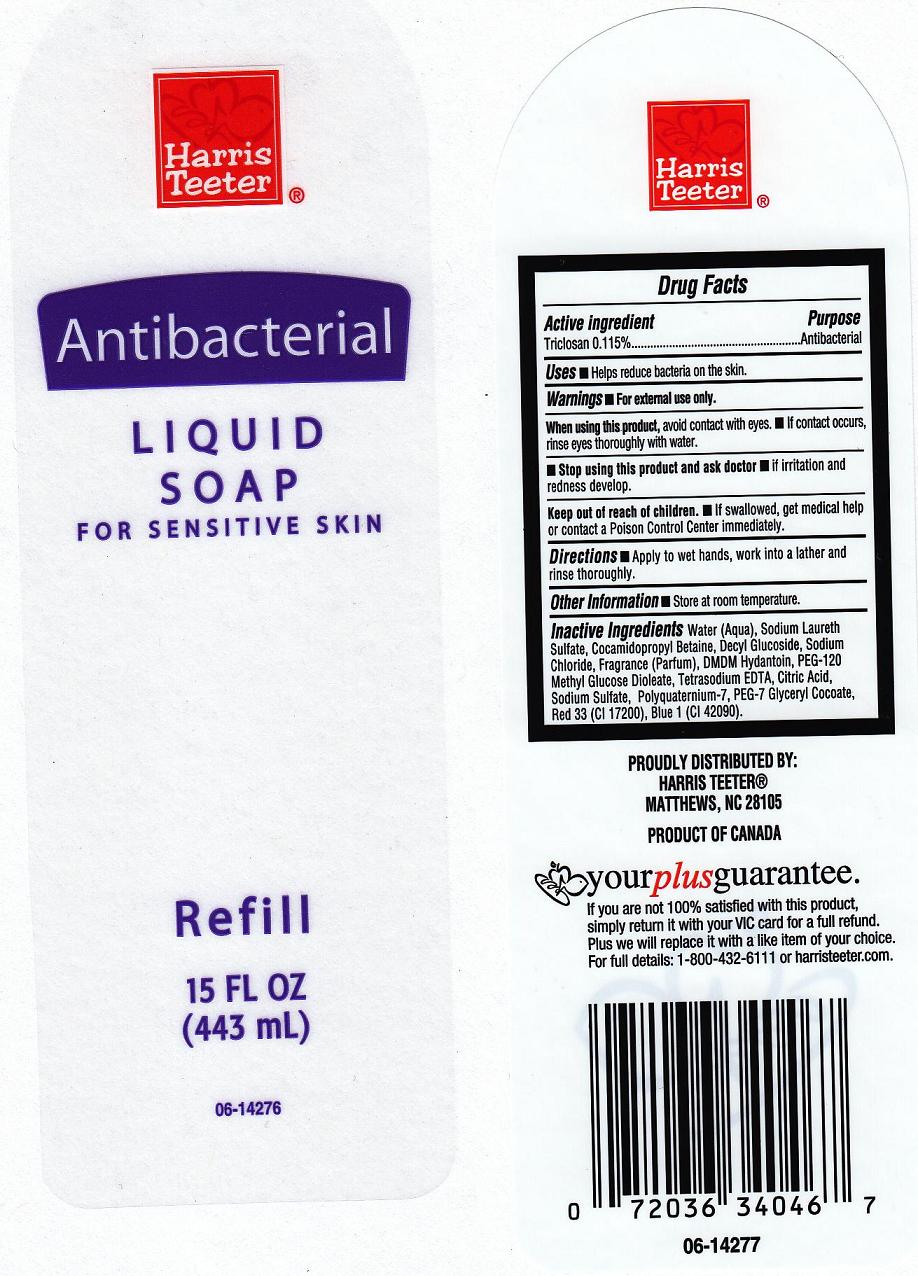
HARRIS TEETER ANTIBACTERIAL - triclosan soap
Harris Teeter
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Active Ingredient
Triclosan 0.115%
Warnings
For external use only.
When using this product
- Avoid contact with eyes. If contact occurs, rinse with water.
Stop using this product and ask doctor if
- irritation or redness develops and lasts.
Keep out of reach of children
In case of accidental ingestion, get medical help or contact a Poison Control Center immediately.
Directions
Apply onto wet hands, work into a lather, rinse thoroughly.
Uses
Helps reduce bacteria on the skin
Inactive Ingredients
Water, Sodium Laureth Sulfate,
Cocamide MEA, Cocamidopropyl Betaine, Decyl Glucoside, Sodium Chloride, Fragrance (Parfum), DMDM
Hydantoin, PEG-120 Methyl Glucose Dioleate, Citric Acid, Tetrasodium
EDTA, Polyquaternium-7, PEG- 7 Glyceryl Cocoate, Red 33 (CI 17200), Blue 1 (CI
42090).
Package Front and Back Labels
ht.jpg