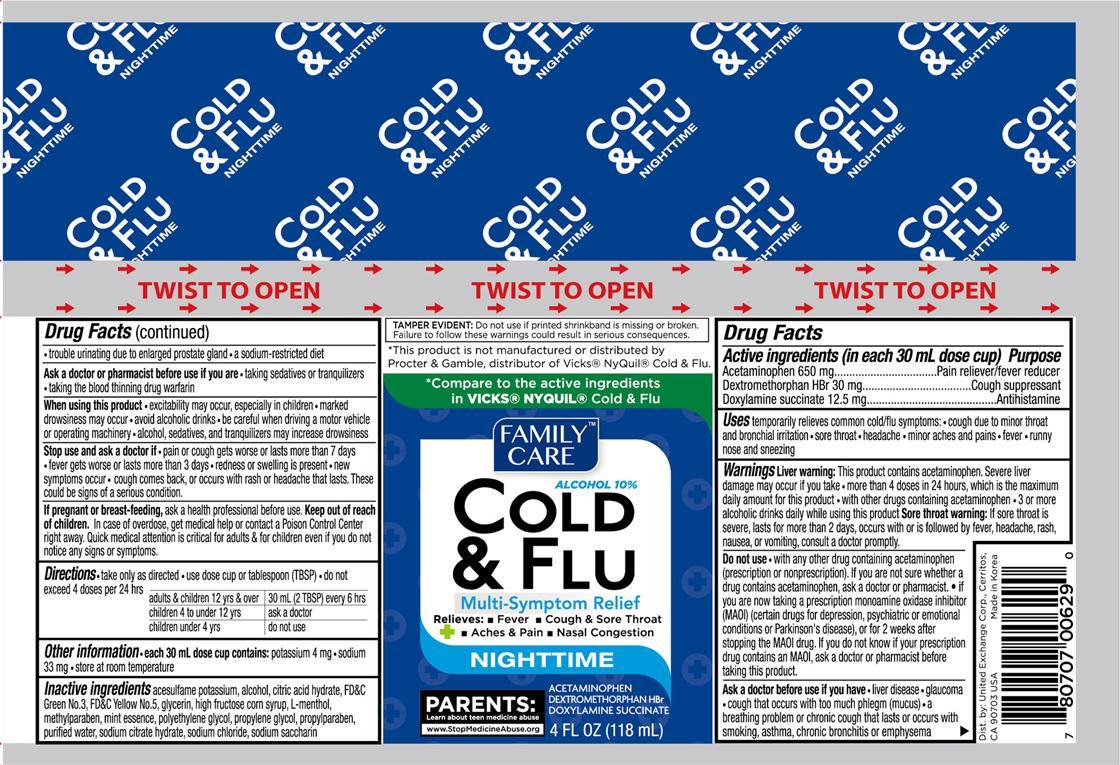
Active ingredients (in each 30 mL dose cup) Purpose
Acetaminophen 650 mg....................................................Pain reliever/fever reducer
Dextromethorphan HBr 30 mg.....................................................Cough suppressant
Doxylamine succinate 12.5 mg............................................................Antihistamine
Uses
temporarily relieves common cold/flu symptoms:
- cough due to minor throat and bronchial irritation
- sore throat
- headache
- minor aches and pains
- fever
- running nose and sneezing
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4 doses in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks daily while using this product
Sore throat warning: if sore throat is severe, lasts for more than 2 days, occurs with or is followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- liver disease
- glaucoma
- cough that occurs with too much phlegm (mucus)
- a breathing problem or chronic cough that lasts or occurs with smoking, asthma, chronic bronchitis or emphysema
- trouble urinating due to enlarged prostate gland
- a sodium-restricted diet
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers
- taking the blood thinning drug warfarin
When using this product
- excitability may occur, especially in children
- marked drowsiness may occur
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
- alcohol, sedatives, and tranquilizers may increase drowsiness
Stop use and ask a doctor if
- pain or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occurs
- cough comes back, or occurs with rash or headache that lasts. These could be signs of a serious condition.
Keep our of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults & for children even if you do not notice any signs or symptoms.
Directions
- take only as directed
- use dose cup or tablespoon (TBSP)
- do not exceed 4 doses per 24 hrs
adults & children 12 yrs & over: 30 mL (2 TBSP) every 6 hrs
children 4 to under 12 yrs: ask a doctor
children under 4 yrs: do not use
Other information
- each 30 mL dose cup contains: potassium 4 mg
- sodium 33 mg
- store at room temperature
Inactive ingredietns
acesulfame potassium, alcohol, citric acid hydrate, FD&C Green No. 3, FD&C No. 5, glycerin, high fructose corn syrup, L-menthol, methylparaben, mint essence, polyethylene glycol, propylene glycol, propylparaben, purified water, sodium citrate hydrate, sodium chloride, sodium saccharin