SUPPLEMENT FACTS
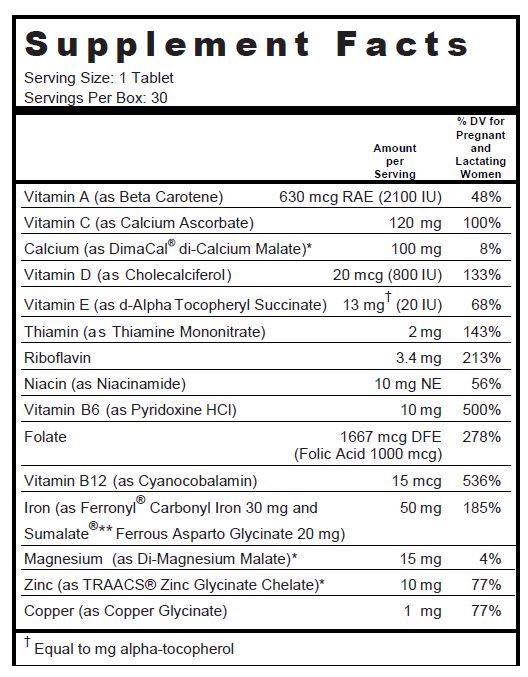
Other Ingredients: Microcrystalline Cellulose, DiCal Phosphate, Croscarmellose Sodium, Pink Color Coating (Hydroxypropylmethyl Cellulose, Polyvinyl Alcohol, Titanium Dioxide, Polyethylene Glycol, Talc and FDC Red #40 Lake), Fumed Silica, Stearic Acid, Magnesium Stearate, Acacia and Povidone K30.
VEGETARIAN FRIENDLY
GLUTEN-, LACTOSE- AND SUGAR-FREE
OB Complete® Premier is a prescription multivitamin/multimineral indicated for use in improving the nutritional status of women prior to conception, throughout pregnancy, and in the postnatal period for both lactating and non-lactating mothers.
CONTRAINDICATIONS
OB Complete® Premier should not be used by patients with a known hypersensitivity to any of the listed ingredients.
WARNINGS
| WARNING: Accidental overdose of iron-containing products is a leading cause of fatal poisoning in children under 6. KEEP OUT OF REACH OF CHILDREN. In case of accidental overdose, call a doctor or poison control center immediately. |
PRECAUTIONS
Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where vitamin B12 is deficient. Folic acid in doses above 0.1 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurologic manifestations remain progressive. Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
The patient’s medical conditions and consumption of other drugs, herbs, and/or supplements should be considered.
DRUG INTERACTIONS
OB Complete® Premier tablets are not recommended for and should not be given to patients receiving levodopa because the action of levodopa is antagonized by pyridoxine. There is a possibility of increased bleeding due to pyridoxine interaction with anticoagulants (e.g., Aspirin, Heparin, Clopidogrel).
ADVERSE REACTIONS
Adverse reactions have been reported with specific vitamins and minerals but generally at levels substantially higher than those contained herein. However, allergic and idiosyncratic reactions are possible at lower levels. Iron, even at the usual recommended levels, has been associated with gastrointestinal intolerance in some patients.
HOW SUPPLIED
OB Complete® Premier is supplied in a carton of 30 tablets, packaged in 3 child resistant unit-dose blister cards of 10 tablets per card.
Product Code: 68025-043-30
STORAGE
Store at controlled room temperature 15°-30°C (59°-86°F) [See USP]. Protect from light, moisture and excessive heat.
KEEP THIS PRODUCT OUT OF REACH OF CHILDREN.
*DimaCal and TRAACS are registered trademarks of Albion International, Inc. Malates covered by U.S. Patent No. 6,706,904. Chelate covered by U.S. Patent No. 7,838,042 and patents pending.
**Sumalate® (Ferrous Asparto Glycinate) is a registered trademark of Albion International, Inc. and is covered by the following patents: U.S. Pat. No. 6,716,814; U.S. Pat. No. 8,007,846 and U.S. Pat. No. 8,425,956.
For use on the order of a healthcare practitioner
Call your doctor about side effects. To report side effects, call Vertical Pharmaceuticals at 1-770-509-4500 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
PLR-OBCPR-00005-2 Rev. 02/2022
Distributed by:
Vertical Pharmaceuticals, LLC
Alpharetta, GA 30005

