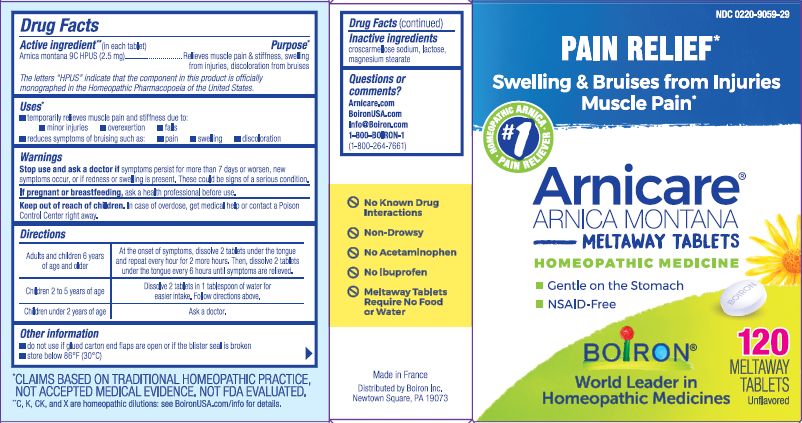
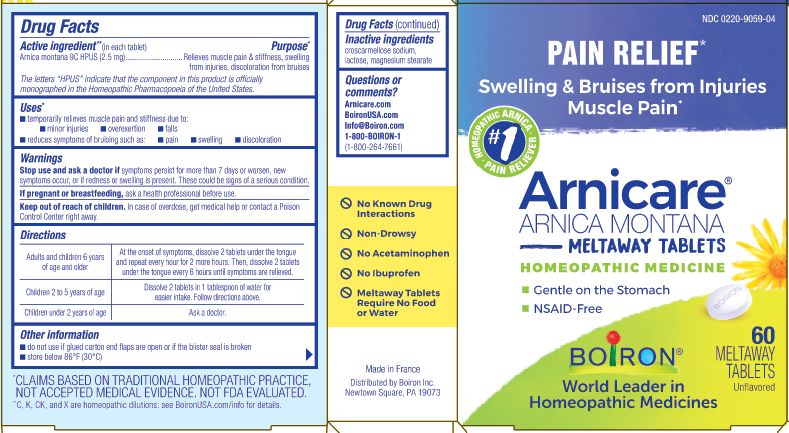
Active ingredient** (in each tablet)
Arnica Montana 9C HPUS (2.5 mg)
The letters "HPUS" indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
Purpose*
Arnica montana 9C HPUS (2.5 mg) ... Relieves muscle pain & stiffness, swelling from injuries, discoloration from bruises.
Uses*
temporarily relieves muscle pain and stiffness due to:
- minor injuries
- overexertion
- falls
- reduces symptoms of brusing such as:
- pain
- swelling
- discoloration
Stop use and ask a doctor if symptoms persist for more than 7 days or worsen, new symptoms occur, or if redness or swelling is present. These could be signs of a serious condition.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
Adults and children 6 years of age or older - At the onset of symptoms, dissolve 2 tablets under the tongue and repeat every hour for 2 more hours. Then, dissolve 2 tablets under the tongue every 6 hours until symptoms are relieved.
Children 2 to 5 years of age - Dissolve 2 tablets in 1 tablespoon for easier intake. Follow directions above.
Children under 2 years of age - Ask a doctor.
- do not use if glued carton end flaps are open or if the blister seal is broken
- store below 86°F (30°C)
No known drug interactions
Non-drowsy
No Acetaminophen
No Ibuprofen
Meltaway Tablets Require No Food or Water
Paid Relief*
Swelling & Bruise from Injuries Muscle Pain*
Gentle on the Stomach
NSAID-Free
60 Meltaway Tablets unflavored
120 Meltaway Tablets unflavored
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
**C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.