures, may be life-threatening or cause death. Hypoglycemia can impair concentration ability and reaction time; this may place the patient and others at risk in situations where these abilities are important (e.g., driving or operating other machinery).
Hypoglycemia can happen suddenly and symptoms may differ in each patient and change over time in the same patient. Symptomatic awareness of hypoglycemia may be less pronounced in patients with longstanding diabetes, in patients with diabetic neuropathy, in patients using medications that block the sympathetic nervous system (e.g., beta-blockers) [see Drug Interactions (7)], or in patients who experience recurrent hypoglycemia.
Risk Factors for Hypoglycemia
The risk of hypoglycemia after an injection is related to the duration of action of the insulin and, in general, is highest when the glucose lowering effect of the insulin is maximal. As with all insulins, the glucose lowering effect time course of HUMULIN N may vary in different individuals or at different times in the same individual and depends on many conditions, including the area of injection as well as the injection site blood supply and temperature [see Clinical Pharmacology (12.2)].
Other factors which may increase the risk of hypoglycemia include changes in meal pattern (e.g., macronutrient content or timing of meals), changes in level of physical activity, or changes to concomitant drugs [see Drug Interactions (7)]. Patients with renal or hepatic impairment may be at higher risk of hypoglycemia [see Use in Specific Populations (8.6, 8.7)].
Risk Mitigation Strategies for Hypoglycemia
Patients and caregivers must be educated to recognize and manage hypoglycemia. Self-monitoring of blood glucose plays an essential role in the prevention and management of hypoglycemia. In patients at higher risk for hypoglycemia and patients who have reduced symptomatic awareness of hypoglycemia, increased frequency of blood glucose monitoring is recommended.
5.4 Hypoglycemia Due to Medication Errors
Accidental mix-ups between insulin products have been reported. To avoid medication errors between HUMULIN N and other insulins, instruct patients to always check the insulin label before each injection.
5.5 Hypersensitivity Reactions
Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulins, including HUMULIN N. If hypersensitivity reactions occur, discontinue HUMULIN N; treat per standard of care and monitor until symptoms and signs resolve [see Adverse Reactions (6)]. HUMULIN N is contraindicated in patients who have had hypersensitivity reactions to HUMULIN N or any of its excipients.
5.6 Hypokalemia
All insulins, including HUMULIN N, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Monitor potassium levels in patients at risk for hypokalemia if indicated (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations).
5.7 Fluid Retention and Heart Failure with Concomitant Use of PPAR-gamma Agonists
Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)-gamma agonists, can cause dose-related fluid retention, when used in combination with insulin. Fluid retention may lead to or exacerbate heart failure. Patients treated with insulin, including HUMULIN N, and a PPAR-gamma agonist should be observed for signs and symptoms of heart failure. If heart failure develops, it should be managed according to current standards of care, and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
6 ADVERSE REACTIONS
The following adverse reactions are discussed elsewhere in the labeling:
- Hypoglycemia [see Warnings and Precautions (5.3)].
- Hypoglycemia Due to Medication Errors [see Warnings and Precautions (5.4)].
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Hypokalemia [see Warnings and Precautions (5.6)].
The following additional adverse reactions have been identified during post-approval use of HUMULIN N. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or to establish a causal relationship to drug exposure.
Allergic Reactions
Some patients taking HUMULIN N have experienced erythema, local edema, and pruritus at the site of injection. These conditions were usually self-limiting. Severe cases of generalized allergy (anaphylaxis) have been reported [see Warnings and Precautions (5.5)].
Peripheral Edema
Some patients taking HUMULIN N have experienced sodium retention and edema, particularly if previously poor metabolic control is improved by intensified insulin therapy.
Lipodystrophy
Administration of insulin subcutaneously, including HUMULIN N, has resulted in lipoatrophy (depression in the skin) or lipohypertrophy (enlargement or thickening of tissue) [see Dosage and Administration (2.2)] in some patients.
Localized Cutaneous Amyloidosis
Localized cutaneous amyloidosis at the injection site has occurred. Hyperglycemia has been reported with repeated insulin injections into areas of localized cutaneous amyloidosis; hypoglycemia has been reported with a sudden change to an unaffected injection site.
7 DRUG INTERACTIONS
| Drugs that May Increase the Risk of Hypoglycemia | |
| Drugs: | Antidiabetic agents, ACE inhibitors, angiotensin II receptor blocking agents, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, pentoxifylline, pramlintide, salicylates, somatostatin analog (e.g., octreotide), and sulfonamide antibiotics |
| Intervention: | Dose adjustment and increased frequency of glucose monitoring may be required when HUMULIN N is co-administered with these drugs. |
| Drugs that May Decrease the Blood Glucose Lowering Effect of HUMULIN N | |
| Drugs: | Atypical antipsychotics (e.g., olanzapine and clozapine), corticosteroids, danazol, diuretics, estrogens, glucagon, isoniazid, niacin, oral contraceptives, phenothiazines, progestogens (e.g., in oral contraceptives), protease inhibitors, somatropin, sympathomimetic agents (e.g., albuterol, epinephrine, terbutaline), and thyroid hormones. |
| Intervention: | Dose adjustment and increased frequency of glucose monitoring may be required when HUMULIN N is co-administered with these drugs. |
| Drugs that May Increase or Decrease the Blood Glucose Lowering Effect of HUMULIN N | |
| Drugs: | Alcohol, beta-blockers, clonidine, and lithium salts. Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia. |
| Intervention: | Dose adjustment and increased frequency of glucose monitoring may be required when HUMULIN N is co-administered with these drugs. |
| Drugs that May Blunt Signs and Symptoms of Hypoglycemia | |
| Drugs: | Beta-blockers, clonidine, guanethidine, and reserpine |
| Intervention: | Increased frequency of glucose monitoring may be required when HUMULIN N is co-administered with these drugs. |
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from published studies over decades have not established an association with human insulin use during pregnancy and major birth defects, miscarriage, or adverse maternal or fetal outcomes (see Data). There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy (see Clinical Considerations). Animal reproduction studies were not performed.
The estimated background risk of major birth defects is 6-10% in women with pre-gestational diabetes with a HbA1c >7% and has been reported to be as high as 20-25% in women with a HbA1c >10%. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Disease-associated maternal and/or embryo/fetal risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia-related morbidity.
Data
Human Data
While available studies cannot definitively establish the absence of risk, published data from retrospective studies, open-label, randomized, parallel studies and meta-analyses over decades have not established an association with human insulin use during pregnancy and major birth defects, miscarriage, or adverse maternal or fetal outcomes. All available studies have methodological limitations, including lack of blinding, unclear methods or randomization, and small sample size.
8.2 Lactation
Risk Summary
Available data from published literature suggests that exogenous human insulin products, including HUMULIN N, are transferred into human milk. There are no adverse reactions reported in breastfed infants in the literature. There are no data on the effects of exogenous human insulin products, including HUMULIN N on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for HUMULIN N and any potential adverse effects on the breastfed child from HUMULIN N or from the underlying maternal condition.
8.4 Pediatric Use
HUMULIN N is indicated to improve glycemic control in pediatric patients with diabetes mellitus.
8.5 Geriatric Use
The effect of age on the pharmacokinetics and pharmacodynamics of HUMULIN N has not been studied [see Clinical Pharmacology (12.3)]. Patients with advanced age using any insulin, including HUMULIN N, may be at increased risk of hypoglycemia due to co-morbid disease and polypharmacy [see Warnings and Precautions (5.3)].
8.6 Renal Impairment
The effect of renal impairment on the pharmacokinetics and pharmacodynamics of HUMULIN N has not been studied [see Clinical Pharmacology (12.3)]. Patients with renal impairment are at increased risk of hypoglycemia and may require more frequent HUMULIN N dose adjustment and more frequent blood glucose monitoring.
8.7 Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics and pharmacodynamics of HUMULIN N has not been studied [see Clinical Pharmacology (12.3)]. Patients with hepatic impairment are at increased risk of hypoglycemia and may require more frequent HUMULIN N dose adjustment and more frequent blood glucose monitoring.
10 OVERDOSAGE
Excess insulin administration may cause hypoglycemia and hypokalemia [see Warnings and Precautions (5.3, 5.6)]. Mild episodes of hypoglycemia can be treated with oral glucose. Adjustments in drug dosage, meal patterns, or physical activity level may be needed. More severe episodes with coma, seizure, or neurologic impairment may be treated with a glucagon product for emergency use or concentrated intravenous glucose. Sustained carbohydrate intake and observation may be necessary because hypoglycemia may recur after apparent clinical recovery. Hypokalemia must be corrected appropriately.
11 DESCRIPTION
Insulin isophane human is produced by recombinant DNA technology utilizing a non-pathogenic laboratory strain of Escherichia coli. Insulin isophane human is a suspension of crystals produced from combining human insulin and protamine sulfate under appropriate conditions for crystal formation. The amino acid sequence of insulin isophane human is identical to human insulin and has the empirical formula C257H383N65O77S6 with a molecular weight of 5.808 kDa.
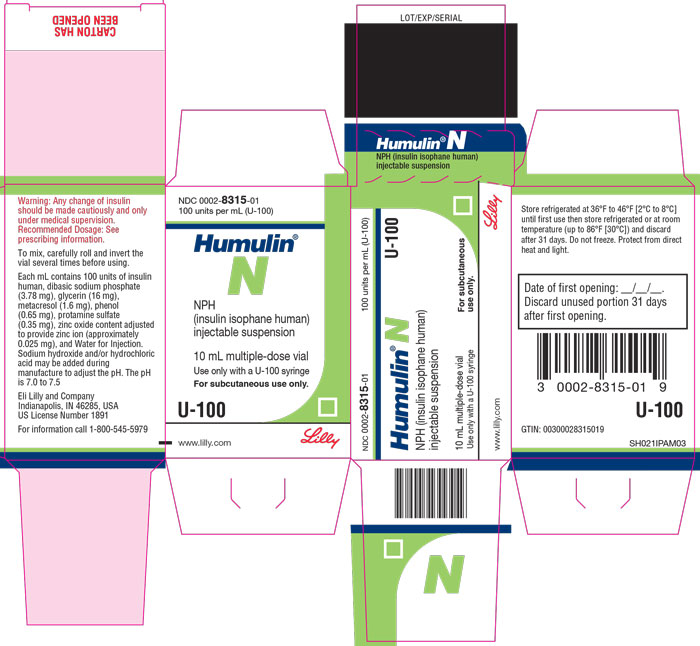
HUMULIN N (insulin isophane human) injectable suspension is an intermediate acting human insulin. It is a sterile, white and cloudy suspension that contains insulin isophane human suspension (NPH) for subcutaneous use. Each milliliter of HUMULIN N contains 100 units of insulin human, dibasic sodium phosphate (3.78 mg), glycerin (16 mg), metacresol (1.6 mg), phenol (0.65 mg), protamine sulfate (0.35 mg), zinc oxide content adjusted to provide zinc ion (approximately 0.025 mg for the vials or approximately 0.035 mg for the KwikPen), and Water for Injection. The pH is 7.0 to 7.5. Sodium hydroxide and/or hydrochloric acid may be added during manufacture to adjust the pH.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
HUMULIN N lowers blood glucose by stimulating peripheral glucose uptake by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulins inhibit lipolysis and proteolysis, and enhance protein synthesis.
12.2 Pharmacodynamics
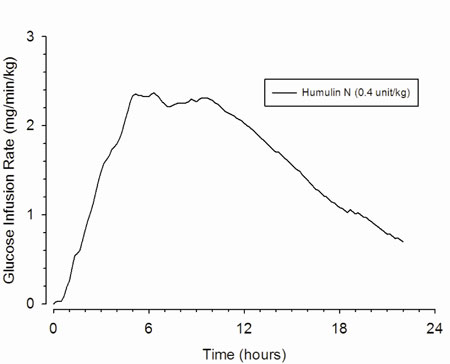
HUMULIN N is an intermediate-acting insulin with a slower onset of action and a longer duration of activity than that of regular human insulin. In a study in which healthy subjects (n=16) received subcutaneous injections of HUMULIN N (0.4 unit/kg) on 4 occasions, the median maximum effect occurred at 6.5 hours (range: 2.8 to 13 hours). In this study, insulin activity was measured by the rate of glucose infusions.
The time course of action of insulin, such as HUMULIN N may vary in different individuals or within the same individual. The parameters of HUMULIN N activity (time of onset, peak time, and duration) as designated in Figure 1 should be considered only as general guidelines. The rate of insulin absorption and consequently the onset of activity is known to be affected by the site of injection, physical activity level, and other variables [see Warnings and Precautions (5.3)].
12.3 Pharmacokinetics
Absorption — In healthy subjects given subcutaneous doses of HUMULIN N (0.4 unit/kg), median peak serum concentration of insulin occurred at approximately 4 hours (range: 1 to 12 hours) after dosing.
Metabolism — The uptake and degradation of insulin occurs predominantly in liver, kidney, muscle, and adipocytes, with the liver being the major organ involved in the clearance of insulin.
Elimination — Because of the absorption-rate limited kinetics of insulin mixtures, a true half-life cannot be accurately estimated from the terminal slope of the concentration versus time curve. In healthy subjects given subcutaneous doses of HUMULIN N (0.4 unit/kg), the mean apparent half-life was approximately 4.4 hours (range: 1-84 hours).
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
HUMULIN N (insulin isophane human) injectable suspension, 100 units/mL (U-100), is a white and cloudy suspension available as:
| 10 mL multiple-dose vial | NDC 0002-8315-01 (HI-310) |
| 3 mL multiple-dose vial | NDC 0002-8315-17 (HI-313) |
| 5 x 3 mL single-patient-use HUMULIN N KwikPen | NDC 0002-8805-59 (HP-8805) |
Each prefilled HUMULIN N KwikPen is for use by a single patient. HUMULIN N KwikPens must never be shared between patients, even if the needle is changed. Patients using HUMULIN N vials must never share needles or syringes with another person.
The HUMULIN N KwikPen dials in 1 unit increments.
16.2 Storage and Handling
Dispense in the original sealed carton with the enclosed Instructions for Use.
Protect from heat and light. Do not freeze. Do not use if it has been frozen. See Table 2 below for storage conditions.
|
a When stored at room temperature, HUMULIN N vial can only be used for a total of 31 days including both not in-use (unopened) and in-use (opened) storage time. |
||||
|
b When stored at room temperature, HUMULIN N KwikPen can only be used for a total of 14 days including both not in-use (unopened) and in-use (opened) storage time. |
||||
| Not In-use (Unopened) | In-use (Opened) |
|||
| Room Temperature (up to 86°F [30°C]) | Refrigerated (36° to 46°F [2° to 8°C]) | Room Temperature (up to 86°F [30°C]) | Refrigerated (36° to 46°F [2° to 8°C]) |
|
| 10 mL multiple-dose viala
3 mL multiple-dose viala | 31 days | Until expiration date | 31 days | 31 days |
| 3 mL single-patient-use HUMULIN N KwikPenb | 14 days | Until expiration date | 14 days | Do not refrigerate. |
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
Never Share a HUMULIN N KwikPen or Syringe Between Patients
Advise patients that they must never share a HUMULIN N KwikPen with another person, even if the needle is changed. Advise patients using HUMULIN N vials not to share needles or syringes with another person. Sharing poses a risk for transmission of blood-borne pathogens [see Warnings and Precautions (5.1)].
Hyperglycemia or Hypoglycemia
Instruct patients on self-management procedures including glucose monitoring, proper injection technique, and management of hypoglycemia and hyperglycemia especially at initiation of HUMULIN N therapy. Instruct patients on handling of special situations such as intercurrent conditions (illness, stress, or emotional disturbances), an inadequate or skipped insulin dose, inadvertent administration of an increased insulin dose, inadequate food intake, and skipped meals. Instruct patients on the management of hypoglycemia.
Inform patients that their ability to concentrate and react may be impaired as a result of hypoglycemia. Advise patients who have frequent hypoglycemia or reduced or absent warning signs of hypoglycemia to use caution when driving or operating machinery [see Warnings and Precautions (5.3)].
Advise patients that changes in insulin regimen can predispose to hyperglycemia or hypoglycemia and that changes in insulin regimen should be made under close medical supervision [see Warnings and Precautions (5.2)].
Inform patients that accidental mix-ups between insulin products have been reported. Instruct patients to always carefully check that they are administering the correct insulin (e.g., by checking the insulin label before each injection) to avoid medication errors between HUMULIN N and other insulins.
Hypersensitivity Reactions
Advise patients that hypersensitivity reactions have occurred with HUMULIN N. Inform patients on the symptoms of hypersensitivity reactions [see Warnings and Precautions (5.5)].
PATIENT INFORMATION
HUMULIN®
(HU-mu-lin) N
(insulin isophane human)
injectable suspension, for subcutaneous use
Do not share your HUMULIN N KwikPen or syringes with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
What is HUMULIN N?
- HUMULIN N is a man-made insulin that is used to control high blood sugar in adults and children with diabetes mellitus.
Who should not use HUMULIN N?
Do not use HUMULIN N if you:
- are having an episode of low blood sugar (hypoglycemia).
- have an allergy to HUMULIN N or any of the ingredients in HUMULIN N.
Before using HUMULIN N, tell your healthcare provider about all your medical conditions including, if you:
- have liver or kidney problems.
- take any other medicines, especially ones commonly called TZDs (thiazolidinediones).
- have heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with HUMULIN N.
- are pregnant, planning to become pregnant. Talk with your healthcare provider about the best way to control your blood sugar if you plan to become pregnant or while you are pregnant.
- are breast-feeding or plan to breastfeed. HUMULIN N may pass into your breast milk. Talk with your healthcare provider about the best way to feed your baby while using HUMULIN N.
- are taking new prescription or over-the-counter medicines, vitamins, or herbal supplements.
Before you start using HUMULIN N, talk to your healthcare provider about low blood sugar and how to manage it.
How should I use HUMULIN N?
- Read the Instructions for Use that come with your HUMULIN N.
- Use HUMULIN N exactly as your healthcare provider tells you to. HUMULIN N is injected under the skin (subcutaneously) of your stomach area, buttocks, upper legs or upper arms. Change (rotate) your injection sites within the area you choose with each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not use the exact same spot for each injection. Do not inject where the skin has pits, is thickened, or has lumps. Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Know the type and strength of insulin you use. Do not change the type of insulin you use unless your healthcare provider tells you to. The amount of insulin and the best time for you to take your insulin may need to change if you use different types of insulin.
- Check your blood sugar levels. Ask your healthcare provider what your blood sugars should be and when you should check your blood sugar levels.
- Do not share your HUMULIN N KwikPen or syringes with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
Your HUMULIN N dose may need to change because of:
- change in level of physical activity or exercise, weight gain or loss, increased stress, illness, change in diet.
What should I avoid while using HUMULIN N?
While using HUMULIN N do not:
- drive or operate heavy machinery, until you know how HUMULIN N affects you.
- drink alcohol or use prescription or over-the-counter medicines that contain alcohol.
What are the possible side effects of HUMULIN N?
HUMULIN N may cause serious side effects that can lead to death, including:
-
low blood sugar (hypoglycemia). Signs and symptoms that may indicate low blood sugar include:
- dizziness or light-headedness, sweating, confusion, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability, or mood changes, hunger.
-
serious allergic reaction (whole body reaction). Get medical help right away, if you have any of these symptoms of an allergic reaction:
- a rash over your whole body, trouble breathing, a fast heartbeat, or sweating.
- low potassium in your blood (hypokalemia).
- heart failure. Taking certain diabetes pills called thiazolidinediones or “TZDs” with HUMULIN N may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you already have heart failure it may get worse while you take TZDs with HUMULIN N. Your healthcare provider should monitor you closely while you are taking TZDs with HUMULIN N. Tell your healthcare provider if you have any new or worse symptoms of heart failure including:
- shortness of breath, swelling of your ankles or feet, sudden weight gain.
Treatment with TZDs and HUMULIN N may need to be adjusted or stopped by your healthcare provider if you have new or worse heart failure.
Get emergency medical help if you have:
- trouble breathing, shortness of breath, fast heartbeat, swelling of your face, tongue, or throat, sweating, extreme drowsiness, dizziness, confusion.
The most common side effects of HUMULIN N include:
- low blood sugar (hypoglycemia), allergic reactions including reactions at the injection site, skin thickening or pits at the injection site (lipodystrophy), itching, rash, weight gain, and swelling of your hands and feet.
These are not all the possible side effects of HUMULIN N. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of HUMULIN N:
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about HUMULIN N that is written for health professionals. Do not use HUMULIN N for a condition for which it was not prescribed. Do not give HUMULIN N to other people, even if they have the same symptoms that you have. It may harm them.
What are the ingredients in HUMULIN N?
Active Ingredient: insulin human
Inactive Ingredients: dibasic sodium phosphate, glycerin, metacresol, phenol, protamine sulfate, zinc oxide, and Water for Injection as inactive ingredients. Sodium hydroxide and/or hydrochloric acid may be added to adjust the pH.
For more information, call 1-800-545-5979 or go to www.humulin.com.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Patient Information revised June 2022
Manufactured by:
Eli Lilly and Company, Indianapolis, IN 46285, USA
US License Number 1891
Copyright © 1997, 2022, Eli Lilly and Company. All rights reserved.
LINN-0005-PPI-20220627
Instructions for Use
HUMULIN® (HU-mu-lin) N
(insulin isophane human)
injectable suspension, for subcutaneous use
3 mL or 10 mL multiple-dose vial (100 units/mL)
Read the Instructions for Use before you start taking HUMULIN N and each time you get a new HUMULIN N vial. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your syringes with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
Supplies needed to give your injection:
- a 3 mL or 10 mL multiple-dose HUMULIN N vial
- a U-100 insulin syringe and needle
- 2 alcohol swabs
- 1 sharps container for throwing away used needles and syringes. See “Disposing of used needles and syringes” at the end of these instructions.
| Vial | Syringe |
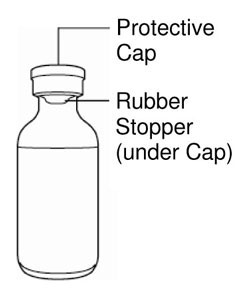 |
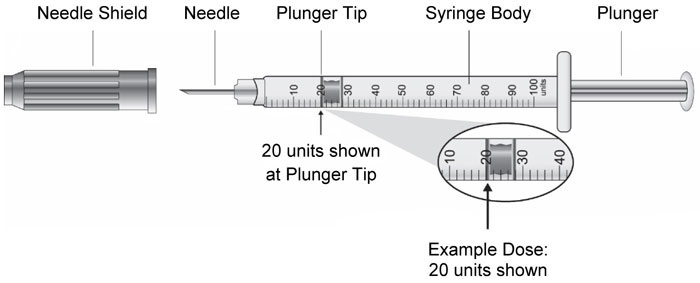 |
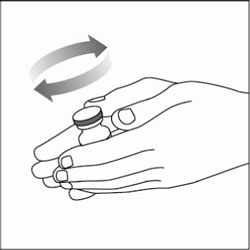
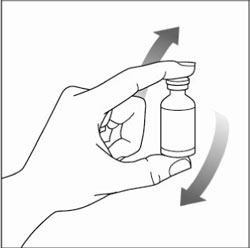
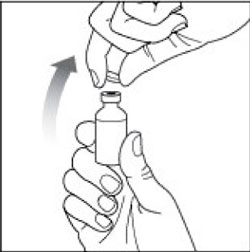
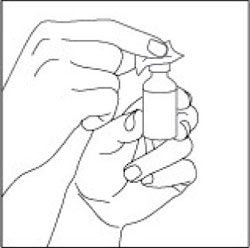
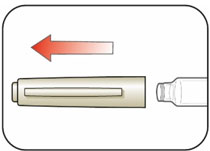
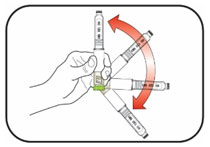
Preparing your HUMULIN N dose:
- Wash your hands with soap and water.
- Check the HUMULIN N label to make sure you are taking the right type of insulin. This is especially important if you use more than 1 type of insulin.
- Do not use HUMULIN N past the expiration date printed on the label or 31 days after you first use it.
- Always use a new syringe or needle for each injection to help ensure sterility and prevent blocked needles. Do not reuse or share your syringes or needles with other people. You may give other people a serious infection or get a serious infection from them.
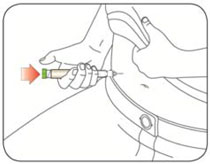
Giving your HUMULIN N injection:
- Inject your insulin exactly as your healthcare provider has shown you.
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not inject where the skin has pits, is thickened, or has lumps. Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
Disposing of used needles and syringes:
- Put your used needles and syringes in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- -
- made of a heavy-duty plastic,
- -
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- -
- upright and stable during use,
- -
- leak-resistant, and
- -
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
How should I store HUMULIN N?
All unopened HUMULIN N vials:
- Store all unopened vials in the refrigerator.
- Do not freeze. Do not use if it has been frozen.
- Keep away from heat and out of direct light.
- Unopened vials can be used until the expiration date on the carton and label, if they have been stored in the refrigerator.
- Unopened vials should be thrown away after 31 days, if they are stored at room temperature.
After HUMULIN N vials have been opened:
- Store opened vials in the refrigerator or at room temperature up to 86°F (30°C) for up to 31 days.
- Keep away from heat and out of direct light.
- Throw away all opened vials after 31 days of use, even if there is still insulin left in the vial.
General information about the safe and effective use of HUMULIN N.
- Keep HUMULIN N vials, syringes, needles, and all medicines out of the reach of children.
- Always use a new syringe or needle for each injection.
- Do not reuse or share your syringes or needles with other people. You may give other people a serious infection or get a serious infection from them.
If you have any questions or problems with your HUMULIN, contact Lilly at 1-800-Lilly-Rx (1-800-545-5979) or call your healthcare provider for help. For more information on HUMULIN and insulin, go to www.humulin.com.
Scan this code to launch the humulin.com website
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Humulin® is a trademark of Eli Lilly and Company.
Instructions for Use revised: June 2022
Manufactured by:
Eli Lilly and Company, Indianapolis, IN 46285, USA
US License Number 1891
Copyright © 1992, 2022, Eli Lilly and Company. All rights reserved.
LINNVL-0005-IFU-20220627
Instructions for Use
HUMULIN® N KwikPen®
(insulin isophane human)
injectable suspension, for subcutaneous use
3 mL single-patient-use pen (100 units/mL)
Read the Instructions for Use before you start taking HUMULIN N and each time you get another KwikPen. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Do not share your HUMULIN N KwikPen with other people, even if the needle has been changed. You may give other people a serious infection or get a serious infection from them.
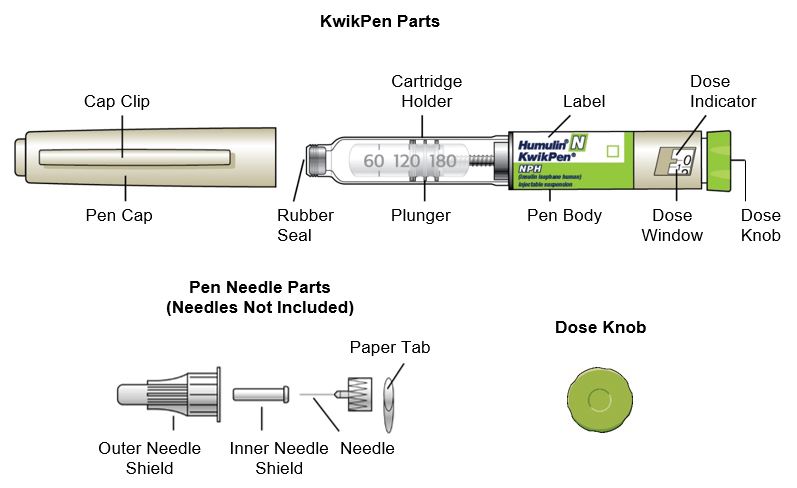
HUMULIN® N KwikPen® (“Pen”) is a disposable single-patient-use prefilled pen containing 300 units of HUMULIN N. You can give yourself more than 1 dose from the Pen. Each turn (click) of the Dose Knob dials 1 unit of insulin. You can give from 1 to 60 units in a single injection. If your dose is more than 60 units, you will need to give yourself more than 1 injection. The Plunger only moves a little with each injection, and you may not notice that it moves. The Plunger will only reach the end of the cartridge when you have used all 300 units in the Pen.
People who are blind or have vision problems should not use the Pen without help from a person trained to use the Pen.
How to recognize your HUMULIN N KwikPen
- Pen color: Beige
- Dose Knob: Light green
- Labels: White label with light green stripe
Supplies you will need to give your injection
- HUMULIN N KwikPen
- KwikPen compatible Needle (Becton, Dickinson and Company Pen Needles recommended)
- Alcohol swab
- Gauze
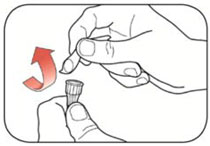
Preparing your Pen
- Wash your hands with soap and water.
- Check your Pen to make sure you are taking the right type of insulin. This is especially important if you use more than 1 type of insulin.
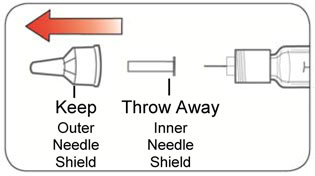
- Do not use your Pen past the expiration date printed on the Label or for more than 14 days after you first start using the Pen.
- Always use a new needle for each injection to help prevent infections and blocked needles. Do not reuse or share your needles with other people. You may give other people a serious infection or get a serious infection from them.
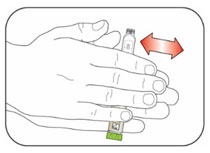
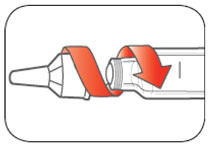
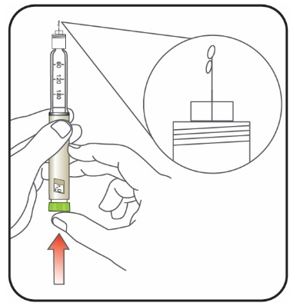
Priming your Pen
Prime before each injection.
- Priming your Pen means removing the air from the Needle and Cartridge that may collect during normal use and ensures that the Pen is working correctly.
- If you do not prime before each injection, you may get too much or too little insulin.
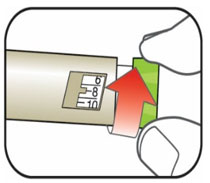
Selecting your dose
- You can give from 1 to 60 units in a single injection.
- If your dose is more than 60 units, you will need to give more than 1 injection.
- -
- If you need help with dividing up your dose the right way, ask your healthcare provider.
- -
- Use a new Needle for each injection and repeat the priming step.
- The Pen will not let you dial more than the number of units left in the Pen.
- If you need to inject more than the number of units left in the Pen, you may either:
- -
- inject the amount left in your Pen and then use a new Pen to give the rest of your dose,
or
- -
- get a new Pen and inject the full dose.
- It is normal to see a small amount of insulin left in the Pen that you can not inject.
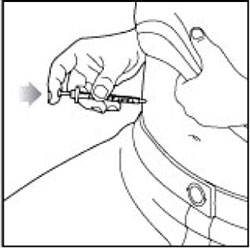
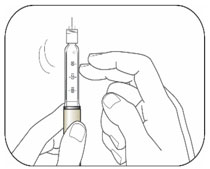
Giving your injection
- Inject your insulin as your healthcare provider has shown you.
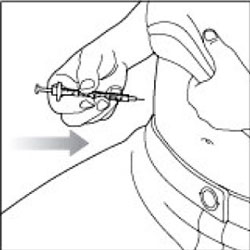
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not inject where the skin has pits, is thickened, or has lumps. Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not try to change your dose while injecting.
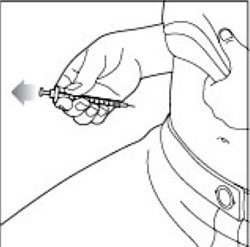
After your injection
Step 15:
|
 |
Step 16:
|
 |
Step 17:
|
 |
- Put your used needles in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- -
- made of a heavy-duty plastic,
- -
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- -
- upright and stable during use,
- -
- leak-resistant, and
- -
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- The used Pen may be discarded in your household trash after you have removed the needle.
Storing your Pen
Unused Pens
- Store unused Pens in the refrigerator at 36°F to 46°F (2°C to 8°C).
- Do not freeze your insulin. Do not use if it has been frozen.
- Unused Pens may be used until the expiration date printed on the Label, if the Pen has been kept in the refrigerator.
In-use Pen
- Store the Pen you are currently using at room temperature [up to 86°F (30°C)]. Keep away from heat and light.
- Throw away the HUMULIN N Pen you are using after 14 days, even if it still has insulin left in it.
General information about the safe and effective use of your Pen
- Keep your Pen and needles out of the reach of children.
- Do not use your Pen if any part looks broken or damaged.
- Always carry an extra Pen in case yours is lost or damaged.
Troubleshooting
- If you can not remove the Pen Cap, gently twist the cap back and forth, and then pull the cap straight off.
- If the Dose Knob is hard to push:
- -
- Pushing the Dose Knob more slowly will make it easier to inject.
- -
- Your Needle may be blocked. Put on a new Needle and prime the Pen.
- -
- You may have dust, food, or liquid inside the Pen. Throw the Pen away and get a new Pen.
If you have any questions or problems with your HUMULIN N KwikPen, contact Lilly at 1-800-LillyRx (1-800-545-5979) or call your healthcare provider for help. For more information on HUMULIN N KwikPen and insulin, go to www.lilly.com.
Scan this code to launch
www.humulin.com
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
HUMULIN® and HUMULIN® KwikPen® are trademarks of Eli Lilly and Company.
Revised: June 2022
Manufactured by:
Eli Lilly and Company
Indianapolis, IN 46285, USA
US License Number 1891
Copyright © 2013, 2022, Eli Lilly and Company. All rights reserved.
| HUMULIN N KwikPen meets the current dose accuracy and functional requirements of ISO 11608-1. |
LINNKP-0008-IFU-20220627
PACKAGE CARTON – HUMULIN N Vial 10 mL 1ct
NDC 0002-8315-01
100 units per mL (U-100)
Humulin® N
NPH
(insulin isophane human)
injectable suspension
10 mL multiple-dose vial
Use only with a U-100 syringe
For subcutaneous use only.
U-100
www.lilly.com
Lilly
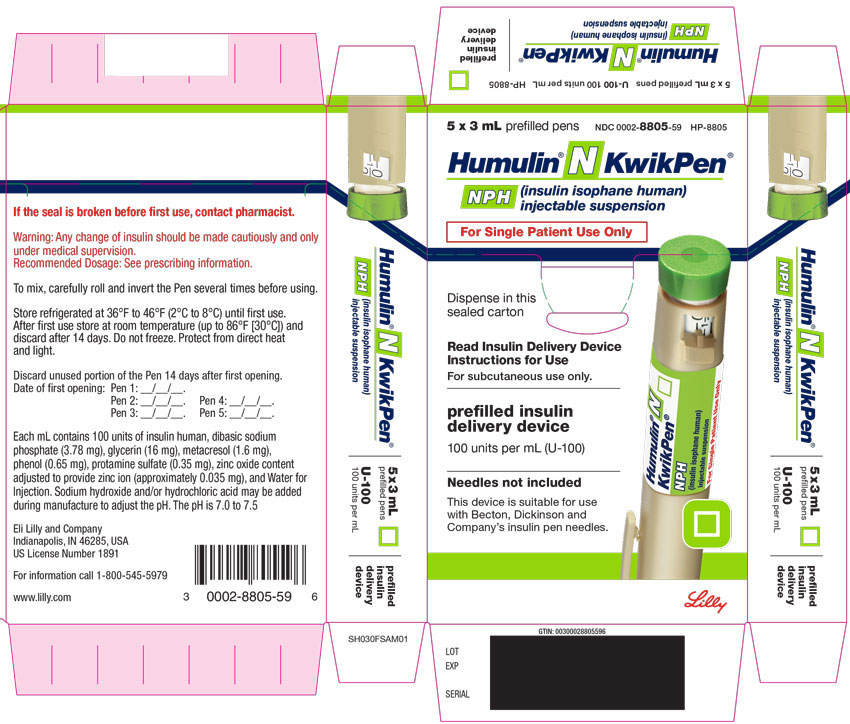
PACKAGE LABEL – Humulin N KwikPen 3mL 5ct
5 x 3 mL prefilled pens
NDC 0002-8805-59
HP-8805
Humulin® N KwikPen®
NPH
(insulin isophane human)
injectable suspension
For Single Patient Use Only
Dispense in this sealed carton
Read Insulin Delivery Device Instructions for Use
For subcutaneous use only.
prefilled insulin delivery device
100 units per mL (U-100)
Needles not included
This device is suitable for use with Becton, Dickinson and Company's insulin pen needles.
Lilly
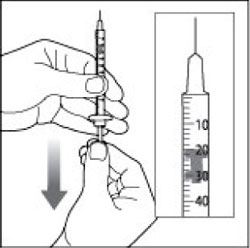 (Example Dose: 20 units shown)
(Example Dose: 20 units shown)
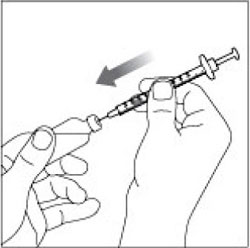
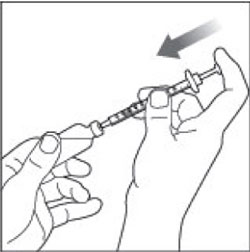
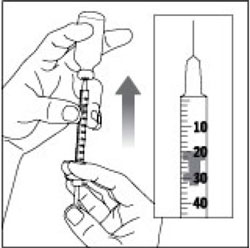
 (Example Dose: 20 units Plunger is shown at 24 units)
(Example Dose: 20 units Plunger is shown at 24 units)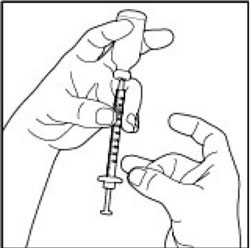
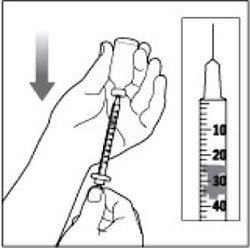
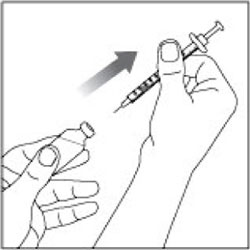
 (Example Dose: 20 units shown)
(Example Dose: 20 units shown)


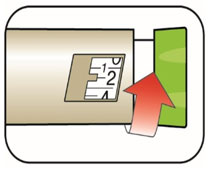
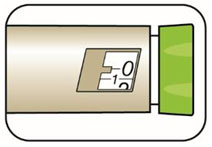
 (Example: 12 units shown in the Dose Window)
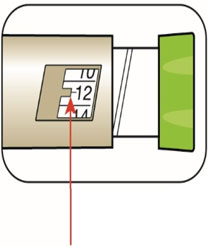
(Example: 12 units shown in the Dose Window)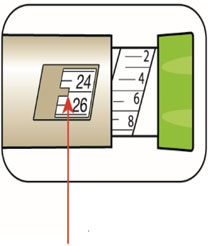 (Example: 25 units shown in the Dose Window)
(Example: 25 units shown in the Dose Window)