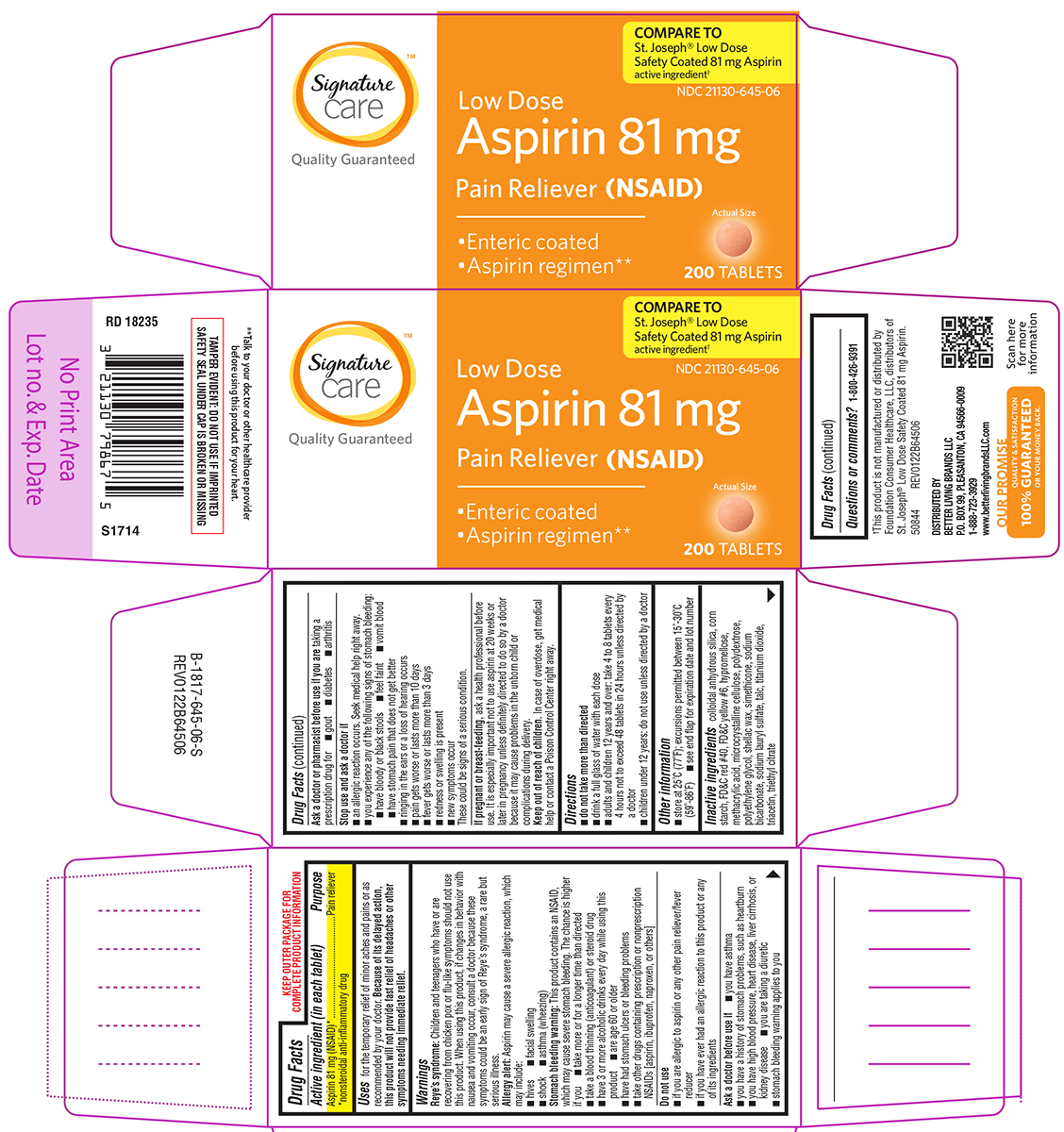
Uses
for the temporary relief of minor aches and pains or as recommended by your doctor. Because of its delayed action, this product will not provide fast relief of headaches or other symptoms needing immediate relief.
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include:
- hives
- facial swelling
- shock
- asthma (wheezing)
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- take more or for a longer time than directed
- take a blood thinning (anticoagulant) or steroid drug
- have 3 or more alcoholic drinks every day while using this product
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
Do not use
- if you are allergic to aspirin or any other pain reliever/fever reducer
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if
- you have asthma
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you are taking a diuretic
- stomach bleeding warning applies to you
Ask a doctor or pharmacist before use if you are
taking a prescription drug for
- gout
- diabetes
- arthritis
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- have bloody or black stools
- feel faint
- vomit blood
- have stomach pain that does not get better
- ringing in the ears or a loss of hearing occurs
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
These could be signs of a serious condition.
Directions
-
do not take more than directed
- drink a full glass of water with each dose
- adults and children 12 years and over: take 4 to 8 tablets every 4 hours not to exceed 48 tablets in 24 hours unless directed by a doctor
- children under 12 years: do not use unless directed by a doctor
Other information
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
colloidal anhydrous silica, corn starch, FD&C red #40, FD&C yellow #6, hypromellose, methacrylic acid, microcrystalline cellulose, polydextrose, polyethylene glycol, shellac wax, simethicone, sodium bicarbonate, sodium lauryl sulfate, talc, titanium dioxide, triacetin, triethyl citrate
Principal Display Panel
Signature™
care
Quality Guaranteed
COMPARE TO
St. Joseph® Low Dose
Safety Coated 81 mg Aspirin
active ingredient†
NDC 21130-645-06
Low Dose
Aspirin 81 mg
Pain Reliever (NSAID)
______________________
• Enteric coated
• Aspirin regimen**
Actual Size
200 TABLETS
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
**Talk to your doctor or other healthcare provider
before using this product for your heart.
†This product is not manufactured or distributed by
Foundation Consumer Healthcare, LLC, distributors of
St. Joseph® Low Dose Safety Coated 81 mg Aspirin.
50844 REV0122B64506
DISTRIBUTED BY
BETTER LIVING BRANDS LLC
P.O. BOX 99, PLEASANTON, CA 94566-0009
1-888-723-3929
www.betterlivingbrandsLLC.com
OUR PROMISE
QUALITY & SATISFACTION
100% GUARANTEED
OR YOUR MONEY BACK.
Signature Care 44-645