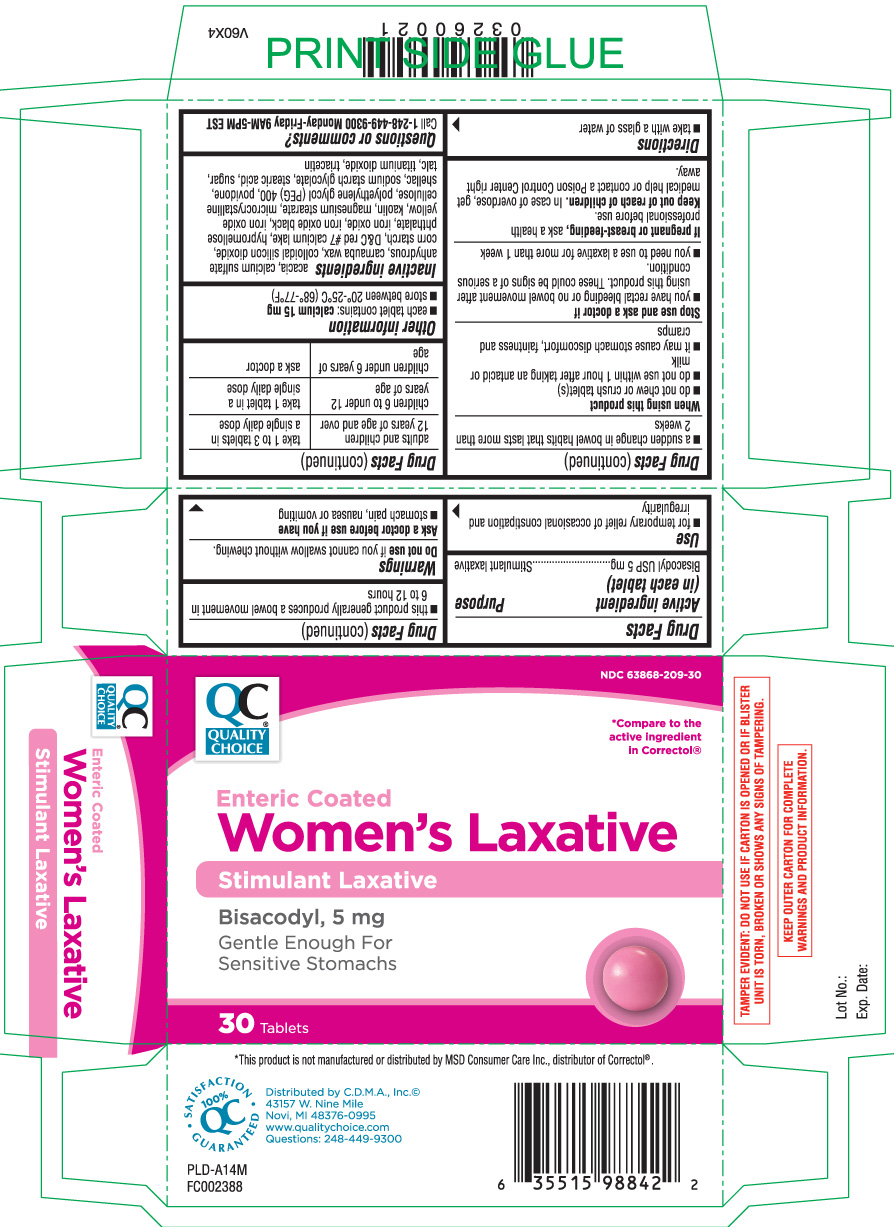
Use
- for temporary relief of occasional constipation and irregularity
- this product generally produces a bowel movement in 6 to 12 hours
Warnings
Ask a doctor before use if you have
- stomach pain, nausea or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
When using this product
- do not chew or crush tablet(s)
- do not use within 1 hour after taking an antacid or milk
- it may cause stomach discomfort, faintness and cramps
Directions
- take with a glass of water
| adults and children 12 years of age and over | take 1 to 3 tablets in a single daily dose |
| children 6 to under 12 years of age | take 1 tablet in a single daily dose |
| children under 6 years of age | ask a doctor |
Inactive ingredients
acacia, calcium sulfate anhydrous, carnauba wax, colloidal silicon dioxide, corn starch, D&C red # 7 calcium lake, hypromellose phthalate, iron oxide, iron oxide black, iron oxide yellow, kaolin, magnesium stearate, microcrystalline cellulose, polyethylene glycol (PEG) 400, povidone, shellac, sodium starch glycolate, stearic acid, sugar, talc, titanium dioxide, and triacetin
Principal Display Panel
Compare to the active ingredient in Correctol®*
Enteric Coated
Women's Laxative
Stimulant laxative
Bisacodyl, 5 mg
Gentle enough for sensitive stomachs
Tablets
*This product is not manufactured or distributed by MSD Consumer Care Inc., distributor of Correctol®.
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BORKEN OR SHOWS ANY SIGNS OF TAMPERING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATON.
Distributed by C.D.M.A., Inc.©
43157 W. Nine Mile
Novi, MI 48376-0995