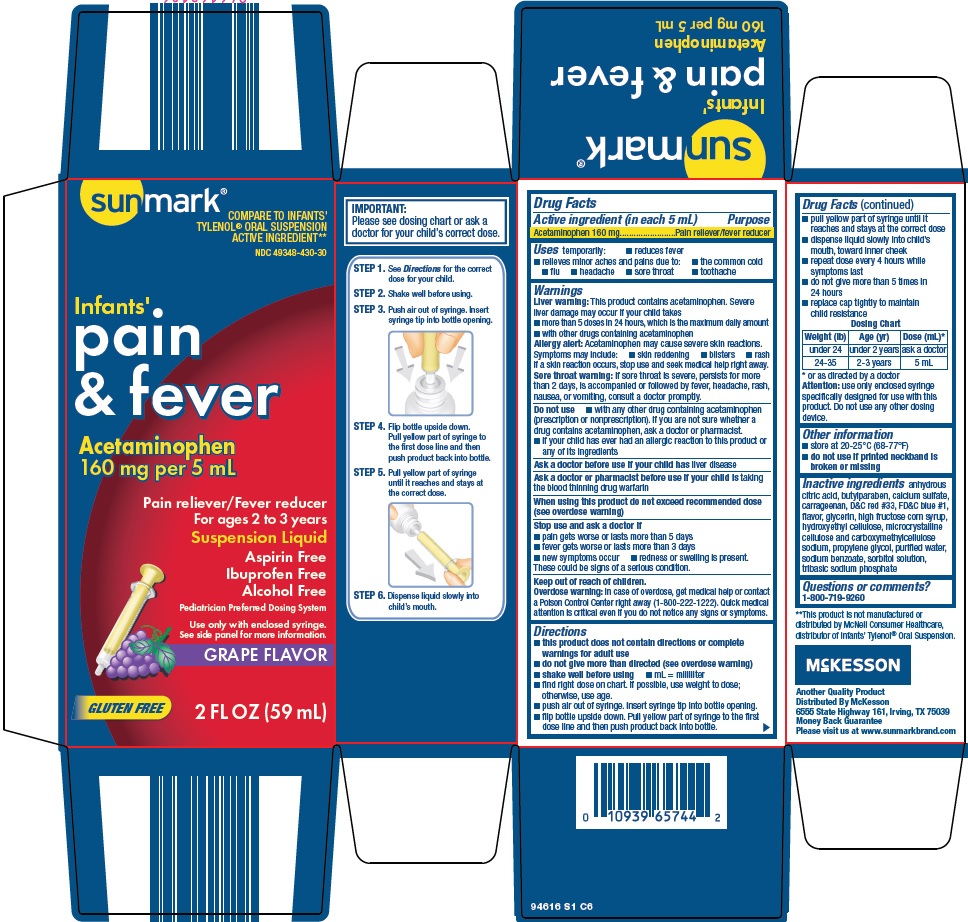
Uses
temporarily:
- •
- reduces fever
- •
- relieves minor aches and pains due to:
- •
- the common cold
- •
- flu
- •
- headache
- •
- sore throat
- •
- toothache
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if your child takes
- •
- more than 5 doses in 24 hours, which is the maximum daily amount
- •
- with other drugs containing acetaminophen
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- •
- skin reddening
- •
- blisters
- •
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- •
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- •
- if your child has ever had an allergic reaction to this product or any of its ingredients
Directions
- •
- this product does not contain directions or complete warnings for adult use
- •
- do not give more than directed (see overdose warning)
- •
- shake well before using
- •
- mL = milliliter
- •
- find right dose on chart. If possible, use weight to dose; otherwise, use age.
- •
- push air out of syringe. Insert syringe tip into bottle opening.
- •
- flip bottle upside down. Pull yellow part of syringe to the first dose line and then push product back into bottle.
- •
- pull yellow part of syringe until it reaches and stays at the correct dose
- •
- dispense liquid slowly into child’s mouth, toward inner cheek
- •
- repeat dose every 4 hours while symptoms last
- •
- do not give more than 5 times in 24 hours
- •
- replace cap tightly to maintain child resistance
|
Dosing Chart |
||
|
Weight (lb) |
Age (yr) |
Dose (mL)* |
|
under 24 |
under 2 years |
ask a doctor |
|
24-35 |
2-3 years |
5 mL |
* or as directed by a doctor
Attention: use only enclosed syringe specifically designed for use with this product. Do not use any other dosing device.
Inactive ingredients
anhydrous citric acid, butylparaben, calcium sulfate, carrageenan, D&C red #33, FD&C blue #1, flavor, glycerin, high fructose corn syrup, hydroxyethyl cellulose, microcrystalline cellulose and carboxymethylcellulose sodium, propylene glycol, purified water, sodium benzoate, sorbitol solution, tribasic sodium phosphate
Package/Label Principal Display Panel
COMPARE TO INFANTS’ TYLENOL® ORAL SUSPENSION ACTIVE INGREDIENT
Infants’
pain & fever
Acetaminophen 160 mg per 5 mL
Pain reliever / Fever reducer
For ages 2 to 3 years
Suspension Liquid
Aspirin Free
Ibuprofen Free
Alcohol Free
Pediatrician Preferred Dosing System
Use only with enclosed syringe.
See side panel for more information.
GRAPE FLAVOR
GLUTEN FREE
2 FL OZ (59 mL)