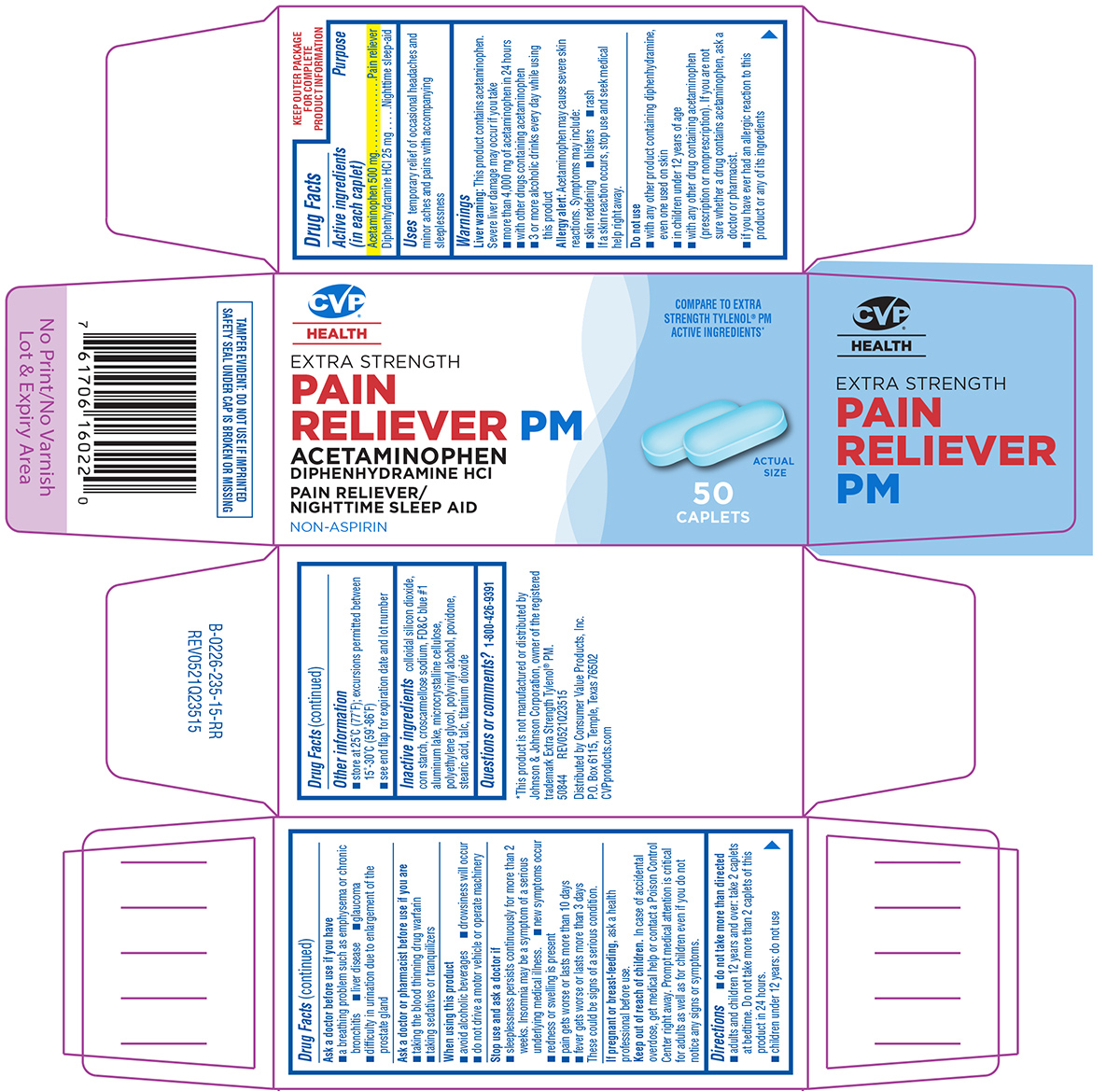
Uses
temporary relief of occasional headaches and minor aches and pains with accompanying sleeplessness
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other product containing diphenhydramine, even one used on skin
- in children under 12 years of age
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- liver disease
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- avoid alcoholic beverages
- drowsiness will occur
- do not drive a motor vehicle or operate machinery
Stop use and ask a doctor if
- sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
- new symptoms occur
- redness or swelling is present
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
These could be signs of a serious condition.
Directions
-
do not take more than directed
- adults and children 12 years and over: take 2 caplets at bedtime. Do not take more than 2 caplets of this product in 24 hours.
- children under 12 years: do not use
Other information
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive ingredients
colloidal silicon dioxide, corn starch, croscarmellose sodium, FD&C blue #1 aluminum lake, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, stearic acid, talc, titanium dioxide
Principal Display Panel
CVP®
HEALTH
COMPARE TO EXTRA
STRENGTH TYLENOL® PM
ACTIVE INGREDIENTS*
EXTRA STRENGTH
PAIN
RELIEVER PM
ACETAMINOPHEN
DIPHENHYDRAMINE HCl
PAIN RELIEVER/
NIGHTTIME SLEEP AID
NON-ASPIRIN
50
CAPLETS
ACTUAL SIZE
*This product is not manufactured or distributed by
Johnson & Johnson Corporation, owner of the registered
trademark Extra Strength Tylenol® PM.
50844 REV0521Q23515
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
Distributed by Consumer Value Products, Inc.
P.O. Box 6115. Temple, Texas 76502
CVPproducts.com
CVP 44-235