Active ingredient
(in each 118 mL delivered dose)
Dibasic sodium phosphate 7 g
Monobasic sodium phosphate 19 g
Warnings
For rectal use only
Ask a doctor before use if you have
- •
- a sodium-restricted diet
- •
- abdominal pain, nausea or vomiting
- •
- kidney disease, heart problems, or are dehydrated
- •
- noticed a sudden change in bowel habits that lasts over 2 weeks
Ask a doctor or pharmacist before use if you are
taking any other drug. Take this product two or more hours before or after other drugs. Laxatives may affect how other drugs work.
When using this product
- •
- do not use more than directed. Serious side effects may occur from excess dosage.
- •
- do not use for more than 3 days
Directions
Do not use more unless directed by a doctor. See Warnings.
|
adults & children 12 years and over |
one bottle once daily |
|
children 2 to under 12 years |
1/2 bottle once daily |
|
children under 2 years |
do not use |
CAUTION:
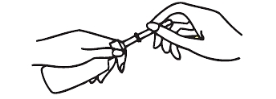
Remove green protective shield before inserting. Hold bottle upright, grasping bottle cap with fingers. Grasp protective shield with other hand and pull gently to remove.
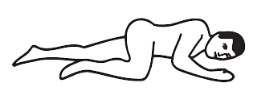
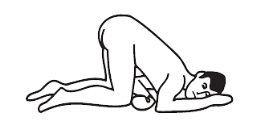
Positioning:
Left-side position: lie on left side with knee bent and arms at rest.
Knee-chest position: kneel, then lower head and chest forward until left side of face is resting on surface. Position arms comfortably.
Administering enema:
- •
- with steady pressure, gently insert enema with tip pointing toward navel.
- •
- squeeze bottle until recommended dose is expelled (it is not necessary to empty unit completely. Bottle contains more liquid than needed for effective use. A small amount of liquid will remain in bottle after squeezing).
- •
- remove tip from rectum
- •
- stop using if tip is hard to insert. Forcing the tip into the rectum can cause injury (especially if you have hemorrhoids). If enema tip causes rectal bleeding or pain, get immediate medical care.
- •
- maintain position until urge to evacuate is strong (usually 1 to 5 minutes)
- •
- if no urge is felt after 5 minutes of using, try to empty bowel. Call a doctor promptly if no liquid comes out of the rectum after 30 minutes because dehydration could occur.
Other Information
- •
- each 118 mL contains: sodium 4.3 g
- •
- store at room temperature 20-25 deg C (68-77 deg F)
- •
- this product generally produces bowel movement in 1 to 5 minutes
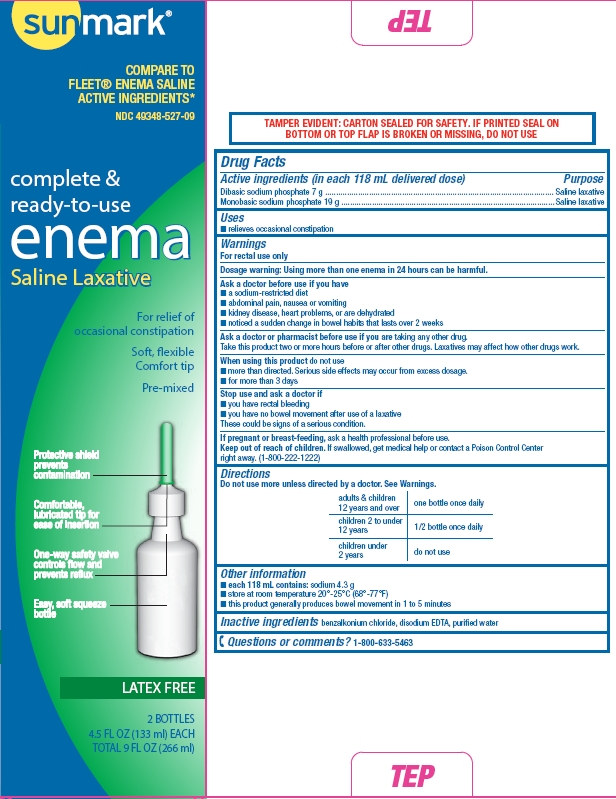
Side and Principal Display Panel - box
Sunmark
Compare to Fleet Enema Saline Active Ingredients
NDC 49348-005-35
Complete & Ready-to-Use
ENEMA
Saline Laxative
For relief of occasional constipation
Soft, flexible
Comfort tip
Pre-mixed
Twin Pack
Latex Free
2 bottles
4.5 FL OZ (133 ml) each
Total 9 FL OZ (266 ml)