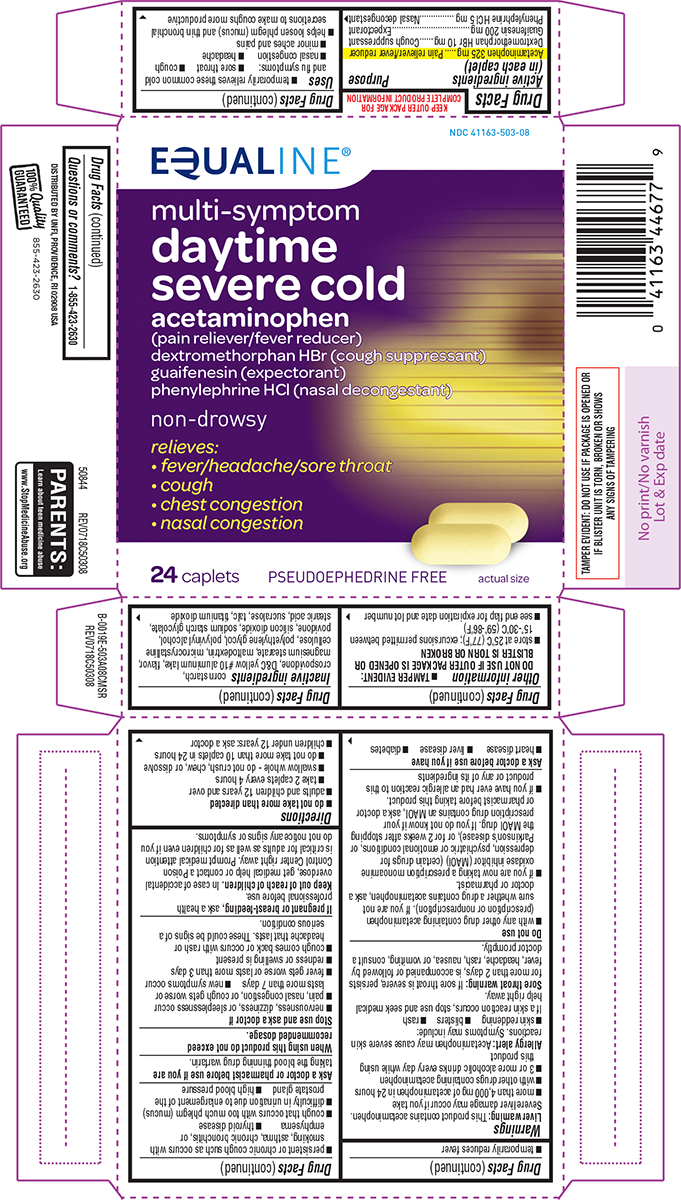
Active ingredients (in each caplet)
Acetaminophen 325 mg
Dextromethorphan HBr 10 mg
Guaifenesin 200 mg
Phenylephrine HCl 5 mg
Uses
- temporarily relieves these common cold and flu symptoms:
- sore throat
- cough
- nasal congestion
- headache
- minor aches and pains
- helps loosen phlegm (mucus) and thin bronchial secretions to make coughs more productive
- temporarily reduces fever
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- heart disease
- liver disease
- diabetes
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- thyroid disease
- cough that occurs with too much phlegm (mucus)
- difficulty in urination due to enlargement of the prostate gland
- high blood pressure
Stop use and ask a doctor if
- nervousness, dizziness, or sleeplessness occur
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- new symptoms occur
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
Directions
- do not take more than directed
- adults and children 12 years and over
- take 2 caplets every 4 hours
- swallow whole - do not crush, chew, or dissolve
- do not take more than 10 caplets in 24 hours
- children under 12 years: ask a doctor
Other information
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, crospovidone, D&C yellow #10 aluminum lake, flavor, magnesium stearate, maltodextrin, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, silicon dioxide, sodium starch glycolate, stearic acid, sucralose, talc, titanium dioxide
Principal Display Panel
EQUALINE®
NDC 41163-503-08
multi-symptom
daytime
severe cold
acetaminophen
(pain reliever/fever reducer)
dextromethorphan HBr (cough suppressant)
guaifenesin (expectorant)
phenylephrine HCl (nasal decongestant)
non-drowsy
relieves:
• fever/headache/sore throat
• cough
• chest congestion
• nasal congestion
24 caplets PSEUDOEPHEDRINE FREE actual size
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR
IF BLISTER UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERING
50844 REV0718C50308
DISTRIBUTED BY UNFI, PROVIDENCE, RI 02908 USA
100% Quality Guaranteed
855-423-2630
Equaline B-0019E-503A08CMSR REV0718C