BACTROBAN- mupirocin calcium ointment
GlaxoSmithKline LLC
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use BACTROBAN Nasal Ointment safely and effectively. See full prescribing information for BACTROBAN Nasal Ointment.
BACTROBAN (mupirocin) nasal ointment Initial U.S. Approval: 1987 INDICATIONS AND USAGEBACTROBAN nasal ointment is an RNA synthetase inhibitor antibacterial indicated for the eradication of nasal colonization with methicillin-resistant Staphylococcus aureus (MRSA) in adult and pediatric patients (aged 12 years and older) and healthcare workers as part of a comprehensive infection control program to reduce the risk of infection among patients at high risk of MRSA infection during institutional outbreaks of infections with this microorganism. (1) Limitations of Use (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHSNasal ointment: 20 mg (2% w/w) of mupirocin in single-use 1-gram tubes. (3) CONTRAINDICATIONSKnown hypersensitivity to mupirocin or any of the excipients of BACTROBAN nasal ointment. (4) WARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most frequent adverse reactions (at least 1% in U.S. trials) were headache, rhinitis, respiratory disorders, pharyngitis, taste perversion, burning/stinging, cough, and pruritus. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact GlaxoSmithKline at 1-888-825-5249 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling. Revised: 2/2020 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
BACTROBAN nasal ointment is indicated for the eradication of nasal colonization with methicillin‑resistant Staphylococcus aureus (MRSA) in adult and pediatric patients (aged 12 years and older) and healthcare workers as part of a comprehensive infection control program to reduce the risk of infection among patients at high risk of MRSA infection during institutional outbreaks of infections with this microorganism.
Limitations of Use
- •
- There are insufficient data at this time to establish that this product is safe and effective as part of an intervention program to prevent autoinfection of high-risk patients from their own nasal colonization with Staphylococcus aureus (S. aureus).
- •
- There are insufficient data at this time to recommend use of BACTROBAN nasal ointment for general prophylaxis of any infection in any patient population.
2 DOSAGE AND ADMINISTRATION
- •
- For Intranasal Use Only.
- •
- Apply approximately one-half of the ointment from the single-use tube into 1 nostril and the other half into the other nostril twice daily (morning and evening) for 5 days.
- •
- After application, close the nostrils by pressing together and releasing the sides of the nose repetitively for approximately 1 minute. This will spread the ointment throughout the nares.
- •
- Do not apply BACTROBAN nasal ointment concurrently with any other intranasal products [see Clinical Pharmacology (12.3)].
- •
- The single-use 1-gram tube will deliver a total of approximately 0.5 grams of the ointment (approximately 0.25 grams per nostril).
- •
- Discard the tube after usage. Do not re-use.
3 DOSAGE FORMS AND STRENGTHS
BACTROBAN nasal ointment is a white to off-white ointment that contains 20 mg (2% w/w) of mupirocin in a soft white ointment base supplied in single-use 1‑gram tubes.
4 CONTRAINDICATIONS
BACTROBAN nasal ointment is contraindicated in patients with known hypersensitivity to mupirocin or any of the excipients of BACTROBAN nasal ointment.
5 WARNINGS AND PRECAUTIONS
5.1 Severe Allergic Reactions
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash have been reported in patients treated with formulations of BACTROBAN, including BACTROBAN nasal ointment [see Adverse Reactions (6.2)].
5.2 Eye Irritation
Avoid contact with the eyes. In case of accidental contact, rinse well with water. Application of BACTROBAN nasal ointment to the eye under testing conditions has caused severe symptoms such as burning and tearing. These symptoms resolved within days to weeks after discontinuation of the ointment.
5.3 Local Irritation
In the event of a sensitization or severe local irritation from BACTROBAN nasal ointment, usage should be discontinued.
5.4 Clostridium difficile-Associated Diarrhea
Clostridium difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over 2 months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in more detail in other sections of the labeling:
- •
- Severe Allergic Reactions [see Warnings and Precautions (5.1)]
- •
- Eye Irritation [see Warnings and Precautions (5.2)]
- •
- Local Irritation [see Warnings and Precautions (5.3)]
- •
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, 210 domestic (U.S.) and 2,130 foreign adult subjects received BACTROBAN nasal ointment. Less than 1% of domestic or foreign subjects in clinical trials were withdrawn due to adverse reactions.
The most frequently reported adverse reactions in foreign clinical trials were rhinitis (1%), taste perversion (0.8%), and pharyngitis (0.5%).
In domestic clinical trials, 17% (36 of 210) of adults treated with BACTROBAN nasal ointment reported adverse reactions. Table 1 shows the incidence of adverse reactions that were reported in at least 1% of adults enrolled in clinical trials conducted in the U.S.
Table 1. Adverse Reactions (≥1% Incidence) – Adults in U.S. Trials
|
Adverse Reactions |
% of Subjects Experiencing Reactions BACTROBAN Nasal Ointment (n = 210) |
|
Headache |
9% |
|
Rhinitis |
6% |
|
Respiratory disorder, including upper respiratory tract congestion |
5% |
|
Pharyngitis |
4% |
|
Taste perversion |
3% |
|
Burning/stinging |
2% |
|
Cough |
2% |
|
Pruritus |
1% |
The following adverse reactions were reported in less than 1% of adults enrolled in domestic clinical trials: blepharitis, diarrhea, dry mouth, ear pain, epistaxis, nausea, and rash.
6.2 Postmarketing Experience
In addition to adverse reactions reported from clinical trials, the following reactions have been identified during postmarketing use of BACTROBAN nasal ointment. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These reactions have been chosen for inclusion due to a combination of their seriousness, frequency of reporting, or potential causal relationship to BACTROBAN nasal ointment.
Immune System Disorders
Systemic allergic reactions, including anaphylaxis, urticaria, angioedema, and generalized rash [see Warnings and Precautions (5.1)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are insufficient human data to establish whether there is a drug-associated risk with BACTROBAN nasal ointment in pregnant women. Mupirocin absorption was negligible following repeated intranasal administration of BACTROBAN nasal ointment [see Clinical Pharmacology 12.3)], but the dose regimen used in this study did not mimic recommended clinical use. No developmental toxicity was observed in rats or rabbits treated with mupirocin subcutaneously during organogenesis at doses of 160 or 40 mg per kg per day, respectively (65 and 32 times the human intranasal dose based on calculations of dose divided by the entire body surface area).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. The estimated background risk in the U.S. general population of major birth defects is 2% to 4% and of miscarriage is 15% to 20% of clinically recognized pregnancies.
Data
Animal Data: Developmental toxicity studies have been performed with mupirocin administered subcutaneously to rats and rabbits at doses up to 160 mg per kg per day during organogenesis. This dose is 65 and 130 times, respectively, the human intranasal dose (approximately 20 mg mupirocin per day) based on calculations of dose divided by the entire body surface area. Maternal toxicity was observed (body weight loss/decreased body weight gain and reduced feeding) in both species with no evidence of developmental toxicity in rats. In rabbits, excessive maternal toxicity at the high dose precluded the evaluation of fetal outcomes. There was no developmental toxicity in rabbits at 40 mg per kg per day, 32 times the human intranasal dose based on calculations of dose divided by the entire body surface area.
Mupirocin administered subcutaneously to rats in a pre- and postnatal development study (dosed during late gestation through lactation) was associated with reduced offspring viability in the early postnatal period at a dose of 106.7 mg per kg, in the presence of injection site irritation and/or subcutaneous hemorrhaging. This dose is 37 times the human intranasal dose based on calculations of dose divided by the entire body surface area. The no-observed adverse effect level in this study was 44.2 mg per kg per day, which is 18 times the human intranasal dose.
8.2 Lactation
Risk Summary
It is not known whether mupirocin is present in human milk, has effects on the breastfed child, or has effects on milk production. However, breastfeeding is not expected to result in exposure of the child to the drug due to the negligible systemic absorption of mupirocin in humans following intranasal administration of BACTROBAN nasal ointment [see Clinical Pharmacology (12.3)]. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for BACTROBAN nasal ointment and any potential adverse effects on the breastfed child from BACTROBAN nasal ointment or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of BACTROBAN nasal ointment in children younger than 12 years have not been established [see Clinical Pharmacology (12.3)].
Pharmacokinetic data in neonates and premature infants indicate that, unlike in adults, significant systemic absorption occurred following intranasal administration of BACTROBAN nasal ointment in this population.
11 DESCRIPTION
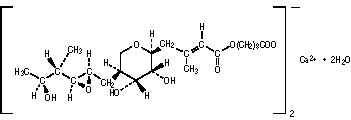
BACTROBAN (mupirocin) nasal ointment, 2% contains the dihydrate crystalline calcium hemi‑salt of the RNA synthetase inhibitor antibacterial, mupirocin. Chemically, it is (αE,2S,3R,4R,5S)-5-[(2S,3S,4S,5S)-2,3-epoxy-5-hydroxy-4-methylhexyl]tetrahydro-3,4-dihydroxy-β-methyl-2H-pyran-2-crotonic acid, ester with 9-hydroxynonanoic acid, calcium salt (2:1), dihydrate.
The molecular formula of mupirocin calcium is (C26H43O9)2Ca•2H2O, and the molecular weight is 1075.3. The molecular weight of mupirocin free acid is 500.6. The structural formula of mupirocin calcium is:
Each gram of BACTROBAN nasal ointment contains 20 mg (2% w/w) of mupirocin equivalent to 21.5 mg (2.15% w/w) of mupirocin calcium. The inactive ingredients are paraffin and a mixture of glycerin esters (SOFTISAN 649).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mupirocin is an RNA synthetase inhibitor antibacterial [see Microbiology (12.4)].
12.3 Pharmacokinetics
Absorption
Following single or repeated intranasal applications of 0.2 grams of BACTROBAN nasal ointment 3 times daily for 3 days to 5 healthy adult male subjects, no evidence of systemic absorption of mupirocin was demonstrated. The dosage regimen used in this trial was for pharmacokinetic characterization only; see Dosage and Administration (2) for proper clinical dosing information.
In this trial, the concentrations of mupirocin in urine and of monic acid in urine and serum were below the limit of determination of the assay for up to 72 hours after the applications. The lowest levels of determination of the assay used were 50 ng per mL of mupirocin in urine, 75 ng per mL of monic acid in urine, and 10 ng per mL of monic acid in serum. Based on the detectable limit of the urine assay for monic acid, one can extrapolate that a mean of 3.3% (range: 1.2% to 5.1%) of the applied dose could be systemically absorbed from the nasal mucosa of adults.
The effect of concurrent application of BACTROBAN nasal ointment with other intranasal products has not been studied [see Dosage and Administration (2)].
Elimination
In a trial conducted in 7 healthy adult male subjects, the elimination half‑life after intravenous administration of mupirocin was 20 to 40 minutes for mupirocin and 30 to 80 minutes for monic acid.
Metabolism: Following intravenous or oral administration, mupirocin is rapidly metabolized. The principal metabolite, monic acid, demonstrates no antibacterial activity.
Excretion: Monic acid is predominantly eliminated by renal excretion.
Specific Populations
Pediatric Patients: The pharmacokinetic properties of mupirocin following intranasal application of BACTROBAN nasal ointment have not been adequately characterized in neonates or other children younger than 12 years, and in addition, the safety and efficacy of the product in children younger than 12 years have not been established.
12.4 Microbiology
Mupirocin is an RNA synthetase inhibitor antibacterial produced by fermentation using the organism Pseudomonas fluorescens.
Mechanism of Action
Mupirocin inhibits bacterial protein synthesis by reversibly and specifically binding to bacterial isoleucyl‑transfer RNA (tRNA) synthetase.
Mupirocin is bactericidal at concentrations achieved topically by intranasal administration. Mupirocin is highly protein bound (greater than 97%) and the effect of nasal secretions on the minimum inhibitory concentrations (MICs) of intranasally applied mupirocin has not been determined.
Resistance
When mupirocin resistance occurs, it results from the production of a modified isoleucyl‑tRNA synthetase, or the acquisition of, by genetic transfer, a plasmid mediating a new isoleucyl-tRNA synthetase. High‑level plasmid‑mediated resistance (MIC ≥512 mcg/mL) has been reported in increasing numbers of isolates of S. aureus and with higher frequency in coagulase‑negative staphylococci. Mupirocin resistance occurs with greater frequency in methicillin‑resistant than methicillin‑susceptible staphylococci.
Cross Resistance
Due to its mode of action, mupirocin does not demonstrate cross resistance with other classes of antimicrobial agents.
Antimicrobial Activity
BACTROBAN nasal ointment has been shown to be active against most isolates of MRSA, both in vitro and in clinical trials [see Indications and Usage (1)].
Susceptibility Test Methods
High-level mupirocin resistance (≥512 mcg/mL) may be determined using standard disk diffusion or broth microdilution tests.1,2 The significance of these results, with regard to decolonization regimens, should be evaluated at each medical facility, in conjunction with laboratory, medical, and infection control staff.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long‑term studies in animals to evaluate carcinogenic potential of mupirocin calcium have not been conducted.
Results of the following studies performed with mupirocin calcium or mupirocin sodium in vitro and in vivo did not indicate a potential for genotoxicity: rat primary hepatocyte unscheduled DNA synthesis, sediment analysis for DNA strand breaks, Salmonella reversion test (Ames), Escherichia coli mutation assay, metaphase analysis of human lymphocytes, mouse lymphoma assay, and bone marrow micronuclei assay in mice.
In a fertility/reproductive performance study (with dosing through lactation), mupirocin administered subcutaneously to male and female rats at doses up to 100 mg per kg per day, which is 41 times the human intranasal dose (approximately 20 mg mupirocin per day) based on calculations of dose divided by the entire body surface area, did not result in impaired fertility or impaired reproductive performance attributable to mupirocin.
14 CLINICAL STUDIES
All adequate and well-controlled trials of this product were vehicle-controlled; therefore, no data from direct, head-to-head comparisons with other products are available. The safety and effectiveness of applications of this medication for greater than 5 days have not been established. There are no human clinical or pre-clinical animal data to support the use of this product in a chronic manner or in manners other than those described in this prescribing information.
In clinical trials, 210 domestic (US) and 2,130 foreign adult subjects received BACTROBAN nasal ointment. Greater than 90% of subjects in clinical trials had eradication of nasal colonization 2 to 4 days after therapy was completed. Approximately 30% recolonization was reported in 1 domestic trial within 4 weeks after completion of therapy. These eradication rates were clinically and statistically superior to those reported in subjects in the vehicle-treated arms of the adequate and well-controlled trials. Those treated with vehicle had eradication rates of 5% to 30% at 2 to 4 days post-therapy with 85% to 100% recolonization within 4 weeks.
15 REFERENCES
- 1.
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing; Twenty-sixth Informational Supplement. CLSI document M100-S26. Clinical and Laboratory Standards Institute, 950 West Valley Rd., Suite 2500, Wayne, PA 19087, USA, 2016.
- 2.
- Patel J, Gorwitz RJ, et al. Mupirocin Resistance. Clinical Infectious Diseases. 2009; 49(6); 935-41.
16 HOW SUPPLIED/STORAGE AND HANDLING
BACTROBAN nasal ointment is a white to off-white ointment that contains 20 mg (2% w/w) of mupirocin.
BACTROBAN nasal ointment, 2% is supplied in single-use 1‑gram tubes.
NDC 0029-1526-03 Single-use 1‑gram tube in Package of 10: NDC 0029-1526-11.
Store between 20°C and 25°C (68°F and 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). Do not refrigerate.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Advise the patient to administer BACTROBAN nasal ointment as follows:
- •
- Use BACTROBAN nasal ointment only as directed by the healthcare provider. Avoid contact of BACTROBAN nasal ointment with the eyes. If BACTROBAN nasal ointment gets in or near the eyes, rinse thoroughly with water.
- •
- Wash your hands before and after applying BACTROBAN nasal ointment.
- •
- Apply approximately one‑half of the BACTROBAN nasal ointment from the single‑use tube directly into 1 nostril and the other half into the other nostril.
- •
- Press the sides of the nose together and gently massage after application to spread the ointment throughout the inside of the nostrils.
- •
- Discard the tube after using. Do not re-use.
- •
- Report to the healthcare provider any signs of local adverse reactions. BACTROBAN nasal ointment should be stopped and the healthcare provider contacted if irritation, severe itching, or rash occurs.
- •
- Report to the healthcare provider or go to the nearest emergency room if severe allergic reactions, such as swelling of the lips, face, or tongue, or wheezing occur [see Warnings and Precautions (5.1)].
- •
- It is important that you take the full course of BACTROBAN nasal ointment. Do not stop early because the amount of bacteria in your nose may not be reduced.
BACTROBAN is owned by or licensed to the GSK group of companies. The other brand listed is a trademark owned by or licensed to its owner and is not owned by or licensed to the GSK group of companies. The maker of this brand is not affiliated with and does not endorse the GSK group of companies or its products.
GlaxoSmithKline
Research Triangle Park, NC 27709
©2020 GSK group of companies or its licensor.
BBN:8PI
PHARMACIST‑DETACH HERE AND GIVE INSTRUCTIONS TO PATIENT
|
PATIENT INFORMATION BACTROBAN (BACK-troh-ban) (mupirocin) nasal ointment |
|
What is BACTROBAN nasal ointment? BACTROBAN nasal ointment is a prescription medicine used to reduce the amount of methicillin-resistant Staphylococcus aureus (MRSA) bacteria in your nose. It is not known if BACTROBAN nasal ointment is safe and effective in children under 12 years of age. |
|
Who should not use BACTROBAN nasal ointment? Do not use BACTROBAN nasal ointment if:
|
|
What should I tell my healthcare provider before using BACTROBAN nasal ointment? Before using BACTROBAN nasal ointment, tell your healthcare provider about all of your medical conditions including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Do not mix BACTROBAN nasal ointment with other intranasal products. |
|
How should I use BACTROBAN nasal ointment?
|
|
What are the possible side effects of BACTROBAN nasal ointment? BACTROBAN nasal ointment may cause serious side effects, including:
○ hives ○ trouble breathing or wheezing ○ swelling of your face, lips, mouth, or tongue ○ dizziness, fast heartbeat or pounding in your chest ○ a rash over your whole body
These are not all the possible side effects of BACTROBAN nasal ointment. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store BACTROBAN nasal ointment?
|
|
General information about the safe and effective use of BACTROBAN nasal ointment. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use BACTROBAN nasal ointment for a condition for which it was not prescribed. Do not give BACTROBAN nasal ointment to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about BACTROBAN nasal ointment that is written for health professionals. |
|
What are the ingredients in BACTROBAN nasal ointment? Active Ingredient: mupirocin calcium Inactive Ingredients: paraffin and SOFTISAN 649 BACTROBAN is owned by or licensed to the GSK group of companies. The other brand listed is a trademark owned by or licensed to its owner and is not owned by or licensed to the GSK group of companies. The maker of this brand is not affiliated with and does not endorse the GSK group of companies or its products. ©2020 GSK group of companies or its licensor. GlaxoSmithKline, Research Triangle Park, NC 27709 BBN:4PIL For more information, call 1-888-825-5249. |
|
PRINCIPAL DISPLAY PANEL
NDC 0029-1526-11
BACTROBAN
(mupirocin nasal ointment, 2%
10 x 1.0 gram Single-Use Tubes
This shrink-wrapped unit represents one 5-day course of therapy.
Store at 20o - 25oC (68o – 77oF); excursions permitted 15o - 30oC (59o – 86oF).. Do not refrigerate.
Do not use if seal on tube nozzle is broken.
Usual Dosage: For intranasal use only. Apply one-half the contents of a tube in one nostril. Apply other half of tube contents in other nostril. See accompanying prescribing information.
GlaxoSmithKline
Research Triangle Park, NC 27709
- DFT-01678-4035086 Rev. 11/19
| BACTROBAN
mupirocin calcium ointment |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - GlaxoSmithKline LLC (167380711) |