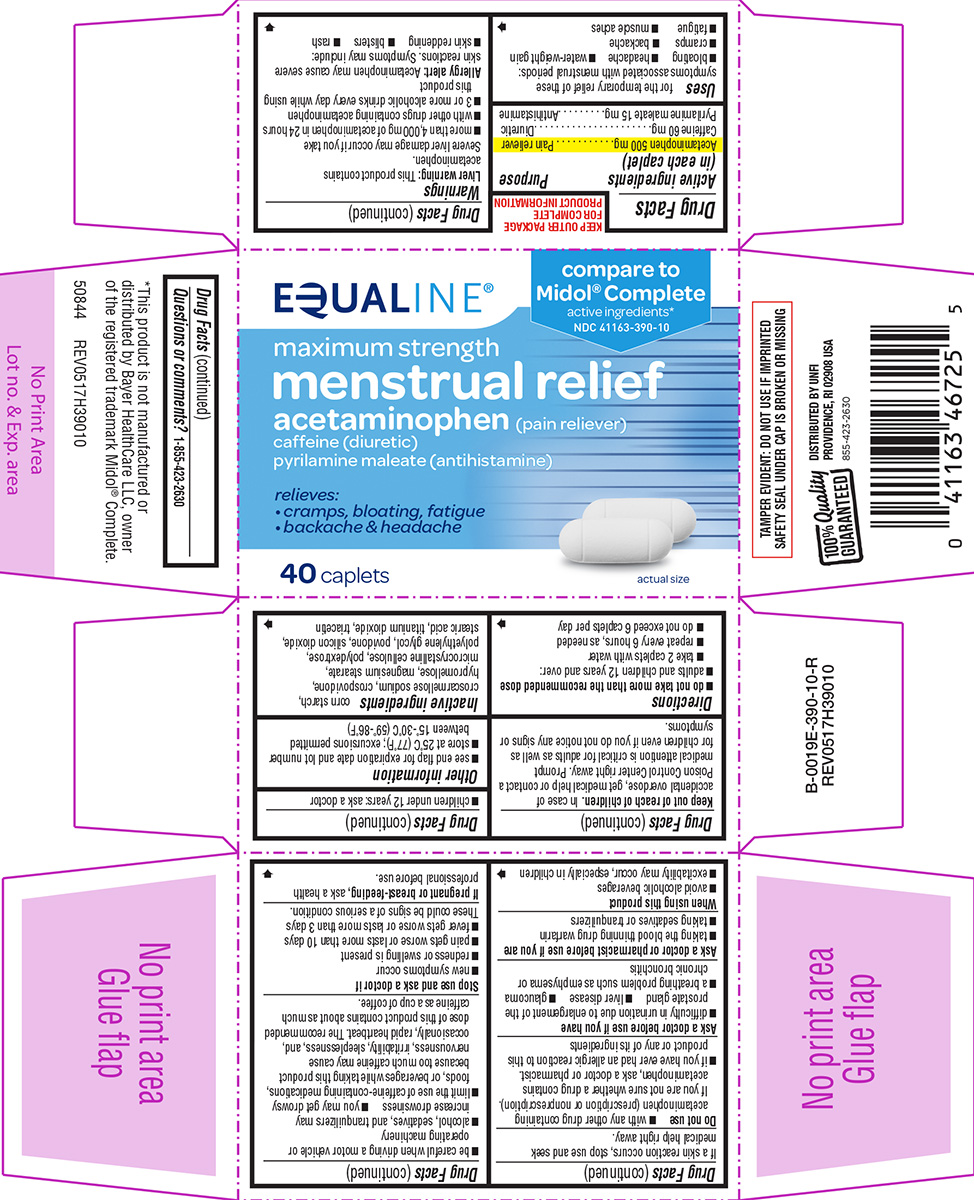
Uses
for the temporary relief of these symptoms associated with menstrual periods:
- bloating
- headache
- water-weight gain
- cramps
- backache
- fatigue
- muscle aches
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- difficulty in urination due to enlargement of the prostate gland
- liver disease
- glaucoma
- a breathing problem such as emphysema or chronic bronchitis
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- avoid alcoholic beverages
- excitability may occur, especially in children
- be careful when driving a motor vehicle or operating machinery
- alcohol, sedatives, and tranquilizers may increase drowsiness
- you may get drowsy
- limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heartbeat. The recommended dose of this product contains about as much caffeine as a cup of coffee.
Directions
- do not take more than the recommended dose
- adults and children 12 years and over:
- take 2 caplets with water
- repeat every 6 hours, as needed
- do not exceed 6 caplets per day
- children under 12 years: ask a doctor
Other information
- see end flap for expiration date and lot number
- store at 25ºC (77ºF); excursions permitted between 15º-30ºC (59º-86ºF)
Inactive ingredients
corn starch, croscarmellose sodium, crospovidone, hypromellose, magnesium stearate, microcrystalline cellulose, polydextrose, polyethylene glycol, povidone, silicon dioxide, stearic acid, titanium dioxide, triacetin
Principal Display Panel
EQUALINE®
compare to
Midol® Complete
active ingredients*
NDC 41163-390-10
maximum strength
menstrual relief
acetaminophen (pain reliever)
caffeine (diuretic)
pyrilamine maleate (antihistamine)
relieves:
• cramps, bloating, fatigue
• backache & headache
40 caplets
actual size
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING
*This product is not manufactured or
distributed by Bayer HealthCare LLC, owner
of the registered trademark Midol® Complete.
50844 REV0517H39010
100% Quality GUARANTEED
DISTRIBUTED BY UNFI
PROVIDENCE, RI 02908 USA
855-423-2630
Equaline 44-390