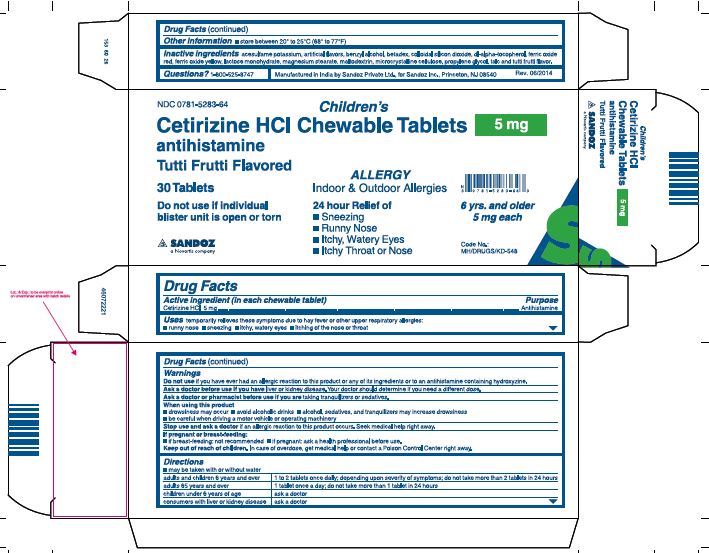
Keep Out of Reach of Children
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Uses
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- •
- runny nose
- •
- sneezing
- •
- itchy, watery eyes
- •
- itching of the nose or throat
Warnings
Do not use if you have ever had an allergic reactions to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Ask a doctor or pharmacist before use if you are taking tranquilizers or sedatives.
When using this product
- •
- drowsiness may occur
- •
- avoid alcoholic drinks
- •
- alcohol, sedatives, and tranquilizers may increase drowsiness
- •
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding:
- •
- if breast-feeding: not recommended
- •
- if pregnant: ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- •
- may be taken with or without water
|
adults and children 6 years and over |
1 to 2 tablets once daily; depending upon severity of symptoms; do not take more than 2 tablets in 24 hours |
|
adults 65 years and over |
1 tablet once a day; do not take more than 1 tablet in 24 hours |
|
children under 6 years of age |
ask a doctor |
|
consumers with liver or kidney disease |
ask a doctor |
Inactive ingredients
acesulfame potassium, artificial flavors, benzyl alcohol, betadex, colloidal silicon dioxide, dl-alpha-tocopherol, ferric oxide red, ferric oxide yellow, lactose monohydrate, magnesium stearate, maltodextrin, microcrystalline cellulose, propylene glycol, talc and tutti frutti flavor
Questions? 1-800-525-8747
Manufactured in India by Sandoz Private Ltd.,
for Sandoz Inc., Princeton, NJ 08540
Rev.06/2014
Principal Display Panel
NDC 0781-5283-64
Children's
Cetirizine HCl Chewable Tablets
5 mg
antihistamine
Tutti Frutti Flavored
30 Tablets
Do not use if individual blister unit is open or torn
ALLERGY
Indoor & Outdoor Allergies
24 hour Relief of
- •
- Sneezing
- •
- Runny Nose
- •
- Itchy, Watery Eyes
- •
- Itchy Throat or Nose
6 yrs. and older 5 mg each