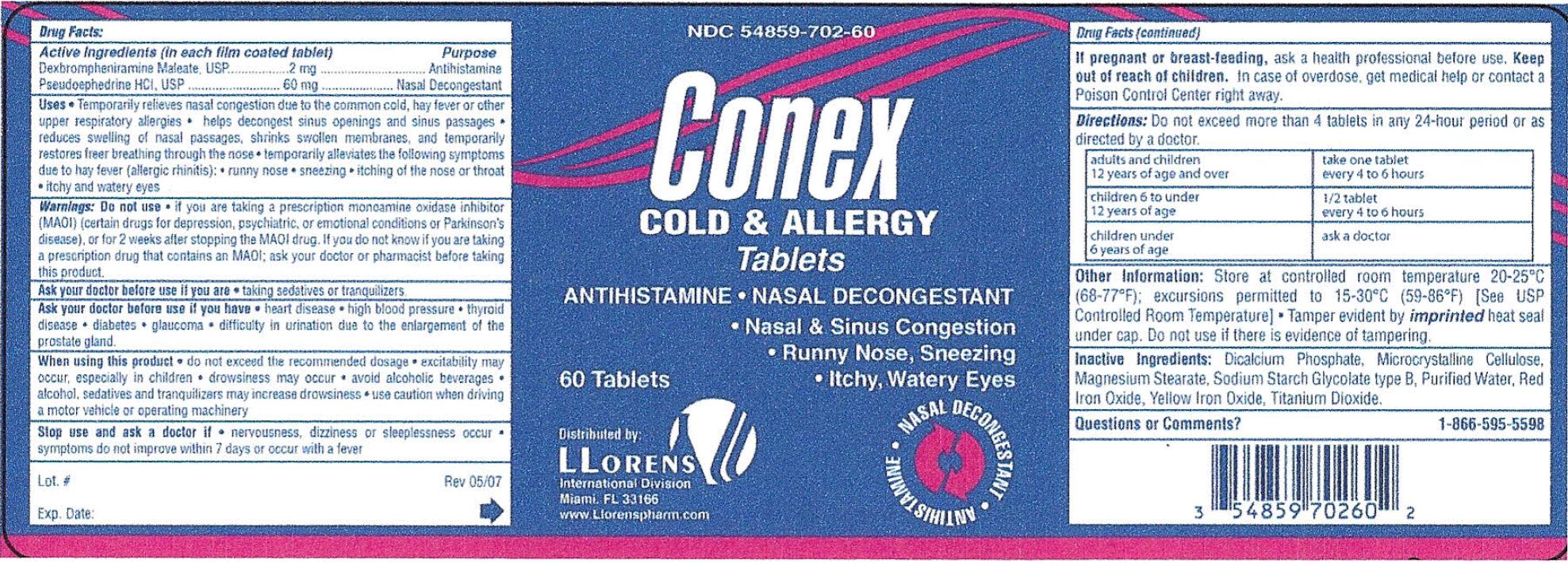
Active Ingredients (in each film coated tablet) Purpose
Dexbrompheniramine Maleate, USP ................ 2 mg .................. Antihistamine
Pseudeophedrine HCl, USP .................... 60 mg ..........................Nasal Decogestant
Uses
- Temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- helps decongest sinus openings and sinus passages
- Reduces swelling of nasal passages, shrinks swollen membranes, and temporarily restores freer breathing through the nose
- Temporarily alleviates the following symptoms due to hay fever (allergic rhinitis): runny nose, sneezing, itching of the nose or throat, itching and watery eyes.
Warnings:
Ask a doctor before you use if you are- taking sedatives or tranquilizers.
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- difficulty in urination due to the enlargement of the prostate gland.
- do not exceed the recommended dosage
- excitability may occur, especially in children
- drowsiness may occur
- avoid alcoholic beverages
- alcohol, sedatives and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- nervousness, dizziness or sleepiness occur
- symptoms do not improve withing 7 days or occur with a fever
Keep out of the reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Do not use if you are taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions
or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if you are taking a prescription drug that contains MAOI;
ask your doctor or pharmacist before taking this product.
Directions; Do not exceed more than 4 tablets in any 24-hour period or as directed by a doctor.
| Adults and children 12 years of age and over | Take one tablet every 4 to 6 hours |
| Children 6 to under 12 years of age | 1/2 tablet every 4 to 6 hours |
| Children under 6 years of age | Ask a doctor |
Other Information: Store at controlled room temperature 20 - 25 degree celsius (68 - 77 degree fahrenheit); excursions permitted to
15 - 30 degree celsius ( 59 - 86 degree fahrenheit) [ See USP Controlled Room Temperature] Tamper evident by imprinted heat seal
under cap. Do not use if there is evidence of tampering.