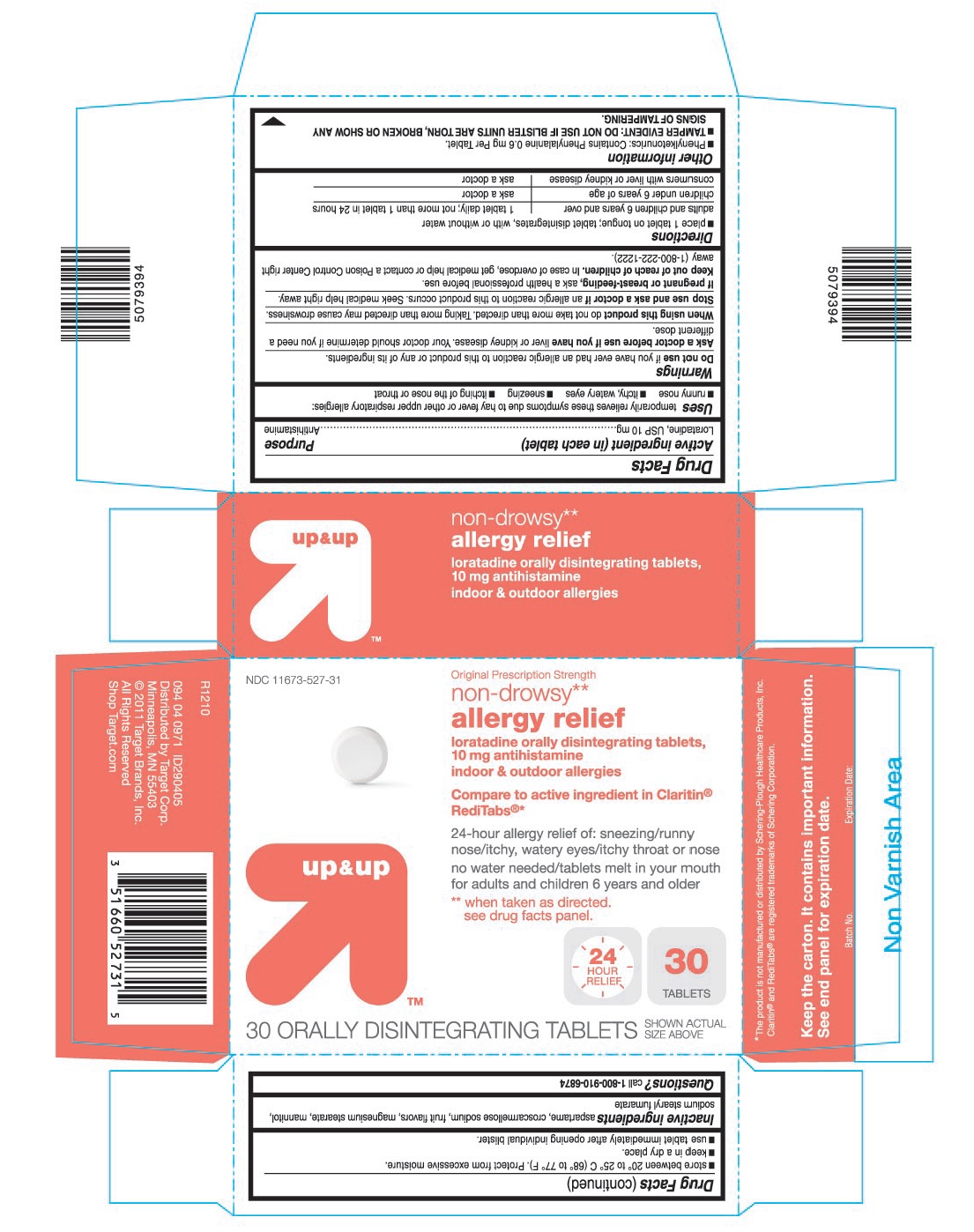
USES
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
WARNINGS
Ask a doctor before use if you have
Liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
Do not take more than directed. Taking more than directed may cause drowsiness.
OTHER INFORMATION
- Phenylketonurics: Contains Phenylalanine 0.6 mg Per Tablet.
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
- store between 20° to 25° C (68° to 77° F). Protect from excessive moisture.
- keep in a dry place.
- use tablet immediately after opening individual blister.
INACTIVE INGREDIENTS
aspartame, croscarmellose sodium, fruit flavors, magnesium stearate, mannitol, sodium stearyl fumarate
PRINCIPAL DISPLAY PANEL
Original Prescription Strength
loratadine orally disintegrating tablets, 10 mg
Compare to active ingredient in Alavert®*
24-hour allergy relief of: sneezing/
runny nose/itchy nose and throat
no water needed/tablets melt in your mouth
for adults and children 6 years and older
12 ORALLY DISINTEGRATING TABLETS
Distributed by Target Corporation
Original Prescription Strength
loratadine orally disintegrating tablets,
Compare to active ingredient in Claritin®Reditabs®*
24-hour allergy relief of: sneezing/runny
nose/itchy, watery eyes/itchy throat or nose
no water needed/tablets melt in your mouth
for adults and children 6 years and older