Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Warnings
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Directions
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other information
- store between 20° to 25° C (68° to 77° F)
- protect from excessive moisture
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
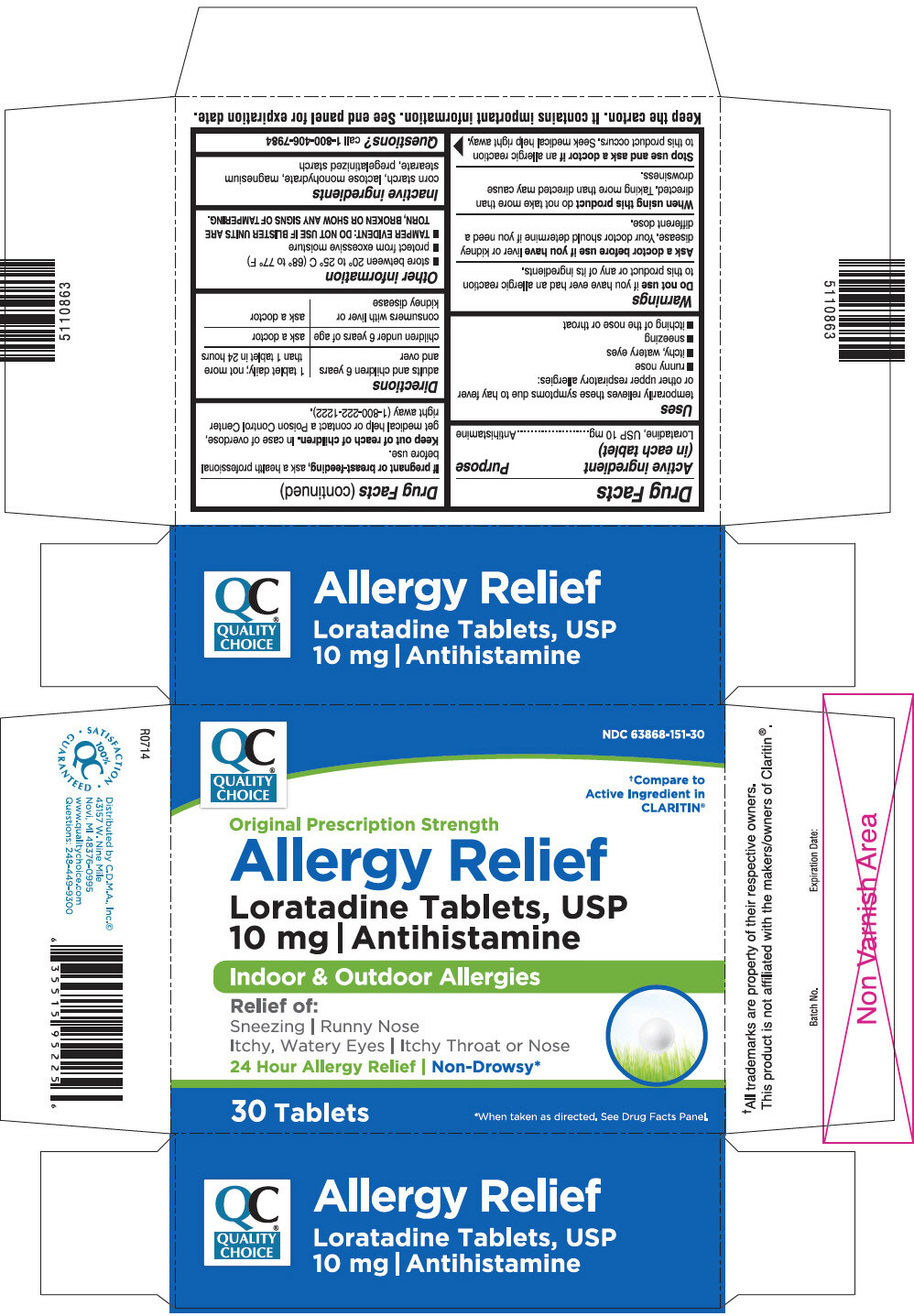
PRINCIPAL DISPLAY PANEL - 30 Tablet Blister Pack Carton
QUALITY
CHOICE
NDC 63868-151-30
†Compare to
Active Ingredient in
CLARITIN®
Original Prescription Strength
Allergy Relief
Loratadine Tablets, USP
10 mg | Antihistamine
Indoor & Outdoor Allergies
Relief of:
Sneezing | Runny Nose
Itchy, Watery Eyes | Itchy Throat or Nose
24 Hour Allergy Relief | Non-Drowsy*
30 Tablets
*When taken as directed. See Drug Facts Panel.