Uses
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- temporarily relieves sinus congestion and pressure
Warnings
Do not use
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- diabetes
- thyroid disease
- heart disease
- high blood pressure
- trouble urinating due to enlargement of the prostate gland
Directions
- adults and children 12 years and older: take 1 tablet every 4 to 6 hours. Do not take more than 4 tablets in 24 hours.
- children under 12 years of age: do not use
Other information
- store at controlled room temperature 15˚-30˚C (59˚-86˚F)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, hypromellose, lactose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, silicon dioxide, stearic acid
Principal display panel
The product packaging shown below represents a sample of that currently in use. Additional packaging may also be available
MAJOR®
FOR PHARMACY USE ONLY.
NOT FOR RETAIL SALE.
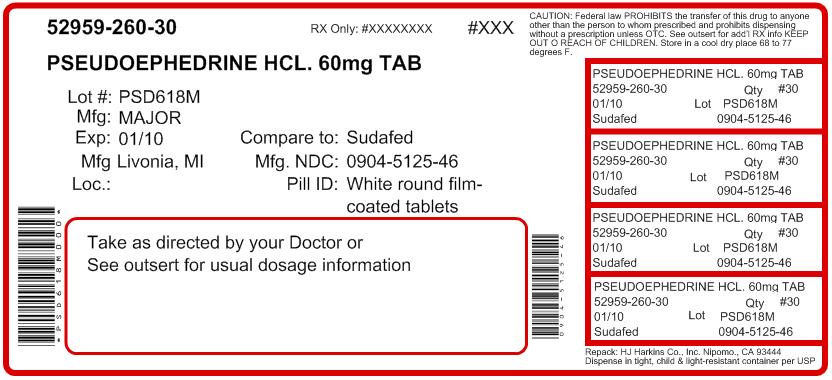
NDC 0904-5125-59
SudoGest™
NASAL DECONGESTANT 60 mg
Pseudoephedrine Hydrochloride 60 mg
Relieves Nasal and Sinus Congestion due to Colds or Hay Fever
Without Drowsiness
100 TABLETS
TAMPER EVIDENT: DO NOT USE IF IMPRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
50844 Rev. 0807L11302
Distributed by Major Pharmaceuticals
31778 Enterprise Drive
Livonia, MI 48150 USA M-17
Repacked by H.J. Harkins Company, Inc.
Nipomo, CA 93444
Rev. 10/11 Re-order No. 700324
Product Packaging