FULL PRESCRIBING INFORMATION
WARNING: FETAL TOXICITY
When pregnancy is detected, discontinue amlodipine and benazepril hydrochloride as soon as possible [see Warnings and Precautions (5.1)].
Drugs that act directly on the renin-angiotensin system (RAS) can cause injury and death to the developing fetus [see Warnings and Precautions (5.1)].
2 DOSAGE AND ADMINISTRATION
2.1 General Considerations
The recommended initial dose is amlodipine 2.5 mg and benazepril 10 mg orally once-daily.
Begin therapy with amlodipine and benazepril hydrochloride capsules only after a patient has either (a) failed to achieve the desired antihypertensive effect with amlodipine or benazepril monotherapy, or (b) demonstrated inability to achieve adequate antihypertensive effect with amlodipine therapy without developing edema.
The antihypertensive effect of amlodipine and benazepril hydrochloride capsules is largely attained within 2 weeks. If blood pressure remains uncontrolled, the dose may be titrated up to amlodipine 10 mg and benazepril 40 mg once-daily. The dosing should be individualized and adjusted according to the patient’s clinical response.
In clinical trials of amlodipine and benazepril combination therapy using amlodipine doses of 2.5 to 10 mg and benazepril doses of 10 to 40 mg, the antihypertensive effects increased with increasing dose of amlodipine in all patient groups, and the effects increased with increasing dose of benazepril in nonblack groups.
3 DOSAGE FORMS AND STRENGTHS
Amlodipine and benazepril hydrochloride capsules USP are available as follows:
2.5 mg/10 mg, 5 mg/10 mg, 5 mg/20 mg, 5 mg/40 mg, 10 mg/20 mg, and 10 mg/40 mg.
4 CONTRAINDICATIONS
- Do not coadminister aliskiren with angiotensin receptor blockers (ARBs), angiotensin-converting enzyme (ACE) inhibitors, including amlodipine and benazepril hydrochloride capsules in patients with diabetes.
- Amlodipine and benazepril hydrochloride capsules are contraindicated in patients with a history of angioedema, with or without previous ACE inhibitor treatment, or patients who are hypersensitive to benazepril, to any other ACE inhibitor, to amlodipine, or to any of the excipients of amlodipine and benazepril hydrochloride capsules.
- Amlodipine and benazepril hydrochloride capsules are contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer amlodipine and benazepril hydrochloride capsules within 36 hours of switching to or from a neprilysin inhibitor, e.g., sacubitril/valsartan [see Warnings and Precautions (5.1)].
5 WARNINGS AND PRECAUTIONS
5.1 Fetal Toxicity
Amlodipine Benazepril hydrochloride can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the renin-angiotensin system during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Resulting oligohydramnios can be associated with fetal lung hypoplasia and skeletal deformations. Potential neonatal adverse effects include skull hypoplasia, anuria, hypotension, renal failure, and death. When pregnancy is detected, discontinue amlodipine and benazepril hydrochloride as soon as possible [see Use in Specific Populations (8.1)].
5.2 Angioedema and Anaphylactoid Reactions
Head and Neck Angioedema: Angioedema of the face, extremities, lips, tongue, glottis, and larynx has been reported in patients treated with benazepril. This may occur at any time during treatment. Angioedema associated with edema of the larynx, tongue, or glottis can compromise the airway and be fatal. If laryngeal stridor or angioedema of the face, tongue, or glottis occurs, discontinue treatment with amlodipine and benazepril hydrochloride and treat immediately. When involvement of the tongue, glottis, or larynx appears likely to cause airway obstruction, appropriate therapy, e.g., administer subcutaneous epinephrine injection 1:1000 (0.3 to 0.5 mL), promptly [see Adverse Reactions (6)].
Patients with a history of angioedema may be at increased risk for angioedema while receiving amlodipine and benazepril hydrochloride. Black patients receiving ACE inhibitors have a higher incidence of angioedema compared to nonblacks.
Patients receiving coadministration of ACE inhibitor and mTOR (mammalian target of rapamycin) inhibitor (e.g., temsirolimus, sirolimus, everolimus) therapy or a neprilysin inhibitor may be at increased risk for angioedema [see Drug Interactions (7)].
Intestinal Angioedema: Intestinal angioedema has been reported in patients treated with ACE inhibitors. These patients presented with abdominal pain (with or without nausea or vomiting); in some cases there was no prior history of facial angioedema and C-1 esterase levels were normal. The angioedema was diagnosed by procedures including abdominal CT scan or ultrasound, or at surgery, and symptoms resolved after stopping the ACE inhibitor. Intestinal angioedema should be included in the differential diagnosis of patients on ACE inhibitors presenting with abdominal pain.
Anaphylactoid Reactions During Desensitization: Two patients undergoing desensitizing treatment with hymenoptera (wasp sting) venom while receiving ACE inhibitors sustained life-threatening anaphylactoid reactions.
Anaphylactoid Reactions During Membrane Exposure: Anaphylactoid reactions have been reported in patients dialyzed with high-flux membranes and treated concomitantly with an ACE inhibitor. Anaphylactoid reactions have also been reported in patients undergoing low-density lipoprotein apheresis with dextran sulfate absorption.
5.3 Increased Angina and/or Myocardial Infarction
Worsening angina and acute myocardial infarction can develop after starting or increasing the dose of amlodipine, particularly in patients with severe obstructive coronary artery disease.
5.4 Hypotension
Amlodipine and benazepril hydrochloride can cause symptomatic hypotension, sometimes complicated by oliguria, progressive azotemia, acute renal failure, or death. Symptomatic hypotension is most likely to occur in patients who have heart failure, severe aortic or mitral stenosis, obstructive hypertrophic cardiomyopathy or have been volume or salt depleted as a result of diuretic therapy, dietary salt restriction, dialysis, diarrhea, or vomiting. Correct volume and salt depletion before starting therapy with benazepril. If hypotension occurs, place the patient in the supine position and give physiological saline intravenously if needed. Continue treatment with benazepril once blood pressure and volume have returned to normal.
In patients with congestive heart failure, start amlodipine and benazepril hydrochloride therapy under close medical supervision; follow closely for the first 2 weeks of treatment and whenever the dose of the benazepril component is increased or a diuretic is added or its dose increased.
In patients undergoing surgery or during anesthesia with agents that produce hypotension, benazepril will block the angiotensin II formation that could otherwise occur secondary to compensatory renin release. Hypotension that occurs as a result of this mechanism can be corrected by volume expansion.
5.5 Impaired Renal Function
Monitor renal function periodically in patients treated with amlodipine and benazepril hydrochloride. Changes in renal function, including acute renal failure, can be caused by drugs that affect the RAS. Patients whose renal function may depend in part on the activity of the RAS (e.g., patients with renal artery stenosis, severe heart failure, post-myocardial infarction or volume depletion) or who are on Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) or ARBs may be at particular risk of developing acute renal failure on amlodipine and benazepril hydrochloride. Consider withholding or discontinuing therapy in patients who develop a clinically significant decrease in renal function on amlodipine and benazepril hydrochloride.
5.6 Hyperkalemia
Monitor serum potassium periodically in patients receiving amlodipine and benazepril hydrochloride. Drugs that affect the RAS can cause hyperkalemia. Risk factors for the development of hyperkalemia include renal insufficiency, diabetes mellitus, and the concomitant use of potassium-sparing diuretics, potassium supplements, and/or potassium-containing salt substitutes. In U.S. placebo-controlled trials of amlodipine and benazepril hydrochloride, hyperkalemia [serum potassium at least 0.5 mEq/L greater than the upper limit of normal (ULN)] not present at baseline occurred in approximately 1.5% of hypertensive patients receiving amlodipine and benazepril hydrochloride. Increases in serum potassium were generally reversible.
5.7 Hepatitis and Hepatic Failure
There have been rare reports of predominantly cholestatic hepatitis and isolated cases of acute liver failure, some of them fatal, in patients on ACE inhibitors. The mechanism is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevation of hepatic enzymes should discontinue the ACE inhibitor and be kept under medical surveillance.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The adverse reaction information from clinical trials does, however, provide a basis for identifying the adverse events that appear to be related to drug use and for approximating rates.
Amlodipine and benazepril hydrochloride has been evaluated for safety in over 2,991 patients with hypertension; over 500 of these patients were treated for at least 6 months, and over 400 were treated for more than 1 year.
In a pooled analysis of 5 placebo-controlled trials involving amlodipine and benazepril hydrochloride doses up to 5/20, the reported side effects were generally mild and transient, and there was no relationship between side effects and age, sex, race, or duration of therapy. Discontinuation of therapy due to side effects was required in approximately 4% of patients treated with amlodipine and benazepril hydrochloride and in 3% of patients treated with placebo.
The most common reasons for discontinuation of therapy with amlodipine and benazepril hydrochloride in these studies were cough and edema (including angioedema).
The peripheral edema associated with amlodipine use is dose-dependent. When benazepril is added to a regimen of amlodipine, the incidence of edema is substantially reduced.
The addition of benazepril to a regimen of amlodipine should not be expected to provide additional antihypertensive effect in African-Americans. However, all patient groups benefit from the reduction in amlodipine-induced edema.
The side effects considered possibly or probably related to study drug that occurred in these trials in more than 1% of patients treated with amlodipine and benazepril hydrochloride are shown in the table below. Cough was the only adverse event with at least possible relationship to treatment that was more common on amlodipine and benazepril hydrochloride (3.3%) than on placebo (0.2%).
| *Edema refers to all edema, such as dependent edema, angioedema, facial edema. | ||||
| Benazepril and Amlodipine
| Benazepril
| Amlodipine
| Placebo
|
|
| N = 760
| N = 554
| N = 475
| N = 408
|
|
| Cough
| 3.3 | 1.8 | 0.4 | 0.2 |
| Headache
| 2.2 | 3.8 | 2.9 | 5.6 |
| Dizziness
| 1.3 | 1.6 | 2.3 | 1.5 |
| Edema*
| 2.1 | 0.9 | 5.1 | 2.2 |
The incidence of edema was greater in patients treated with amlodipine monotherapy (5.1%) than in patients treated with amlodipine and benazepril hydrochloride (2.1%) or placebo (2.2%).
Other side effects considered possibly or probably related to study drug that occurred in U.S. placebo-controlled trials of patients treated with amlodipine and benazepril hydrochloride or in postmarketing experience were the following:
Body as a Whole: Asthenia and fatigue.
CNS: Insomnia, nervousness, anxiety, tremor, and decreased libido.
Dermatologic: Flushing, hot flashes, rash, skin nodule, and dermatitis.
Digestive: Dry mouth, nausea, abdominal pain, dyspepsia, and esophagitis.
Hematologic: Neutropenia.
Musculoskeletal: cramps, and muscle cramps.
Urogenital: Sexual problems, such as impotence, and polyuria.
Monotherapies of benazepril and amlodipine have been evaluated for safety in clinical trials in over 6,000 and 11,000 patients, respectively. The observed adverse reactions to the monotherapies in these trials were similar to those seen in trials of amlodipine and benazepril hydrochloride.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In postmarketing experience with benazepril, there have been rare reports of Stevens-Johnson syndrome, pancreatitis, hemolytic anemia, pemphigus, thrombocytopenia, paresthesia, dysgeusia, orthostatic symptoms and hypotension, angina pectoris and arrhythmia, pruritus, photosensitivity reaction, arthralgia, arthritis, myalgia, blood urea nitrogen (BUN) increase, serum creatinine increase, renal impairment, vision impairment, agranulocytosis, neutropenia.
Rare reports in association with use of amlodipine: gingival hyperplasia, tachycardia, jaundice, and hepatic enzyme elevations (mostly consistent with cholestasis severe enough to require hospitalization), leukocytopenia, allergic reaction, hyperglycemia, dysgeusia, hypoesthesia, paresthesia, syncope, peripheral neuropathy, hypertonia, visual impairment, diplopia, hypotension, vasculitis, rhinitis, gastritis, hyperhidrosis, pruritus, skin discoloration, urticaria, erythema multiform, muscle spasms, arthralgia, micturition disorder, nocturia, erectile dysfunction, malaise, weight decrease or gain.
Other potentially important adverse experiences attributed to other ACE inhibitors and calcium channel blockers include: eosinophilic pneumonitis (ACE inhibitors) and gynecomastia (CCBs).
7 DRUG INTERACTIONS
7.1 Drug/Drug Interactions
Amlodipine
Simvastatin: Coadministration of simvastatin with amlodipine increases the systemic exposure of simvastatin. Limit the dose of simvastatin in patients on amlodipine to 20 mg daily.
CYP3A4 Inhibitors: Coadministration with CYP3A inhibitors (moderate and strong) results in increased systemic exposure to amlodipine and may require dose reduction. Monitor for symptoms of hypotension and edema when amlodipine is coadministered with CYP3A4 inhibitors to determine the need for dose adjustment.
CYP3A4 Inducers: No information is available on the quantitative effects of CYP3A4 inducers on amlodipine. Blood pressure should be monitored when amlodipine is coadministered with CYP3A4 inducers (e.g. rifampicin, St. John’s Wort).
Benazepril
Potassium Supplements and Potassium-Sparing Diuretics: Benazepril can attenuate potassium loss caused by thiazide diuretics. Potassium-sparing diuretics (spironolactone, amiloride, triamterene, and others) or potassium supplements can increase the risk of hyperkalemia. If concomitant use of such agents is indicated, the patient’s serum potassium should be monitored frequently.
Lithium: Increased serum lithium levels and symptoms of lithium toxicity have been reported in patients receiving ACE inhibitors during therapy with lithium. When coadministering amlodipine and benazepril hydrochloride and lithium, frequent monitoring of serum lithium levels is recommended.
Gold: Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy.
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors): In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with ACE inhibitors, including benazepril, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving benazepril and NSAID therapy.
The antihypertensive effect of ACE inhibitors, including benazepril, may be attenuated by NSAIDs.
Antidiabetic Agents: In rare cases, diabetic patients receiving an ACE inhibitor (including benazepril) concomitantly with insulin or oral antidiabetics may develop hypoglycemia. Such patients should therefore be advised about the possibility of hypoglycemic reactions and should be monitored accordingly.
Mammalian Target of Rapamycin (mTOR) Inhibitors: The risk of angioedema may be increased in patients receiving coadministration of ACE inhibitors and mTOR inhibitors (e.g., temsirolimus, sirolimus, everolimus).
Dual Blockade of the Renin-Angiotensin System (RAS): Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Most patients receiving the combination of two RAS inhibitors do not obtain any additional benefit compared to monotherapy. In general, avoid combined use of RAS inhibitors. Closely monitor blood pressure, renal function and electrolytes in patients on amlodipine and benazepril hydrochloride and other agents that block the RAS.
Do not coadminister aliskiren with amlodipine and benazepril hydrochloride in patients with diabetes. Avoid use of aliskiren with amlodipine and benazepril hydrochloride in patients with renal impairment [glomerular filtration rate (GFR) < 60 mL/min].
Neprilysin Inhibitor: Patients taking concomitant neprilysin inhibitors may be at increased risk for angioedema [see Warnings and Precautions (5.1)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Amlodipine and benazepril hydrochloride can cause fetal harm when administered to a pregnant woman. Use of drugs that act on the RAS during the second and third trimesters of pregnancy reduces fetal renal function and increases fetal and neonatal morbidity and death. Most epidemiologic studies examining fetal abnormalities after exposure to antihypertensive use in the first trimester have not distinguished drugs affecting the RAS from other antihypertensive agents.
When pregnancy is detected, discontinue amlodipine and benazepril hydrochloride as soon as possible.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Hypertension in pregnancy increases the maternal risk for pre-eclampsia, gestational diabetes, premature delivery, and delivery complications (e.g., need for cesarean section, and post-partum hemorrhage). Hypertension increases the fetal risk for intrauterine growth restriction and intrauterine death. Pregnant women with hypertension should be carefully monitored and managed accordingly.
Fetal/Neonatal Adverse Reactions
Oligohydramnios in pregnant women who use drugs affecting the renin-angiotensin system in the second and third trimesters of pregnancy can result in the following: reduced fetal renal function leading to anuria and renal failure, fetal lung hypoplasia, skeletal deformations, including skull hypoplasia, hypotension and death.
Perform serial ultrasound examinations to assess the intra-amniotic environment. Fetal testing may be appropriate, based on the week of gestation. Patients and physicians should be aware, however, that oligohydramnios may not appear until after the fetus has sustained irreversible injury. If oligohydramnios is observed, consider alternative drug treatment. Closely observe neonates with histories of in utero exposure to amlodipine and benazepril hydrochloride for hypotension, oliguria, and hyperkalemia. In neonates with a history of in utero exposure to amlodipine and benazepril hydrochloride, if oliguria or hypotension occurs, support blood pressure and renal perfusion. Exchange transfusions or dialysis may be required as a means of reversing hypotension and replacing renal function.
Data
Animal Data
Benazepril and Amlodipine:
When rats received benazepril:amlodipine at doses ranging from 5:2.5 to 50:25 mg/kg/day, dystocia was observed at an increasing dose-related incidence at all doses tested. On a body surface area basis, the 2.5 mg/kg/day dose of amlodipine is twice the amlodipine dose delivered when the maximum recommended dose of amlodipine and benazepril hydrochloride is given to a 60 kg patient. Similarly, the 5 mg/kg/day dose of benazepril is approximately equivalent with the benazepril dose delivered when the maximum recommended dose of amlodipine and benazepril hydrochloride is given to a 60 kg patient. No teratogenic effects were seen when benazepril and amlodipine were administered in combination to pregnant rats or rabbits. Rats received doses of up to 50:25 mg (benazepril:amlodipine)/kg/day (12 times the MRHD on a body surface area basis, assuming a 60 kg patient). Rabbits received doses of up to 1.5:0.75 mg/kg/day (equivalent to the maximum recommended dose of amlodipine and benazepril hydrochloride given to a 60 kg patient).
8.2 Lactation
Risk Summary
Minimal amounts of unchanged benazepril and of benazeprilat are excreted into the breast milk of lactating women treated with benazepril, so that a newborn child ingesting nothing but breast milk would receive less than 0.1% of the maternal doses of benazepril and benazeprilat. Limited available data from a published clinical lactation study reports that amlodipine is present in human milk at an estimated median relative infant dose of 4.2%. No adverse effects of amlodipine on the breastfed infant have been observed. There is no available information on the effects of amlodipine or benazepril on milk production.
8.5 Geriatric Use
In geriatric patients, exposure to amlodipine is increased, thus consider lower initial doses of amlodipine and benazepril hydrochloride [see Clinical Pharmacology (12.3)].
Of the total number of patients who received amlodipine and benazepril hydrochloride in U.S. clinical studies of amlodipine and benazepril hydrochloride, over 19% were 65 years or older while about 2% were 75 years or older. Overall differences in effectiveness or safety were not observed between these patients and younger patients. Clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
Exposure to amlodipine is increased in patients with hepatic insufficiency, thus consider using lower doses of amlodipine and benazepril hydrochloride [see Clinical Pharmacology (12.3)].
8.7 Renal Impairment
In patients with severe renal impairment systemic exposure to benazepril is increased. The recommended dose of benazepril in this subgroup is 5 mg which is not an available strength with amlodipine and benazepril hydrochloride. Amlodipine and benazepril hydrochloride is not recommended in patients with severe renal impairment. No dose adjustment of amlodipine and benazepril hydrochloride is needed in patients with mild or moderate impairment of renal function [see Dosage and Administration (2.2), Warnings and Precautions (5.7) and Clinical Pharmacology (12.3)].
10 OVERDOSAGE
Only a few cases of human overdose with amlodipine have been reported. One patient was asymptomatic after a 250 mg ingestion; another, who combined 70 mg of amlodipine with an unknown large quantity of a benzodiazepine, developed refractory shock and died.
Human overdoses with any combination of amlodipine and benazepril have not been reported. In scattered reports of human overdoses with benazepril and other ACE inhibitors, there are no reports of death.
Treatment: Patients should be admitted to hospital and, generally, should be managed in an intensive care setting, with continuous monitoring of cardiac function, blood gases, and blood biochemistry. Emergency supportive measures such as artificial ventilation or cardiac pacing should be instituted if appropriate.
In the event of a potentially life-threatening oral overdose, use induction of vomiting or gastric lavage and/or activated charcoal to remove the drug from the gastrointestinal tract (only if presented within 1 hour after ingestion of amlodipine and benazepril hydrochloride).
Other clinical manifestations of overdose should be managed symptomatically based on modern methods of intensive care.
To obtain up-to-date information about the treatment of overdose, a good resource is your certified Regional Poison-Control Center. Telephone numbers of certified poison-control centers are listed in the Physicians’ Desk Reference (PDR). In managing overdose, consider the possibilities of multiple-drug overdoses, drug-drug interactions, and unusual drug kinetics in your patient.
The most likely effect of overdose with amlodipine and benazepril hydrochloride is vasodilation, with consequent hypotension and tachycardia. Simple repletion of central fluid volume (Trendelenburg positioning, infusion of crystalloids) may be sufficient therapy, but pressor agents (norepinephrine or high-dose dopamine) may be required. With abrupt return of peripheral vascular tone, overdoses of other dihydropyridine calcium channel blockers have sometimes progressed to pulmonary edema, and patients must be monitored for this complication.
Analyses of bodily fluids for concentrations of amlodipine, benazepril, or their metabolites are not widely available. Such analyses are, in any event, not known to be of value in therapy or prognosis.
No data are available to suggest physiologic maneuvers (e.g., maneuvers to change the pH of the urine) that might accelerate elimination of amlodipine, benazepril, or their metabolites. Benazeprilat is only slightly dialyzable; attempted clearance of amlodipine by hemodialysis or hemo-perfusion has not been reported, but amlodipine’s high protein binding makes it unlikely that these interventions will be of value.
Angiotensin II could presumably serve as a specific antagonist-antidote to benazepril, but angiotensin II is essentially unavailable outside of scattered research laboratories.
11 DESCRIPTION
Amlodipine and benazepril hydrochloride capsules USP are a combination of amlodipine besylate and benazepril hydrochloride.
Benazepril hydrochloride USP is a white to off-white, crystalline powder, soluble (greater than 100 mg/mL) in water, in ethanol, and in methanol. Benazepril hydrochloride’s chemical name is 3-[[1-(ethoxycarbonyl)-3-phenyl-(1S)-propyl]amino]-2,3,4,5-tetrahydro-2-oxo-1H-1-(3S)-benzazepine-1-acetic acid monohydrochloride; its structural formula is:
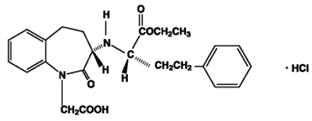
Its molecular formula is C24H28N2O5•HCl, and its molecular weight is 460.96.
Benazeprilat, the active metabolite of benazepril, is a nonsulfhydryl ACE inhibitor. Benazepril is converted to benazeprilat by hepatic cleavage of the ester group.
Amlodipine besylate USP is a white or almost white powder, slightly soluble in water and sparingly soluble in ethanol. Its chemical name is (R,S)3-ethyl-5-methyl-2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate benzenesulfonate; its structural formula is:
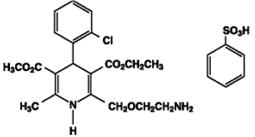
Its molecular formula is C20H25ClN2O5•C6H6O3S, and its molecular weight is 567.1.
Amlodipine besylate is the besylate salt of amlodipine, a dihydropyridine calcium channel blocker.
Amlodipine and benazepril hydrochloride is available as capsules containing amlodipine besylate USP (3.5 mg, 6.9 mg or 13.9 mg, equivalent to 2.5 mg, 5 mg or 10 mg of amlodipine respectively), with 10 mg, 20 mg, or 40 mg of benazepril hydrochloride USP providing for the following available combinations: 2.5 mg/10 mg, 5 mg/10 mg, 5 mg/20 mg, 5 mg/40 mg, 10 mg/20 mg, and 10 mg/40 mg.
The inactive ingredients of the capsules are colloidal silicon dioxide, crospovidone, gelatin, magnesium stearate, microcrystalline cellulose, povidone, sodium lauryl sulfate, and titanium dioxide. In addition, the hard gelatin capsule shells of 5 mg/10 mg contains iron oxide black, iron oxide red, and iron oxide yellow, 5 mg/20 mg contains iron oxide red, 5 mg/40 mg and 10 mg/40 mg contains FD&C Blue 1, FD&C Red 3, and 10 mg/20 mg contains D&C Red 28, FD&C Blue 1, FD&C Red 40, and FD&C Yellow 5. The capsules are printed with edible ink containing black iron oxide and shellac.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Benazepril
Benazepril and benazeprilat inhibit ACE in human subjects and in animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex.
Inhibition of ACE results in decreased plasma angiotensin II, which leads to decreased vasopressor activity and to decreased aldosterone secretion. The latter decrease may result in a small increase of serum potassium. Hypertensive patients treated with benazepril and amlodipine for up to 56 weeks had elevations of serum potassium up to 0.2 mEq/L [see Warnings and Precautions (5.6)].
Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity. In animal studies, benazepril had no inhibitory effect on the vasopressor response to angiotensin II and did not interfere with the hemodynamic effects of the autonomic neurotransmitters acetylcholine, epinephrine, and norepinephrine.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of amlodipine and benazepril hydrochloride remains to be elucidated.
While the mechanism through which benazepril lowers blood pressure is believed to be primarily suppression of the renin-angiotensin aldosterone system, benazepril has an antihypertensive effect even in patients with low-renin hypertension.
Amlodipine
Amlodipine is a dihydropyridine calcium antagonist (calcium ion antagonist or slow channel blocker) that inhibits the transmembrane influx of calcium ions into vascular smooth muscle and cardiac muscle. Experimental data suggest that amlodipine binds to both dihydropyridine and nondihydropyridine binding sites. The contractile processes of cardiac muscle and vascular smooth muscle are dependent upon the movement of extracellular calcium ions into these cells through specific ion channels. Amlodipine inhibits calcium ion influx across cell membranes selectively, with a greater effect on vascular smooth muscle cells than on cardiac muscle cells. Negative inotropic effects can be detected in vitro but such effects have not been seen in intact animals at therapeutic doses. Serum calcium concentration is not affected by amlodipine. Within the physiologic pH range, amlodipine is an ionized compound (pKa = 8.6), and its kinetic interaction with the calcium channel receptor is characterized by a gradual rate of association and dissociation with the receptor binding site, resulting in a gradual onset of effect.
Amlodipine is a peripheral arterial vasodilator that acts directly on vascular smooth muscle to cause a reduction in peripheral vascular resistance and reduction in blood pressure.
12.2 Pharmacodynamics
Benazepril
Single and multiple doses of 10 mg or more of benazepril cause inhibition of plasma ACE activity by at least 80% to 90% for at least 24 hours after dosing. For up to 4 hours after a 10 mg dose, pressor responses to exogenous angiotensin I were inhibited by 60% to 90%.
Administration of benazepril to patients with mild-to-moderate hypertension results in a reduction of both supine and standing blood pressure to about the same extent, with no compensatory tachycardia. Symptomatic postural hypotension is infrequent, although it can occur in patients who are salt and/or volume depleted [see Warnings and Precautions (5.4)].
The antihypertensive effects of benazepril were not appreciably different in patients receiving high- or low-sodium diets.
In normal human volunteers, single doses of benazepril caused an increase in renal blood flow but had no effect on glomerular filtration rate.
Amlodipine
Following administration of therapeutic doses to patients with hypertension, amlodipine produces vasodilation resulting in a reduction of supine and standing blood pressures. These decreases in blood pressure are not accompanied by a significant change in heart rate or plasma catecholamine levels with chronic dosing.
With chronic once-daily administration, antihypertensive effectiveness is maintained for at least 24 hours. Plasma concentrations correlate with effect in both young and elderly patients. The magnitude of reduction in blood pressure with amlodipine is also correlated with the height of pretreatment elevation; thus, individuals with moderate hypertension (diastolic pressure 105 to 114 mmHg) had about 50% greater response than patients with mild hypertension (diastolic pressure 90 to 104 mmHg). Normotensive subjects experienced no clinically significant change in blood pressure (+1/-2 mmHg).
In hypertensive patients with normal renal function, therapeutic doses of amlodipine resulted in a decrease in renal vascular resistance and an increase in glomerular filtration rate and effective renal plasma flow without change in filtration fraction or proteinuria.
As with other calcium channel blockers, hemodynamic measurements of cardiac function at rest and during exercise (or pacing) in patients with normal ventricular function treated with amlodipine have generally demonstrated a small increase in cardiac index without significant influence on dP/dt or on left ventricular end diastolic pressure or volume. In hemodynamic studies, amlodipine has not been associated with a negative inotropic effect when administered in the therapeutic dose range to intact animals and humans, even when coadministered with beta blockers to humans.
Amlodipine does not change sinoatrial (SA) nodal function or atrioventricular (AV) conduction in intact animals or humans. In clinical studies in which amlodipine was administered in combination with beta blockers to patients with either hypertension or angina, no adverse effects on electrocardiographic parameters were observed.
Amlodipine has demonstrated beneficial clinical effects in patients with chronic stable angina, vasospastic angina and angiographically documented coronary artery disease.
12.3 Pharmacokinetics
The rate and extent of absorption of benazepril and amlodipine from amlodipine and benazepril hydrochloride are the same as when administered as individual tablets. Absorption from the individual tablets is not influenced by the presence of food in the gastrointestinal tract; food effects on absorption from amlodipine and benazepril hydrochloride have not been studied.
Absorption: Following oral administration of amlodipine and benazepril hydrochloride, peak plasma concentrations of amlodipine are reached in 6 to 12 hours. Absolute bioavailability has been calculated as between 64% and 90%. Following oral administration of amlodipine and benazepril hydrochloride, the peak plasma concentrations of benazepril are reached in 0.5 to 2 hours. The cleavage of the ester group (primarily in the liver) converts benazepril to its active metabolite, benazeprilat, which reaches peak plasma concentrations in 1.5 to 4 hours. The extent of absorption of benazepril is at least 37%. Amlodipine and benazepril exhibit dose proportional pharmacokinetics between the therapeutic dose range of 2.5 and 10 mg and 10 and 20 mg, respectively.
Distribution: The apparent volume of distribution of amlodipine is about 21 L/kg. In vitro studies indicate that approximately 93% of circulating amlodipine is bound to plasma proteins in hypertensive patients. The apparent volume of distribution of benazeprilat is about 0.7 L/kg. Approximately 93% of circulating amlodipine is bound to plasma proteins, and the bound fraction of benazeprilat is slightly higher. On the basis of in vitro studies, benazeprilat’s degree of protein binding should be unaffected by age, by hepatic dysfunction, or—over the therapeutic concentration range—by concentration.
Metabolism: Amlodipine is extensively (approximately 90%) metabolized in the liver to inactive metabolites. Benazepril is extensively metabolized to form benazeprilat as the main metabolite, which occurs by enzymatic hydrolysis, mainly in the liver. Two minor metabolites are the acyl glucuronide conjugates of benazepril and benazeprilat.
Elimination: Amlodipine elimination from plasma is biphasic with a terminal elimination half-life of approximately 30 to 50 hours. Steady-state plasma levels are reached after once-daily dosing for 7 to 8 days. Ten percent of unchanged drug and 60% of amlodipine metabolites are excreted in urine. Effective elimination half-life of amlodipine is 2 days. Benazepril is eliminated mainly by metabolic clearance. Benazeprilat is eliminated via the kidneys and the bile; renal excretion is the main route in patients with normal renal function. In the urine, benazepril accounts for less than 1% and benazeprilat for about 20% of an oral dose. Elimination of benazeprilat is biphasic with an initial half-life of about 3 hours and a terminal half-life of about 22 hours. Benazeprilat’s effective elimination half-life is 10 to 11 hours, while that of amlodipine is about 2 days, so steady-state levels of the 2 components are achieved after about a week of once-daily dosing.
Specific Populations
Geriatric Patients: No specific clinical studies were performed to understand the impact of age on the pharmacokinetics of amlodipine and benazepril as fixed dose combination. As individual component amlodipine is extensively metabolized in the liver. In the elderly, clearance of amlodipine is decreased with resulting increases in peak plasma levels, elimination half-life and area-under-the-plasma-concentration curve [see Use in Specific Populations (8.5)].
Hepatic Impairment: Patients with hepatic insufficiency have decreased clearance of amlodipine with a resulting increase in AUC of approximately 40 to 60%. Pharmacokinetics of benazepril is not significantly influenced by hepatic impairment [see Use in Specific Populations (8.6)].
Renal Impairment: The disposition of benazepril and benazeprilat in patients with mild-to-moderate renal insufficiency (CrCl greater than 30 mL/min) is similar to that in patients with normal renal function. In patients with CrCl less than or equal to 30 mL/min, peak benazeprilat levels and the effective half-life increase, resulting in higher systemic exposures. Pharmacokinetics of amlodipine is not significantly influenced by renal impairment [see Dosage and Administration (2.2), Use in Specific Populations (8.7) and Warnings and Precautions (5.7)].
Drug Interactions
Amlodipine
In vitro data in human plasma indicate that amlodipine has no effect on the protein binding of digoxin, phenytoin, warfarin, and indomethacin.
Cimetidine: Coadministration of amlodipine with cimetidine did not alter the pharmacokinetics of amlodipine.
Grapefruit juice: Coadministration of 240 mL of grapefruit juice with a single oral dose of amlodipine 10 mg in 20 healthy volunteers had no significant effect on the pharmacokinetics of amlodipine.
Maalox® (antacid): Coadministration of the antacid Maalox with a single dose of amlodipine had no significant effect on the pharmacokinetics of amlodipine.
Sildenafil: A single 100 mg dose of sildenafil in subjects with essential hypertension had no effect on the pharmacokinetic parameters of amlodipine. When amlodipine and sildenafil were used in combination, each agent independently exerted its own blood pressure lowering effect.
Atorvastatin: Coadministration of multiple 10 mg doses of amlodipine with 80 mg of atorvastatin resulted in no significant change in the steady-state pharmacokinetic parameters of atorvastatin.
Digoxin: Coadministration of amlodipine with digoxin did not change serum digoxin levels or digoxin renal clearance in normal volunteers.
Ethanol (alcohol): Single and multiple 10 mg doses of amlodipine had no significant effect on the pharmacokinetics of ethanol.
Warfarin: Coadministration of amlodipine with warfarin did not change the warfarin prothrombin response time.
Simvastatin: Coadministration of multiple doses of 10 mg of amlodipine with 80 mg simvastatin resulted in a 77% increase in exposure to simvastatin compared to simvastatin alone.
CYP3A Inhibitors: Coadministration of a 180 mg daily dose of diltiazem with 5 mg amlodipine in elderly hypertensive patients resulted in a 60% increase in amlodipine systemic exposure. Erythromycin coadministration in healthy volunteers did not significantly change amlodipine systemic exposure. However, strong inhibitors of CYP3A4 (e.g., ketoconazole, itraconazole, ritonavir) may increase the plasma concentrations of amlodipine to a greater extent.
Benazepril
The pharmacokinetic properties of benazepril are not affected by hydrochlorothiazide, furosemide, chlorthalidone, digoxin, propranolol, atenolol, nifedipine, amlodipine, naproxen, acetylsalicylic acid, or cimetidine. Likewise the administration of benazepril does not substantially affect the pharmacokinetics of these medications.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity and mutagenicity studies have not been conducted with this combination. However, these studies have been conducted with amlodipine and benazepril alone (see below). No adverse effects on fertility occurred when the benazepril:amlodipine combination was given orally to rats of either sex at doses up to 15:7.5 mg (benazepril:amlodipine)/kg/day, prior to mating and throughout gestation.
Benazepril: No evidence of carcinogenicity was found when benazepril was administered to rats and mice for up to 2 years at doses of up to 150 mg/kg/day. When compared on the basis of body surface area, this dose is 18 and 9 times (rats and mice, respectively) the maximum recommended human dose (MRHD) (calculations assume a patient weight of 60 kg). No mutagenic activity was detected in the Ames test in bacteria, in an in vitro test for forward mutations in cultured mammalian cells, or in a nucleus anomaly test. At doses of 50 to 500 mg/kg/day (6 to 60 times the MRHD on a body surface area basis), benazepril had no adverse effect on the reproductive performance of male and female rats.
Amlodipine: Rats and mice treated with amlodipine maleate in the diet for up to 2 years, at concentrations calculated to provide daily dosage levels of 0.5, 1.25, and 2.5 mg amlodipine/kg/day, showed no evidence of a carcinogenic effect of the drug. For the mouse, the highest dose was, on a body surface area basis, similar to the MRHD of 10 mg amlodipine/day. For the rat, the highest dose was, on a body surface area basis, about two and a half times the MRHD (Calculations based on a 60 kg patient). Mutagenicity studies conducted with amlodipine maleate revealed no drug-related effects at either the gene or chromosome level. There was no effect on the fertility of rats treated orally with amlodipine maleate (males for 64 days and females for 14 days prior to mating) at doses of up to 10 mg amlodipine/kg/day (about 10 times the MRHD of 10 mg/day on a body surface area basis).
14 CLINICAL STUDIES
Over 950 patients received amlodipine and benazepril hydrochloride once-daily in 6 double-blind, placebo-controlled studies. The antihypertensive effect of a single dose persisted for 24 hours, with peak reductions achieved 2 to 8 hours after dosing.
Once-daily doses of benazepril and amlodipine using benazepril doses of 10 to 20 mg and amlodipine doses of 2.5 to 10 mg decreased seated pressure (systolic/diastolic) 24 hours after dosing by about 10 to 25/6 to 13 mmHg.
In 2 studies in patients not adequately controlled on either benazepril 40 mg alone (n = 329) or amlodipine 10 mg alone (n = 812) once-daily doses of amlodipine and benazepril hydrochloride 10 mg/40 mg further decreased seated blood pressure compared to the respective monotherapy alone.
Combination therapy was effective in blacks and nonblacks. Both components contributed to the antihypertensive efficacy in nonblacks, but virtually all of the antihypertensive effect in blacks could be attributed to the amlodipine component. Among nonblack patients in placebo-controlled trials comparing amlodipine and benazepril hydrochloride to the individual components, the blood pressure lowering effects of the combination were shown to be additive and in some cases synergistic.
During chronic therapy with amlodipine and benazepril hydrochloride, the maximum reduction in blood pressure with any given dose is generally achieved after 1 to 2 weeks. The antihypertensive effects of amlodipine and benazepril hydrochloride have continued during therapy for at least 1 year. Abrupt withdrawal of amlodipine and benazepril hydrochloride has not been associated with a rapid increase in blood pressure.
16 HOW SUPPLIED/STORAGE AND HANDLING
Amlodipine and benazepril hydrochloride is available as capsules containing amlodipine besylate USP (3.5 mg, 6.9 mg or 13.9 mg, equivalent to 2.5 mg, 5 mg or 10 mg of amlodipine respectively), with 10 mg, 20 mg, or 40 mg of benazepril hydrochloride USP providing for the following available combinations: 2.5 mg/10 mg, 5 mg/10 mg, 5 mg/20 mg, 5 mg/40 mg, 10 mg/20 mg, and 10 mg/40 mg. They are available as follows:
Amlodipine and Benazepril Hydrochloride Capsules USP, 2.5 mg/10 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of white cap and white body imprinted with ‘I’ on white cap and ‘96’ on white body with black edible ink.
Bottles of 100 NDC 65862-582-01
Bottles of 500 NDC 65862-582-05
Amlodipine and Benazepril Hydrochloride Capsules USP, 5 mg/10 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of light brown cap and light brown body imprinted with ‘I’ on light brown cap and ‘97’ on light brown body with black edible ink.
Bottles of 100 NDC 65862-583-01
Bottles of 500 NDC 65862-583-05
Amlodipine and Benazepril Hydrochloride Capsules USP, 5 mg/20 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of pink cap and pink body imprinted with ‘I’ on pink cap and ‘98’ on pink body with black edible ink.
Bottles of 100 NDC 65862-584-01
Bottles of 500 NDC 65862-584-05
Amlodipine and Benazepril Hydrochloride Capsules USP, 5 mg/40 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of light blue cap and light blue body imprinted with ‘J’ on light blue cap and ‘01’ on light blue body with black edible ink.
Bottles of 100 NDC 65862-585-01
Bottles of 500 NDC 65862-585-05
Amlodipine and Benazepril Hydrochloride Capsules USP, 10 mg/20 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of purple cap and purple body imprinted with ‘J’ on purple cap and ‘02’ on purple body with black edible ink.
Bottles of 100 NDC 65862-586-01
Bottles of 500 NDC 65862-586-05
Amlodipine and Benazepril Hydrochloride Capsules USP, 10 mg/40 mg are white to pale yellow colored powder filled in empty hard gelatin capsule shells, size “0” of dark blue cap and dark blue body imprinted with ‘J’ on dark blue cap and ‘03’ on dark blue body with black edible ink.
Bottles of 100 NDC 65862-587-01
Bottles of 500 NDC 65862-587-05
Storage: Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from moisture. Dispense in tight container (USP).
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Pregnancy: Advise female patients of childbearing age about the consequences of exposure to amlodipine and benazepril hydrochloride during pregnancy. Discuss treatment options with women planning to become pregnant. Ask patients to report pregnancies to their physicians as soon as possible [see Warnings and Precautions (5.1) and Use in Specific Populations (8.1)].
Symptomatic Hypotension: Advise patients that lightheadedness can occur, especially during the first days of therapy, and that it should be reported to their healthcare provider. Tell patients that if syncope occurs to discontinue amlodipine and benazepril hydrochloride capsules until the physician has been consulted. Caution all patients that inadequate fluid intake, excessive perspiration, diarrhea, or vomiting can lead to an excessive fall in blood pressure, with the same consequences of lightheadedness and possible syncope [see Warnings and Precautions (5.4)].
Hyperkalemia: Advise patients not to use salt substitutes without consulting their healthcare provider [see Drug Interactions (7)].
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 032, India
Revised: 11/2021
Patient Information
Amlodipine and Benazepril Hydrochloride Capsules USP
(am loe' di peen and ben az' e pril hye" droe klor' ide)
Read this Patient Information leaflet before you start taking amlodipine and benazepril hydrochloride capsules and each time you get a refill. There may be new information. This leaflet does not replace talking with your doctor. If you have any questions, ask your doctor or pharmacist.
What is the most important information I should know about amlodipine and benazepril hydrochloride capsules?
- Amlodipine and benazepril hydrochloride capsules can cause harm or death to an unborn baby.
- Talk to your doctor about other ways to lower your blood pressure if you plan to become pregnant.
- If you get pregnant while taking amlodipine and benazepril hydrochloride capsules, tell your doctor right away.
What are amlodipine and benazepril hydrochloride capsules?
Amlodipine and benazepril hydrochloride capsules contain 2 prescription medicines that work together to lower blood pressure: amlodipine besylate, a calcium channel blocker, and benazepril hydrochloride, an ACE inhibitor. Your doctor will prescribe amlodipine and benazepril hydrochloride capsules only after other medicines haven’t worked.
High Blood Pressure (hypertension). Blood pressure is the force of blood in your blood vessels. You have high blood pressure when the force is too much. Amlodipine and benazepril hydrochloride capsules can help your blood vessels relax so your blood pressure is lower.
Amlodipine and benazepril hydrochloride capsules have not been studied in children.
Who should not take amlodipine and benazepril hydrochloride capsules?
Don’t take amlodipine and benazepril hydrochloride capsules if you are allergic to any of the ingredients. There is a complete list at the end of this leaflet.
What should I tell my Doctor before taking amlodipine and benazepril hydrochloride capsules?
Tell your doctor about all your medical conditions, including if:
- you are pregnant or plan to become pregnant. See “What is the most important information I should know about amlodipine and benazepril hydrochloride capsules?”
- you are breastfeeding. Amlodipine and benazepril hydrochloride is present in human milk. It is not known whether amlodipine and benazepril hydrochloride affects your breastfed baby or milk production.
- you have a heart condition
- you have liver problems
- you have kidney problems
- you are about to have an operation (including dental surgery) or emergency treatment
- you are suffering from several episodes of vomiting or diarrhea
- you are treated for hyperkalemia (too much potassium in the blood)
Keep a list of your medicines with you, including vitamins and natural or herbal remedies, to show your doctor or pharmacist. Some of your other medicines and amlodipine and benazepril hydrochloride capsules could affect each other, causing serious side effects. Tell your doctor about all your medicines, especially:
- Simvastatin (a medicine used to control elevated cholesterol)
- medicines for high blood pressure or heart failure
- water pills, extra potassium or a salt substitute
- Lithium
- potassium-containing medicines, potassium supplements or salt substitutes containing potassium
- cyclosporine, an immunosuppressant medicine used in transplanted patients to reduce the risk of organ rejection
- indomethacin and other nonsteroidal anti-inflammatory drugs (NSAIDs), medicines used to relieve pain and inflammation
- insulin or oral antidiabetics, medicines that help a person with diabetes to control their level of glucose (sugar) in the blood
- gold for the treatment of rheumatoid arthritis
- probenecid, a medicine used to treat gout and hyperuricemia
- medicines used to prevent and treat fungal skin infections (e.g., ketoconazole, itraconazole)
- medicines used to treat AIDS or HIV infections (e.g., ritonavir, indinavir)
- medicines used to treat bacterial infections (e.g., clarithromycin)
- medicines used in organ transplant recipients or for treating some cancers (e.g. temsirolimus, sirolimus, everolimus)
Avoid alcohol until you have discussed the matter with your doctor. Alcohol may make blood pressure fall more and/or increase the possibility of dizziness or fainting.
How do I take amlodipine and benazepril hydrochloride capsules?
- Take amlodipine and benazepril hydrochloride capsules exactly as your doctor tells you.
- Take amlodipine and benazepril hydrochloride capsules at the same time each day, with or without food.
- If you miss a dose, take it as soon as you remember. If it is more than 12 hours, just take your next dose at the regular time.
- Your doctor may test for kidney problems or check your blood potassium level.
- If you take too much amlodipine and benazepril hydrochloride, call your doctor or Poison Control Center, or go to the emergency room.
- Tell all your doctors or dentist you are taking amlodipine and benazepril hydrochloride capsules if you:
- are going to have surgery
- are getting allergy shots for bee stings
- go for kidney dialysis
What are the possible side effects of amlodipine and benazepril hydrochloride capsules?
Amlodipine and benazepril hydrochloride capsules can cause serious side effects including:
- serious allergic reactions that can be life threatening.
Stop amlodipine and benazepril hydrochloride capsules and get emergency help right away if you get:
- swelling of your face, eyelids, lips, tongue or throat
- have trouble swallowing
- asthma (wheezing) or other breathing problems
These allergic reactions are rare but happen more times in people who are African-American.
- low blood pressure (hypotension). Low blood pressure is most likely to happen if you also take water pills, are on a low salt diet, get dialysis treatments, have heart problems or get sick with vomiting or diarrhea. Lie down if you feel faint or dizzy.
-
liver problems. Call your doctor if:
- you have nausea
- you feel more tired or weaker than usual
- you have itching
- your skin or eyes look yellow
- you have pain in your upper right stomach
- you have flu-like symptoms
- kidney problems. Some people will have changes on blood tests for kidney function and need a lower dose of amlodipine and benazepril hydrochloride capsules. Call your doctor if you get swelling in your feet, ankles, or hands or unexplained weight gain.
- more chest pain and heart attacks in people that already have severe heart problems. Get emergency help if you get worse chest pain or chest pain that does not go away.
The more common side effects of amlodipine and benazepril hydrochloride capsules are:
- dizziness, fainting on standing up
- cough (dry, nonproductive, mainly at night, continuing)
- swelling of the feet, ankles, and hands
If any of these affects you severely, tell your doctor.
These are not all the side effects of amlodipine and benazepril hydrochloride capsules. For a complete list, ask your doctor or pharmacist.
How do I store amlodipine and benazepril hydrochloride capsules?
- Store amlodipine and benazepril hydrochloride capsules at room temperature between 20° to 25°C (68° to 77°F).
- Keep amlodipine and benazepril hydrochloride capsules in a closed container in a dry place.
- Keep amlodipine and benazepril hydrochloride capsules and all medicines out of the reach of children.
General information about amlodipine and benazepril hydrochloride capsules
Doctors can also use medicine for a condition that is not in the patient information leaflet. Take amlodipine and benazepril hydrochloride capsules the way your doctor tells you. Do not share it with other people. They may harm them.
For more information, ask your doctor or pharmacist, or call 1-866-850-2876.
What are the ingredients in amlodipine and benazepril hydrochloride capsules?
Active ingredients: amlodipine besylate (the active ingredient found in Norvasc®), benazepril hydrochloride (Lotensin®)
Inactive ingredients: colloidal silicon dioxide, crospovidone, gelatin, magnesium stearate, microcrystalline cellulose, povidone, sodium lauryl sulfate, and titanium dioxide. In addition, the hard gelatin capsule shells of 5 mg/10 mg contains iron oxide black, iron oxide red, and iron oxide yellow, 5 mg/20 mg contains iron oxide red, 5 mg/40 mg and 10 mg/40 mg contains FD&C Blue 1, FD&C Red 3, and 10 mg/20 mg contains D&C Red 28, FD&C Blue 1, FD&C Red 40, and FD&C Yellow 5. The capsules are printed with edible ink containing black iron oxide and shellac.
The brands listed are trademarks of their respective owners and are not trademarks of Aurobindo Pharma Limited
Distributed by:
Aurobindo Pharma USA, Inc.
279 Princeton-Hightstown Road
East Windsor, NJ 08520
Manufactured by:
Aurobindo Pharma Limited
Hyderabad-500 032, India
Revised: 11/2021
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 2.5 mg/10 mg (100 Capsules Bottle)
NDC 65862-582-01
Rx only
Amlodipine and
Benazepril
Hydrochloride
Capsules USP
2.5 mg*/10 mg
AUROBINDO 100 Capsules

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/10 mg (100 Capsules Bottle)
NDC 65862-583-01
Rx only
Amlodipine and
Benazepril
Hydrochloride
Capsules USP
5 mg*/10 mg
AUROBINDO 100 Capsules

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/20 mg (100 Capsules Bottle)
NDC 65862-584-01
Rx only
Amlodipine and
Benazepril
Hydrochloride
Capsules USP
5 mg*/20 mg
AUROBINDO 100 Capsules

PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg/40 mg (100 Capsules Bottle)
NDC 65862-585-01
Rx only
Amlodipine and
Benazepril
Hydrochloride
Capsules USP
5 mg*/40 mg
AUROBINDO 100 Capsules


