Uses
For the temporary relief of minor aches and pains of muscles and joints associated with simple backache, arthritis, strains, bruises, and sprains.
Warnings
For external use only
When using this product
- use only as directed
- avoid contact with the eyes
- do not apply to wounds or damaged skin
- do not bandage tightly
Stop use and ask a doctor if condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
- Children under 2 years of age: Consult a doctor.
Inactive ingredients
activated charcoal, disodium EDTA, ethanol, gelatin, glycerin, polysorbate 80, polyvinyl alcohol, salt, sodium polyacrylate, tartaric acid, water
HYDROGEL PAIN RELIEF PATCH with MENTHOL
- Soothing relief for painful joints and muscles
- Lasts up to 8 hours
- Just peel and stick
- Contains activated charcoal for smoother skin!
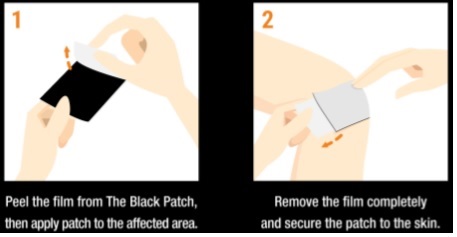
Care Instructions
- The hydrogel must contact the skin for best results. Clean the skin of oil, dirt, lotions, etc. and dry completely.
- It is sometimes helpful to use medical tape around the edges or a tension bandage to hold the patch securely in place.
- The Black Patch is designed to apply easily and remove residue-free. However, due to heat, excessive moisture, or other factors, some hydrogel residue may transfer.
- If residue transfers onto the skin or clothing, simply wash off with warm water or in the laundry.
- The Black Patch is proudly made in America with natural, food grade ingredients. However, it's not edible so keep it out of reach of small children.
Manufactured by
Carbon Innovation
600 Business Park Dr
Lincoln, CA 95648
