Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Warnings
Do not use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Ask a doctor before use if you have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Directions
- do not take more than directed
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 times in 24 hours
|
adults and children 12 | 1 to 2 tablets |
| children 6 to under 12 years | 1 tablet |
| children under 6 years | do not use |
Other information
- each tablet contains: calcium 30 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
- protect from moisture
- see end flap for expiration date and lot number
Inactive ingredients
corn starch, D&C red #27 aluminum lake, dibasic calcium phosphate dihydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, stearic acid, talc, titanium dioxide
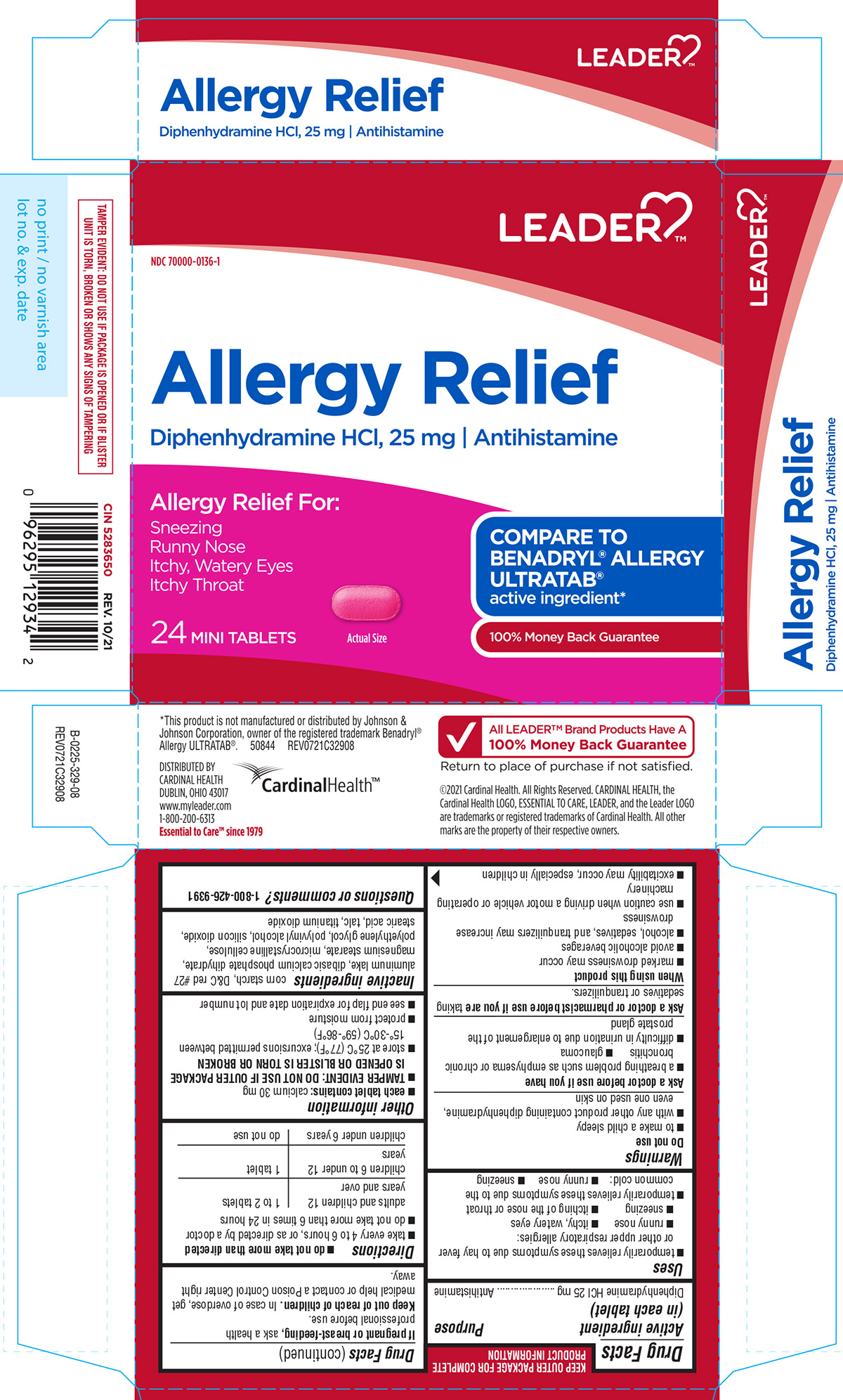
Principal Display Panel
LEADER™
NDC 70000-0136-1
Allergy Relief
Diphenhydramine HCl, 25 mg I Antihistamine
Allergy Relief For:
Sneezing
Runny Nose
Itchy, Watery Eyes
Itchy Throat
24 MINI TABLETS
Actual Size
COMPARE TO BENADRYL® ALLERGY ULTRATAB®
active ingredient*
100% Money Back Guarantee
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Johnson &
Johnson Corporation, owner of the registered trademark Benadryl®
Allergy ULTRATAB®.
50844 REV0721C32908
DISTRIBUTED BY
CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com
1-800-200-6313
Essential to Care™ since 1979
All LEADER™ Brand Products Have A
100% Money Back Guarantee
Return to place of purchase if not satisfied.
©2021 Cardinal Health. All Rights Reserved. CARDINAL HEALTH, the
Cardinal Health LOGO, ESSENTIAL TO CARE, LEADER, and the Leader
LOGO are trademarks or registered trademarks of Cardinal Health. All other
marks are the property of their respective owners.
Leader 44-329