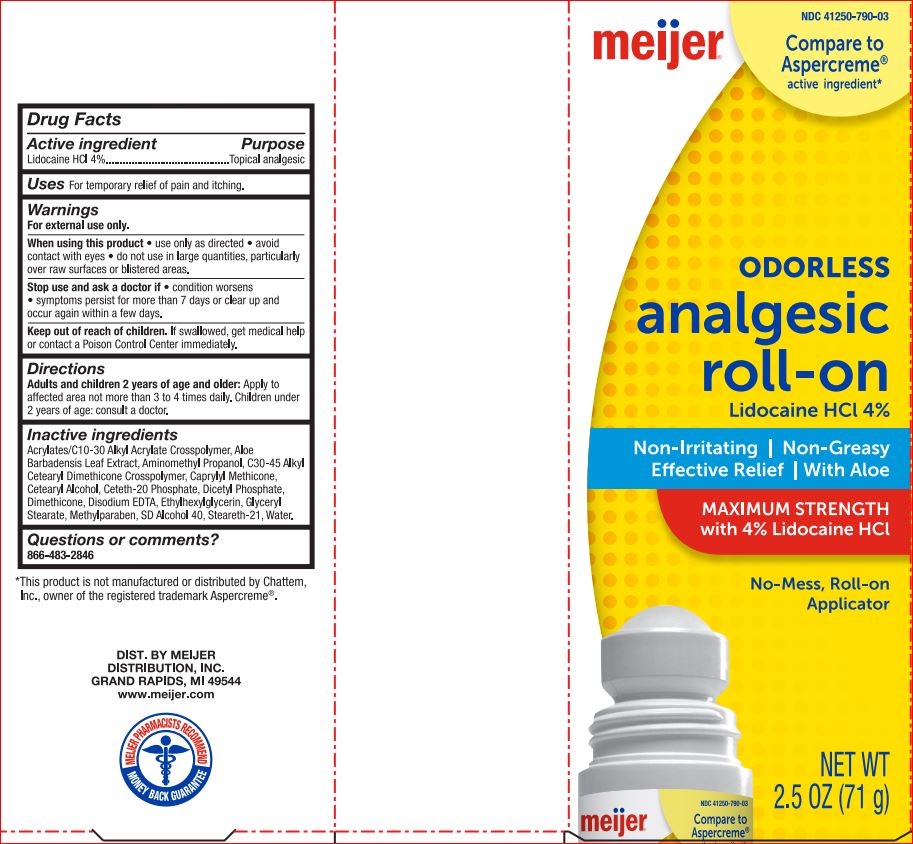
Active ingredient Purpose
Lidocaine HCl 4%..............................................Topical anesthetic
Warnings
For external use only.
When using this product • use only as directed • avoid contact with eyes • do not use in large quantities, particularly over raw surfaces or blistered areas.
Stop use and ask a doctor if • condition worsens • symptoms persist for more than 7 days or clear up and occur again within a few days
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center immediately
Directions
Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily. Children under 2 years of age: consult a doctor.
Inactive ingredients
Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Aloe Barbadensis
Leaf Juice, Aminomethyl Propanol, C30-45 Alkyl Cetearyl
Dimethicone Crosspolymer, Caprylyl Methicone, Cetearyl Alcohol,
Ceteth-20 Phosphate, Dicetyl Phosphate, Dimethicone, Disodium
EDTA, Ethylhexylglycerin, Glyceryl Stearate, Methylparaben, SD
Alcohol 40, Steareth-21, Water.