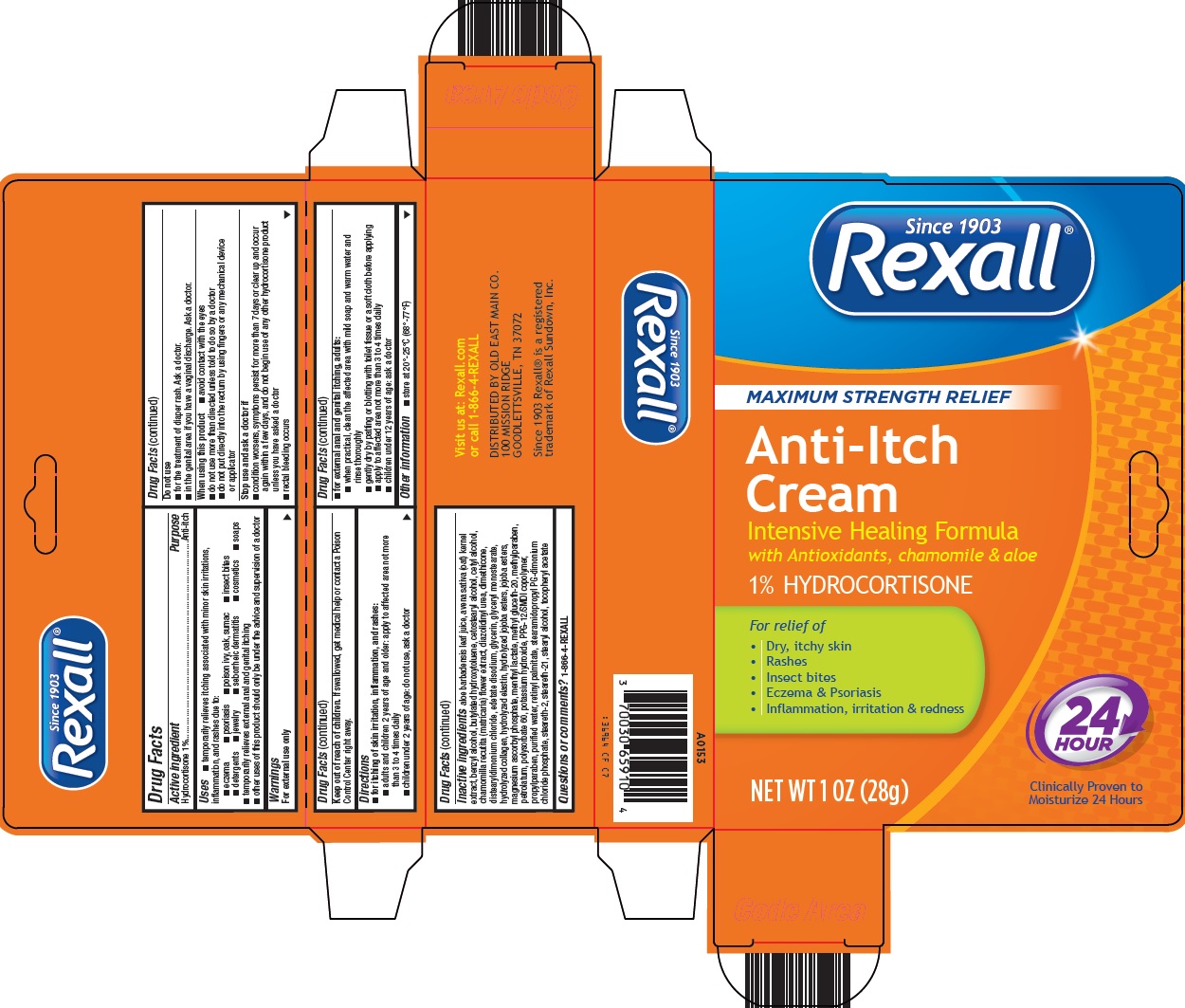
Uses
- •
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
- •
- eczema
- •
- psoriasis
- •
- poison ivy, oak, sumac
- •
- insect bites
- •
- detergents
- •
- jewelry
- •
- cosmetics
- •
- soaps
- •
- seborrheic dermatitis
- •
- temporarily relieves external anal and genital itching
- •
- other uses of this product should only be under the advice and supervision of a doctor
Warnings
For external use only
Do not use
- •
- for the treatment of diaper rash. Ask a doctor.
- •
- in the genital area if you have a vaginal discharge. Ask a doctor.
When using this product
- •
- avoid contact with the eyes
- •
- do not use more than directed unless told to do so by a doctor
- •
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Directions
- •
- for itching of skin irritation, inflammation, and rashes:
- •
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- •
- children under 2 years of age: do not use, ask a doctor
- •
- for external anal and genital itching, adults:
- •
- when practical, clean the affected area with mild soap and warm water and rinse thoroughly
- •
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
- •
- apply to affected area not more than 3 to 4 times daily
- •
- children under 12 years of age: ask a doctor
Inactive ingredients
aloe barbadensis leaf juice, avena sativa (oat) kernel extract, benzyl alcohol, butylated hydroxytoluene, cetostearyl alcohol, cetyl alcohol, chamomilla recutita (matricaria) flower extract, diazolidinyl urea, dimethicone, distearyldimonium chloride, edetate disodium, glycerin, glyceryl monostearate, hydrolyzed collagen, hydrolyzed elastin, hydrolyzed jojoba esters, jojoba esters, magnesium ascorbyl phosphate, menthyl lactate, methyl gluceth-20, methylparaben, petrolatum, polysorbate 60, potassium hydroxide, PPG-12/SMDI copolymer, propylparaben, purified water, retinyl palmitate, stearamidopropyl PG-dimonium chloride phosphate, steareth-2, steareth-21, stearyl alcohol, tocopheryl acetate
Principal Display Panel
Since 1903
Rexall®
MAXIMUM STRENGTH RELIEF
Anti-Itch Cream
Intensive Healing Formula with Antioxidants, chamomile & aloe
1% HYDROCORTISONE
For relief of
• Dry, itchy skin
• Rashes
• Insect bites
• Eczema & Psoriasis
• Inflammation, irritation & redness
24 HOUR
NET WT 1 OZ (28g)
Clinically Proven to Moisturize 24 Hours