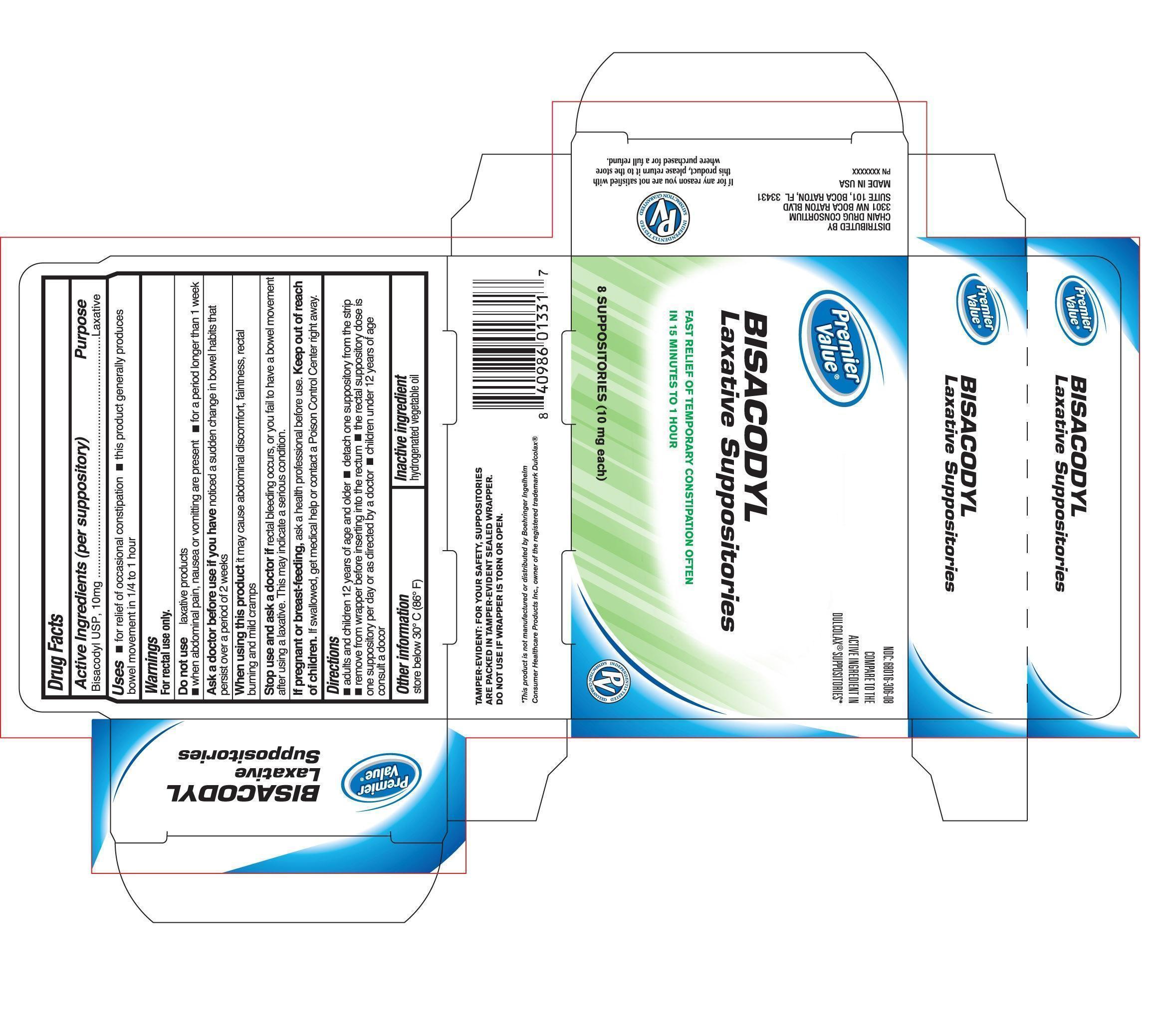
Active Ingredient (in each Suppository)
Active Ingredient (in each Suppository) Purpose
Bisacodyl USP, 10 mg.................................Laxative
Uses
Uses - For relief of occasional constipation -this product generally produces bowel movement in 1/4 hour to 1 hour
Do not use
Do not use laxative products
-when abdominal pain, nausea, or vomiting are present - for a period longer than 1 week
Ask a doctor before use if you have
Ask a doctor before use if you have noticed a sudden change in bowel movement
burning and mild cramps
When using this product
When using this product it may cause abdominal discomfort, faintness, rectal burning, and mild cramps
Stop use and ask a doctor
Stop use and ask a doctor if rectal bleeding occurs, or you fail to have bowel movement after using a laxative. This may indicate a serious condition.
Keep out of reach of children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
-adults and children 12 years of age and older - detach one suppository from the strip
-remove from wrapper before inserting into the rectum - the rectal suppository dose is one suppository per day or as directed by a doctor - children under 12 years of age consult a doctor.