DESCRIPTION
Nystatin is obtained from Streptomyces noursei. It is known to be a mixture, but the composition has not been completely elucidated. Nystatin A is closely related to amphotericin B. Each is a macro-cyclic lactone containing a ketal ring, an all-trans polyene system, and a mycosamine (3-amino-3-deoxyrhamose) moiety.
Its structural formula is:
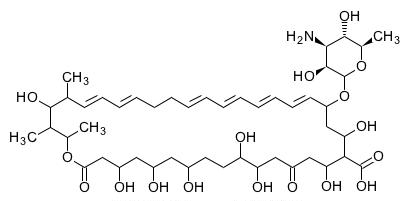
C47H75NO17 M.W. 926.13
Nystatin oral suspension, USP is a cherry-mint flavored, ready-to-use suspension. Each mL of nystatin oral suspension, USP contains 100,000 units of nystatin in a vehicle containing 50% w/v sucrose with the following inactive ingredients: alcohol (less than 1% v/v), carboxymethylcellulose sodium, cherry flavor, citric acid, glycerin, peppermint flavor, purified water, and sodium citrate. Citric acid and/or sodium hydroxide may be used to adjust pH (5.3-7.5).
PRESERVATIVES ADDED: Methylparaben 0.12%, Propylparaben 0.03%
CLINICAL PHARMACOLOGY
Nystatin acts by binding to sterols in the cell membrane of the fungus with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin is absorbed very sparingly following oral administration, with no detectable blood levels when given in the recommended doses.
INDICATIONS AND USAGE
Nystatin oral suspension, USP is indicated for the treatment of infections of the oral cavity caused by Candida albicans.
CONTRAINDICATIONS
Nystatin is contraindicated in patients with a history of hypersensitivity to nystatin or any of the suspension components.
PRECAUTIONS
General
Discontinue treatment with nystatin if sensitization or irritation is reported during use.
Nystatin is not effective in the treatment of systemic mycoses since it is not significantly absorbed from the gastrointestinal tract.
Information for Patients
Patient should be advised to retain nystatin in the mouth as long as possible and to continue its use for at least 2 days after symptoms have subsided.
There should be no interruption or discontinuation of the medication until the prescribed course of treatment is completed, even though symptomatic relief may occur within a few days.
If symptoms of local irritation develop, the physician should be notified immediately.
Laboratory Tests
If there is a lack of therapeutic response, appropriate microbiological studies (e .g., KOH smears and/or cultures) should be repeated to confirm the diagnosis of candidiasis and rule out other pathogens before instituting another course of therapy.
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate the carcinogenic potential of nystatin. In mice exposed to nystatin 50 mg/kg by injection, an increased incidence of chromosomal aberrations, consisting primarily of chromatid breaks, was observed in bone marrow cells. However, there have been no studies to determine the mutagenicity of orally-administered nystatin or its effects on fertility in males or females.
Pregnancy
Nonteratogenic Effects
In one rat reproductive study, nystatin was administered orally to pregnant rats in single doses of 100, 500, or 3000 mg/kg on the ninth day of gestation, or as multiple doses of 500 mg/kg/day on gestation days 1-20, 1-4, 7-10, 11-14, or 15-18. It was found that nystatin had a slight abortive effect when used during the whole period of pregnancy. No abnormalities were seen in surviving fetuses. Although no adverse effects or complications have been attributed to the use of intra-vaginal nystatin in neonates born to women treated during pregnancy, no similar studies evaluating complications of oral nystatin have been conducted.
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Hi-Tech Pharmacal Co., Inc. at 1-800-262-9010 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Gastrointestinal symptoms including diarrhea, gastrointestinal distress, nausea, vomiting and burning of the mouth have been reported. Hypersensitivity reactions including rash, pruritus, and anaphylactoid reaction have also been reported.
OVERDOSAGE
Oral doses of nystatin in excess of five million units daily have caused nausea and gastrointestinal upset.
DOSAGE AND ADMINISTRATION
Infants: 2 mL (200,000 units) four times daily (1 mL in each side of mouth).
Pediatric patients and adults: 4 to 6 mL (400,000 to 600,000 units) four times daily (one-half of dose in each side of mouth).
NOTE: Limited clinical studies in neonates, including premature and low-birth weight neonates, indicate that 1 mL (100,000 units) four times daily is effective.
Local treatment should be continued at least 48 hours after perioral symptoms have disappeared and/or cultures returned to normal. It is recommended that the drug be retained in the mouth as long as possible before swallowing.
HOW SUPPLIED
Nystatin Oral Suspension USP, 100,000 units per mL, is available as a yellow, milky, ready-to-use suspension with a cherry-mint flavor in the following sizes:
60 mL bottles (with child-resistant calibrated dropper)
SHAKE WELL BEFORE USING.
Storage: Store between 15°-30°C (59°-86°F). PROTECT FROM FREEZING.
Dispense in a tight, light resistant container when dispensing from the original container.
KEEP OUT OF REACH OF CHILDREN.
