USE(S)
- temporarily relieves minor aches and pain due to :
- backache
- headache
- menstrual cramps
- minor pain of arthritis
- muscular aches
- the common cold
- toothache
- Temporarily reduces fever
WARNINGS
Allergy alerts: Ibuprofen may cause a severe allergy reaction, especially in people allergic to aspirin.
Symptoms may include:
- asthma (wheezing)
- blisters
- facial swelling
- hives
- rash
- shock
- skin reddening
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains a nonsteroidal anti-inflammatory drug (NSAID), which may cause stomach bleeding. The chance is higher if you:
- are age 60 or older
- have bad stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drug containing prescription NSAID (aspirin, ibuprofen, naproxen, or others)
- have 3 or more alcoholic drinks every day while using this product
- the more or for a longer time than directed
DO NOT USE
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- right before or after heart surgery
ASK A DOCTOR BEFORE USE IF
you have
- problems or serious side effects from taking pain relievers or fever reducers
- stomach problems that last or come back, such as heartburn, upset stomach, or stomach pain
- ulcers
- bleeding problems
- high blood pressure
- heart or kidney disease
- taken a diuretics
- reached age 60 or older
ASK A DOCTOR OR PHARMACIST BEFORE USE IF
you are
- taking any other drugs containg an NSAID (prescription or nonprescription)
- taking a blood thining (anticoagulant) or steriod drug
- under a doctor’s care for any serious condition
- taking aspirin for heart attacks or stroke, because ibuprofen may decrease this benefit of aspirin
- taking any other drug
WHEN USING THIS PRODUCT
- take with food or milk if stomach upset occurs
- long term continuous use may increase the risk of heart attack or stroke
STOP USE AND ASK DOCTOR IF
- you feel faint, vomit blood, or have bloody or black stools.
These are signs of stomach bleeding.
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- stomach pain or upset gets worse or lasts
- redness or swelling is present in painful area
- any new symptoms appear
PREGNANCY/BREASTFEEDING
ask a health professional before use. It is especially important not to use ibuprofen during the last 3 months of pregnancy unless definitely directed to do so by a doctor because it may cause a problems in the unborn child or complications during delivery.
KEEP OUT OF REACH OF CHILDREN
In case of overdose, get medical help or contact a PoisonControlCenter right away.
DIRECTIONS
- do not take more than directed
- the smallest effective dose should be used
- do not take longer than 10 days, unless directed by a doctor
(see Warnings)
| adults and children 12 years and older |
|
| Children under 12 years |
|
INACTIVE INGREDIENT(S)
Colloidal silicon dioxide, Croscarmellose Sodium, Magnesium stearate, Microcrystalline sodium, Pregelatinised starch, talc.
STORAGE
- store between 20-25 0c (68-77 0 F).
- do not use if seal under bottle cap imprinted with” SEALED for YOUR PROTECTION” is broken or missing.
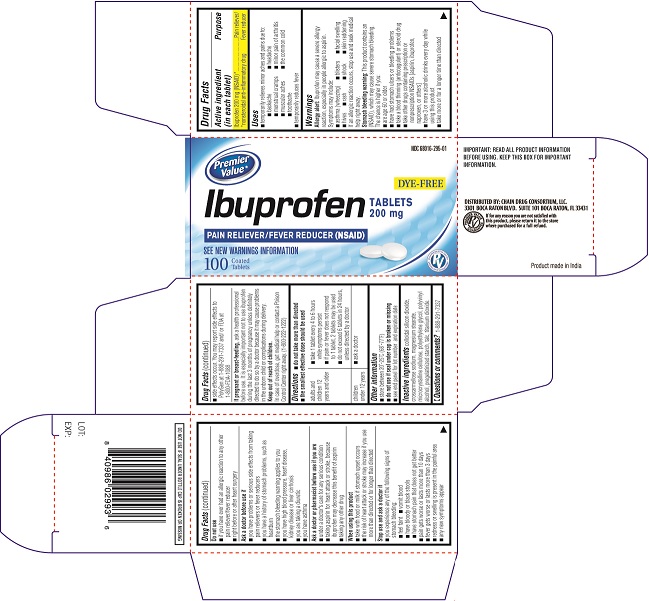
PRINCIPAL DISPLAY PANEL
Carton Label PDP
NDC# 68016-295-01
DYE FREE
Ibuprofen Tablets 200 mg
PAIN RELIEVER/FEVER REDUCER (NSAID)
SEE NEW WARNINGS INFORMATION
100 COATED TABLETS
Bottle Label PDP
NDC# 68016-295-01
DYE FREE
Ibuprofen Tablets 200 mg
PAIN RELIEVER/FEVER REDUCER (NSAID)
SEE NEW WARNINGS INFORMATION
100 COATED TABLETS

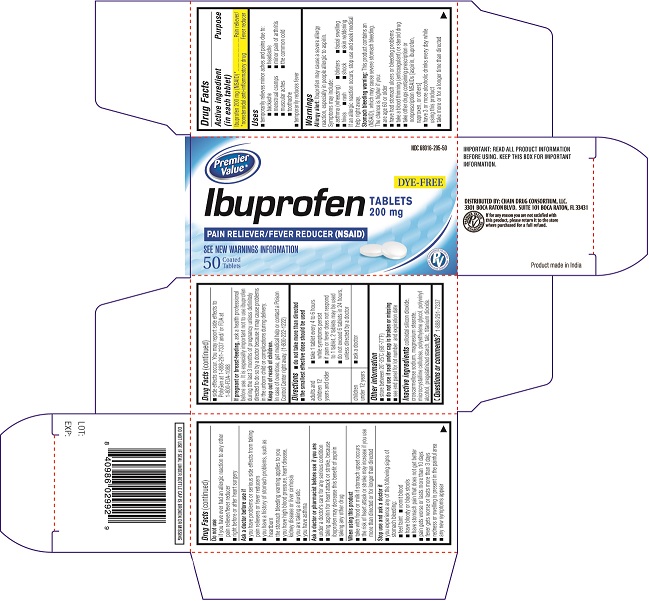
Carton Label PDP
NDC# 68016-295-50
DYE FREE
Ibuprofen Tablets 200 mg
PAIN RELIEVER/FEVER REDUCER (NSAID)
SEE NEW WARNINGS INFORMATION
50 COATED TABLETS
Bottle Label PDP
NDC# 68016-295-50
DYE FREE
Ibuprofen Tablets 200 mg
PAIN RELIEVER/FEVER REDUCER (NSAID)
SEE NEW WARNINGS INFORMATION
50 COATED TABLETS
