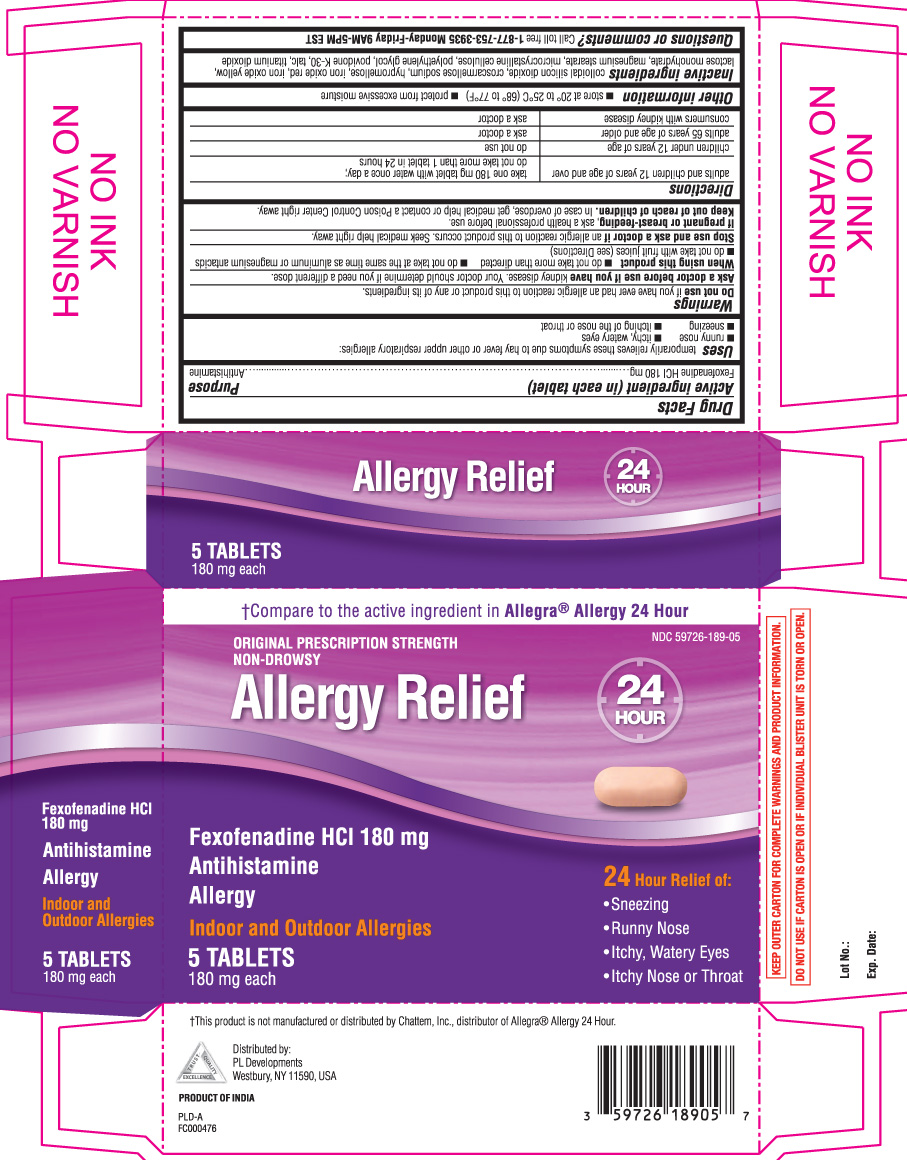
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have
kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Directions
| adults and children 12 years of age and over | take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Inactive ingredients
colloidal silicon dioxide, croscarmellose sodium, hypromellose, iron oxide red, iron oxide yellow, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone k-30, talc, titanium dioxide.
Package/Label Principal Display Panel
†Compare to the active ingredient in Allegra® Allergy 24 hour
ORIGINAL PRESCRIPTION STRENGTH
NON-DROWSY
ALLERGY RELIEF
Fexofenadine HCl 180 mg
Antihistamine
ALLERGY
Indoor and outdoor Allergies
24 Hour Relief of:
- sneezing
- runny nose
- itchy, watery eyes
- itchy nose or throat
†This product is not manufactured or distributed by Chattem Inc., distributor of Allegra® Allergy 24 hour
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DO NOT USE IF CARTON IS OPEN OR IF INDIVIDUAL BLISTER UNIT IS TORN OR OPEN.
Distributed by: PL Developments
200 hicks street
westbury NY 11590
PRODUCT OF INDIA