FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
INTELENCE, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-experienced adult patients and pediatric patients 2 years of age and older [see Microbiology (12.4)and Clinical Studies (14)] .
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Adult Patients
The recommended oral dosage of INTELENCE for adult patients is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal. The type of food does not affect the exposure to INTELENCE [see Clinical Pharmacology (12.3)] .
2.2 Recommended Dosage During Pregnancy
The recommended oral dosage of INTELENCE for pregnant individuals is 200 mg (one 200 mg tablet or two 100 mg tablets) taken twice daily following a meal [see Use in Specific Populations (8.1)] .
2.3 Recommended Dosage in Pediatric Patients (2 Years to Less Than 18 Years of Age)
The recommended dosage of INTELENCE for pediatric patients 2 years to less than 18 years of age and weighing at least 10 kg is based on body weight (see Table 1) not exceeding the recommended adult dosage. INTELENCE should be taken orally, following a meal. The type of food does not affect the exposure to INTELENCE [see Clinical Pharmacology (12.3)] .
| Body Weight
kilograms (kg) | Dose |
|---|---|
| greater than or equal to 10 kg to less than 20 kg | 100 mg twice daily |
| greater than or equal to 20 kg to less than 25 kg | 125 mg twice daily |
| greater than or equal to 25 kg to less than 30 kg | 150 mg twice daily |
| greater than or equal to 30 kg | 200 mg twice daily |
2.4 Method of Administration
Instruct patients to swallow the INTELENCE tablet(s) whole with liquid such as water. Patients who are unable to swallow the INTELENCE tablet(s) whole may disperse the tablet(s) in water. Instruct the patient to do the following:
- place the tablet(s) in 5 mL (1 teaspoon) of water, or at least enough liquid to cover the medication,
- stir well until the water looks milky,
- add approximately 15 mL (1 tablespoon) of liquid. Water may be used but other liquids, such as orange juice or milk, may improve taste. Patients should not place the tablets in orange juice or milk without first adding water. The use of warm (temperature greater than 104°F [greater than 40°C]) or carbonated beverages should be avoided.
- drink the mixture immediately,
- rinse the glass several times with orange juice, milk or water and completely swallow the rinse each time to make sure the patient takes the entire dose.
3 DOSAGE FORMS AND STRENGTHS
- 25 mg white to off-white, oval, scored tablets debossed with "TMC" on one side.
- 100 mg white to off-white oval tablets debossed with "TMC125" on one side and "100" on the other side.
- 200 mg white to off-white, biconvex, oblong tablets debossed with "T200" on one side.
5 WARNINGS AND PRECAUTIONS
5.1 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening and fatal skin reactions have been reported. In clinical trials, these include cases of Stevens-Johnson syndrome, toxic epidermal necrolysis and erythema multiforme. Hypersensitivity reactions including Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) have also been reported and were characterized by rash, constitutional findings, and sometimes organ dysfunction, including hepatic failure. In Phase 3 clinical trials, Grade 3 and 4 rashes were reported in 1.3% of subjects receiving INTELENCE compared to 0.2% of placebo subjects. A total of 2.2% of HIV-1-infected subjects receiving INTELENCE discontinued from Phase 3 trials due to rash [see Adverse Reactions (6.1)] . Rash occurred most commonly during the first 6 weeks of therapy. The incidence of rash was higher in females [see Adverse Reactions (6.1)] . Stevens-Johnson syndrome was reported in 1.1% (2/177) of pediatric patients less than 18 years of age receiving INTELENCE in combination with other HIV-1 antiretroviral agents in an observational study.
Discontinue INTELENCE immediately if signs or symptoms of severe skin reactions or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, angioedema). Clinical status including liver transaminases should be monitored and appropriate therapy initiated. Delay in stopping INTELENCE treatment after the onset of severe rash may result in a life-threatening reaction.
5.2 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
The concomitant use of INTELENCE and other drugs may result in potentially significant drug interactions, some of which may lead to [see Drug Interactions (7.3)]:
- Loss of therapeutic effect of concomitant drug or INTELENCE and possible development of resistance.
- Possible clinically significant adverse reactions from greater exposures of INTELENCE or other concomitant drugs.
See Table 4for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during INTELENCE therapy and review concomitant medications during INTELENCE therapy.
5.3 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including INTELENCE. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jirovecipneumonia (PCP) or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
5.4 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
6 ADVERSE REACTIONS
The following adverse reactions are described in greater detail in other sections:
- Severe skin and hypersensitivity reactions [see Warnings and Precautions (5.1)] .
- Immune reconstitution syndrome [see Warnings and Precautions (5.3)] .
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials Experience in Adults
The safety assessment is based on all data from 1203 subjects in the Phase 3 placebo-controlled trials, TMC125-C206 and TMC125-C216, conducted in antiretroviral treatment-experienced HIV-1-infected adult subjects, 599 of whom received INTELENCE (200 mg twice daily). In these pooled trials, the median exposure for subjects in the INTELENCE arm and placebo arm was 52.3 and 51.0 weeks, respectively. Discontinuations due to adverse drug reactions (ADRs) were 5.2% in the INTELENCE arm and 2.6% in the placebo arm.
The most frequently reported ADR at least Grade 2 in severity was rash (10.0%). Stevens-Johnson syndrome, drug hypersensitivity reaction and erythema multiforme were reported in less than 0.1% of subjects during clinical development with INTELENCE [see Warnings and Precautions (5.1)] . A total of 2.2% of HIV-1-infected subjects in Phase 3 trials receiving INTELENCE discontinued due to rash. In general, in clinical trials, rash was mild to moderate, occurred primarily in the second week of therapy, and was infrequent after Week 4. Rash generally resolved within 1 to 2 weeks on continued therapy. The incidence of rash was higher in women compared to men in the INTELENCE arm in the Phase 3 trials (rash ≥ Grade 2 was reported in 9/60 [15.0%] women versus 51/539 [9.5%] men; discontinuations due to rash were reported in 3/60 [5.0%] women versus 10/539 [1.9%] men) [see Warnings and Precautions (5.1)] . Patients with a history of NNRTI-related rash did not appear to be at increased risk for the development of INTELENCE-related rash compared to patients without a history of NNRTI-related rash.
Common Adverse Reactions
Clinical ADRs of moderate intensity or greater (greater than or equal to Grade 2) and reported in at least 2% of subjects treated with INTELENCE and occurring at a higher rate compared to placebo (excess of 1%) are presented in Table 2. Laboratory abnormalities considered ADRs are included in Table 3.
| Preferred Term | INTELENCE + BR
N=599 % | Placebo + BR
N=604 % |
|---|---|---|
| N=total number of subjects per treatment group; BR=background regimen | ||
| Rash | 10% | 3% |
| Peripheral neuropathy | 4% | 2% |
Less Common Adverse Reactions
Treatment-emergent ADRs occurring in less than 2% of subjects (599 subjects) receiving INTELENCE and of at least moderate intensity (greater than or equal to Grade 2) are listed below by body system:
Cardiac Disorders: myocardial infarction, angina pectoris, atrial fibrillation
Ear and Labyrinth Disorders: vertigo
Eye Disorders: blurred vision
Gastrointestinal Disorders: gastroesophageal reflux disease, flatulence, gastritis, abdominal distension, pancreatitis, constipation, dry mouth, hematemesis, retching, stomatitis
General Disorders and Administration Site Conditions: sluggishness
Hematologic Disorders: hemolytic anemia
Hepatobiliary Disorders: hepatic failure, hepatomegaly, cytolytic hepatitis, hepatic steatosis, hepatitis
Immune System Disorders: drug hypersensitivity, immune reconstitution syndrome
Metabolism and Nutrition Disorders: diabetes mellitus, anorexia, dyslipidemia
Nervous System Disorders: paresthesia, somnolence, convulsion, hypoesthesia, amnesia, syncope, disturbance in attention, hypersomnia, tremor
Psychiatric Disorders: anxiety, sleep disorders, abnormal dreams, confusional state, disorientation, nervousness, nightmares
Renal and Urinary Disorders:acute renal failure
Reproductive System and Breast Disorders: gynecomastia
Respiratory, Thoracic and Mediastinal Disorders: exertional dyspnea, bronchospasm
Skin and Subcutaneous Tissue Disorders: night sweats, lipohypertrophy, prurigo, hyperhidrosis, dry skin, swelling face
Additional ADRs of at least moderate intensity observed in other trials were acquired lipodystrophy, angioneurotic edema, erythema multiforme and hemorrhagic stroke, each reported in no more than 0.5% of subjects.
Laboratory Abnormalities in Treatment-Experienced Patients
Selected Grade 2 to Grade 4 laboratory abnormalities that represent a worsening from baseline observed in adult subjects treated with INTELENCE are presented in Table 3.
| Laboratory Parameter | DAIDS Toxicity Range | INTELENCE + BR
N=599 % | Placebo + BR
N=604 % |
|---|---|---|---|
| ULN=Upper Limit of Normal; BR=background regimen | |||
| GENERAL BIOCHEMISTRY | |||
| Pancreatic amylase | |||
| Grade 2 | > 1.5–2 × ULN | 7% | 8% |
| Grade 3 | > 2–5 × ULN | 7% | 8% |
| Grade 4 | > 5 × ULN | 2% | 1% |
| Lipase | |||
| Grade 2 | > 1.5–3 × ULN | 4% | 6% |
| Grade 3 | > 3–5 × ULN | 2% | 2% |
| Grade 4 | > 5 × ULN | 1% | < 1% |
| Creatinine | |||
| Grade 2 | > 1.4–1.8 × ULN | 6% | 5% |
| Grade 3 | > 1.9–3.4 × ULN | 2% | 1% |
| Grade 4 | > 3.4 × ULN | 0% | < 1% |
| HEMATOLOGY | |||
| Decreased hemoglobin | |||
| Grade 2 | 90–99 g/L | 2% | 4% |
| Grade 3 | 70–89 g/L | < 1% | < 1% |
| Grade 4 | < 70 g/L | < 1% | < 1% |
| White blood cell count | |||
| Grade 2 | 1,500–1,999/mm 3 | 2% | 3% |
| Grade 3 | 1,000–1,499/mm 3 | 1% | 4% |
| Grade 4 | < 1,000/mm 3 | 1% | < 1% |
| Neutrophils | |||
| Grade 2 | 750–999/mm 3 | 5% | 6% |
| Grade 3 | 500–749/mm 3 | 4% | 4% |
| Grade 4 | < 500/mm 3 | 2% | 3% |
| Platelet count | |||
| Grade 2 | 50,000–99,999/mm 3 | 3% | 5% |
| Grade 3 | 25,000–49,999/mm 3 | 1% | 1% |
| Grade 4 | < 25,000/mm 3 | < 1% | < 1% |
| LIPIDS AND GLUCOSE | |||
| Total cholesterol | |||
| Grade 2 | > 6.20–7.77 mmol/L
240–300 mg/dL | 20% | 17% |
| Grade 3 | > 7.77 mmol/L
> 300 mg/dL | 8% | 5% |
| Low density lipoprotein | |||
| Grade 2 | 4.13–4.9 mmol/L
160–190 mg/dL | 13% | 12% |
| Grade 3 | > 4.9 mmol/L
> 190 mg/dL | 7% | 7% |
| Triglycerides | |||
| Grade 2 | 5.65–8.48 mmol/L
500–750 mg/dL | 9% | 7% |
| Grade 3 | 8.49–13.56 mmol/L
751–1200 mg/dL | 6% | 4% |
| Grade 4 | > 13.56 mmol/L
> 1200 mg/dL | 4% | 2% |
| Elevated glucose levels | |||
| Grade 2 | 6.95–13.88 mmol/L
161–250 mg/dL | 15% | 13% |
| Grade 3 | 13.89–27.75 mmol/L
251–500 mg/dL | 4% | 2% |
| Grade 4 | > 27.75 mmol/L
> 500 mg/dL | 0% | < 1% |
| HEPATIC PARAMETERS | |||
| Alanine amino transferase | |||
| Grade 2 | 2.6–5 × ULN | 6% | 5% |
| Grade 3 | 5.1–10 × ULN | 3% | 2% |
| Grade 4 | > 10 × ULN | 1% | < 1% |
| Aspartate amino transferase | |||
| Grade 2 | 2.6–5 × ULN | 6% | 8% |
| Grade 3 | 5.1–10 × ULN | 3% | 2% |
| Grade 4 | > 10 × ULN | < 1% | < 1% |
Patients Co-Infected With Hepatitis B and/or Hepatitis C Virus
In Phase 3 trials TMC125-C206 and TMC125-C216, 139 subjects (12.3%) with chronic hepatitis B and/or hepatitis C virus co-infection out of 1129 subjects were permitted to enroll. AST and ALT abnormalities occurred more frequently in hepatitis B and/or hepatitis C virus co-infected subjects for both treatment groups. Grade 2 or higher laboratory abnormalities that represent a worsening from baseline of AST, ALT or total bilirubin occurred in 27.8%, 25.0% and 7.1% respectively, of INTELENCE-treated co-infected subjects as compared to 6.7%, 7.5% and 1.8% of non-co-infected INTELENCE-treated subjects. In general, adverse events reported by INTELENCE-treated subjects with hepatitis B and/or hepatitis C virus co-infection were similar to INTELENCE-treated subjects without hepatitis B and/or hepatitis C virus co-infection.
Clinical Trials Experience in Pediatric Subjects (2 Years to Less Than 18 years of age)
The safety assessment in pediatric subjects is based on two single-arm trials. TMC125-C213 is a Phase 2 trial in which 101 antiretroviral treatment-experienced HIV-1 infected pediatric subjects 6 years to less than 18 years of age received INTELENCE in combination with other antiretroviral agents (Week 24 analysis). TMC125-C234/IMPAACT P1090 is a Phase 1/2 trial in which 20 antiretroviral treatment-experienced HIV-1 infected pediatric subjects 2 years to less than 6 years of age received INTELENCE in combination with other antiretroviral agents (Week 24 analysis) [see Clinical Studies (14.2)] .
In TMC125-C213, the frequency, type and severity of adverse drug reactions in pediatric subjects 6 years to less than 18 years of age were comparable to those observed in adult subjects, except for rash which was observed more frequently in pediatric subjects. The most common adverse drug reactions in at least 2% of pediatric subjects were rash and diarrhea. Rash was reported more frequently in female subjects than in male subjects (rash ≥ Grade 2 was reported in 13/64 [20.3%] females versus 2/37 [5.4%] males; discontinuations due to rash were reported in 4/64 [6.3%] females versus 0/37 [0%] males). Rash (greater than or equal to Grade 2) occurred in 15% of pediatric subjects from 6 years to less than 18 years of age. In the majority of cases, rash was mild to moderate, of macular/papular type, and occurred in the second week of therapy. Rash was self-limiting and generally resolved within 1 week on continued therapy. The safety profile for subjects who completed 48 weeks of treatment was similar to the safety profile for subjects who completed 24 weeks of treatment.
In TMC125-C234/IMPAACT P1090, the frequency, type and severity of adverse drug reactions in pediatric subjects 2 years to less than 6 years of age through Week 24 were comparable to those observed in adults. The most common adverse drug reactions (any grade) of pediatric subjects were rash (50% [10/20]) and diarrhea (25% [5/20]). In this age group, no subjects had Grade 3 or Grade 4 rash and no subjects discontinued prematurely due to rash. One subject discontinued etravirine due to asymptomatic lipase elevation.
6.2 Postmarketing Experience
The following events have been identified during postmarketing use of INTELENCE. Because these events are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Severe hypersensitivity reactions including DRESS and cases of hepatic failure have been reported [see Warnings and Precautions (5.1)] .
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis
Skin and Subcutaneous Tissue Disorders: Fatal cases of toxic epidermal necrolysis and Stevens-Johnson syndrome have been reported [see Warnings and Precautions (5.1)] .
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect INTELENCE
Etravirine is a substrate of CYP3A, CYP2C9, and CYP2C19. Therefore, co-administration of INTELENCE with drugs that induce or inhibit CYP3A, CYP2C9, and CYP2C19 may alter the therapeutic effect or adverse reaction profile of INTELENCE (see Table 4) [see Clinical Pharmacology (12.3)].
7.2 Potential for INTELENCE to Affect Other Drugs
Etravirine is an inducer of CYP3A and inhibitor of CYP2C9, CYP2C19 and P-glycoprotein (P-gp). Therefore, co-administration of drugs that are substrates of CYP3A, CYP2C9 and CYP2C19 or are transported by P-gp with INTELENCE may alter the therapeutic effect or adverse reaction profile of the co-administered drug(s) (see Table 4) [see Clinical Pharmacology (12.3)].
7.3 Significant Drug Interactions
Table 4 shows significant drug interactions based on which, alterations in dose or regimen of INTELENCE and/or co-administered drug may be recommended. Drugs that are not recommended for co-administration with INTELENCE are also included in Table 4 [see Clinical Pharmacology (12.3)] .
| Concomitant Drug Class:
Drug Name | Effect on Concentration of Etravirine or Concomitant Drug | Clinical Comment |
|---|---|---|
| ↑ = increase; ↓ = decrease; ↔ = no change | ||
| HIV-antiviral agents: integrase strand inhibitors | ||
| dolutegravir * | ↓ dolutegravir
↔ etravirine | Etravirine significantly reduced plasma concentrations of dolutegravir. Using cross -study comparisons to historical pharmacokinetic data for etravirine, dolutegravir did not appear to affect the pharmacokinetics of etravirine. |
| dolutegravir/darunavir/ritonavir * | ↓ dolutegravir
↔ etravirine | The effect of etravirine on dolutegravir plasma concentrations was mitigated by co-administration of darunavir/ritonavir or lopinavir/ritonavir, and is expected to be mitigated by atazanavir/ritonavir. Dolutegravir should only be used with INTELENCE when co-administered with atazanavir/ritonavir, darunavir/ritonavir, or lopinavir/ritonavir. |
| dolutegravir/lopinavir/ritonavir * | ↔ dolutegravir
↔ etravirine |
|
| HIV-antiviral agents: non-nucleoside reverse transcriptase inhibitors (NNRTIs) | ||
| efavirenz
*
nevirapine * | ↓ etravirine | Combining two NNRTIs has not been shown to be beneficial. Concomitant use of INTELENCE with efavirenz or nevirapine may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of INTELENCE. Co-administration of INTELENCE and other NNRTIs is not recommended. |
| delavirdine | ↑ etravirine | Combining two NNRTIs has not been shown to be beneficial. INTELENCE and delavirdine should not be co-administered. |
| rilpivirine | ↓ rilpivirine
↔ etravirine | Combining two NNRTIs has not been shown to be beneficial. Co-administration of INTELENCE and rilpivirine is not recommended. |
| HIV-antiviral agents: protease inhibitors (PIs) | ||
| atazanavir
*
(without ritonavir) | ↓ atazanavir | Co-administration of INTELENCE and atazanavir without low-dose ritonavir is not recommended. |
| atazanavir/ritonavir * | ↓ atazanavir
↔ etravirine | Concomitant use of INTELENCE with atazanavir/ritonavir decreased atazanavir C minbut it is not considered clinically relevant. The mean systemic exposure (AUC) of etravirine after co-administration of INTELENCE with atazanavir/ritonavir in HIV-infected subjects was similar to the mean systemic exposure of etravirine observed in the Phase 3 trials after co-administration of INTELENCE and darunavir/ritonavir (as part of the background regimen). INTELENCE and atazanavir/ritonavir can be co-administered without dose adjustments. |
| atazanavir/cobicistat | ↓ atazanavir
↓ cobicistat | Co-administration of INTELENCE with atazanavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to atazanavir. |
| darunavir/ritonavir * | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when INTELENCE was co-administered with darunavir/ritonavir. Because all subjects in the Phase 3 trials received darunavir/ritonavir as part of the background regimen and etravirine exposures from these trials were determined to be safe and effective, INTELENCE and darunavir/ritonavir can be co-administered without dose adjustments. |
| darunavir/cobicistat | ↓ cobicistat
darunavir: effect unknown | Co-administration of INTELENCE with darunavir/cobicistat is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. |
| fosamprenavir
(without ritonavir) | ↑ amprenavir | Concomitant use of INTELENCE with fosamprenavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of amprenavir. Co-administration of INTELENCE and fosamprenavir without low-dose ritonavir is not recommended. |
| fosamprenavir/ritonavir * | ↑ amprenavir | Due to a significant increase in the systemic exposure of amprenavir, the appropriate doses of the combination of INTELENCE and fosamprenavir/ritonavir have not been established. Co-administration of INTELENCE and fosamprenavir/ritonavir is not recommended. |
| indinavir
*
(without ritonavir) | ↓ indinavir | Concomitant use of INTELENCE with indinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of indinavir. Co-administration of INTELENCE and indinavir without low-dose ritonavir is not recommended. |
| lopinavir/ritonavir * | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced after co-administration of INTELENCE with lopinavir/ritonavir (tablet). Because the reduction in the mean systemic exposures of etravirine in the presence of lopinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, INTELENCE and lopinavir/ritonavir can be co-administered without dose adjustments. |
| nelfinavir
(without ritonavir) | ↑ nelfinavir | Concomitant use of INTELENCE with nelfinavir without low-dose ritonavir may cause a significant alteration in the plasma concentration of nelfinavir. Co-administration of INTELENCE and nelfinavir without low-dose ritonavir is not recommended. |
| ritonavir * | ↓ etravirine | Concomitant use of INTELENCE with ritonavir 600 mg twice daily may cause a significant decrease in the plasma concentration of etravirine and loss of therapeutic effect of INTELENCE. Co-administration of INTELENCE and ritonavir 600 mg twice daily is not recommended. |
| saquinavir/ritonavir * | ↓ etravirine | The mean systemic exposure (AUC) of etravirine was reduced when INTELENCE was co-administered with saquinavir/ritonavir. Because the reduction in the mean systemic exposures of etravirine in the presence of saquinavir/ritonavir is similar to the reduction in mean systemic exposures of etravirine in the presence of darunavir/ritonavir, INTELENCE and saquinavir/ritonavir can be co-administered without dose adjustments. |
| tipranavir/ritonavir * | ↓ etravirine | Concomitant use of INTELENCE with tipranavir/ritonavir may cause a significant decrease in the plasma concentrations of etravirine and loss of therapeutic effect of INTELENCE. Co-administration of INTELENCE and tipranavir/ritonavir is not recommended. |
| CCR5 antagonists | ||
| maraviroc * | ↔ etravirine
↓ maraviroc | When INTELENCE is co-administered with maraviroc in the absence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 600 mg twice daily. No dose adjustment of INTELENCE is needed. |
| maraviroc/darunavir/ritonavir *† | ↔ etravirine
↑ maraviroc | When INTELENCE is co-administered with maraviroc in the presence of a potent CYP3A inhibitor (e.g., ritonavir boosted protease inhibitor), the recommended dose of maraviroc is 150 mg twice daily. No dose adjustment of INTELENCE is needed. |
| Other agents | ||
| Antiarrhythmics:
digoxin * | ↔ etravirine
↑ digoxin | For patients who are initiating a combination of INTELENCE and digoxin, the lowest dose of digoxin should initially be prescribed. For patients on a stable digoxin regimen and initiating INTELENCE, no dose adjustment of either INTELENCE or digoxin is needed. The serum digoxin concentrations should be monitored and used for titration of the digoxin dose to obtain the desired clinical effect. |
| amiodarone
bepridil disopyramide flecainide lidocaine (systemic) mexiletine propafenone quinidine | ↓ antiarrhythmics | Concentrations of these antiarrhythmics may be decreased when co-administered with INTELENCE. INTELENCE and antiarrhythmics should be co-administered with caution. Drug concentration monitoring is recommended, if available. |
| Anticoagulant:
warfarin | ↑ anticoagulants | Warfarin concentrations may be increased when co-administered with INTELENCE. The international normalized ratio (INR) should be monitored when warfarin is combined with INTELENCE. |
| Anticonvulsants:
carbamazepine phenobarbital phenytoin | ↓ etravirine | Carbamazepine, phenobarbital and phenytoin are inducers of CYP450 enzymes. INTELENCE should not be used in combination with carbamazepine, phenobarbital, or phenytoin as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of INTELENCE. |
| Antifungals:
fluconazole * | ↑ etravirine
↔ fluconazole | Co-administration of etravirine and fluconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and fluconazole should be co-administered with caution. No dose adjustment of INTELENCE or fluconazole is needed. |
| voriconazole * | ↑ voriconazole | Co-administration of etravirine and voriconazole significantly increased etravirine exposures. The amount of safety data at these increased etravirine exposures is limited, therefore, etravirine and voriconazole should be co-administered with caution. No dose adjustment of INTELENCE or voriconazole is needed. |
| Antifungals:
itraconazole ketoconazole posaconazole | ↑ etravirine
↓ itraconazole ↓ ketoconazole ↔ posaconazole | Posaconazole, a potent inhibitor of CYP3A4, may increase plasma concentrations of etravirine. Itraconazole and ketoconazole are potent inhibitors as well as substrates of CYP3A4. Concomitant systemic use of itraconazole or ketoconazole and INTELENCE may increase plasma concentrations of etravirine. Simultaneously, plasma concentrations of itraconazole or ketoconazole may be decreased by INTELENCE. Dose adjustments for itraconazole, ketoconazole or posaconazole may be necessary depending on the other co-administered drugs. |
| Antiinfective:
clarithromycin * | ↑ etravirine
↓ clarithromycin ↑ 14-OH-clarithromycin | Clarithromycin exposure was decreased by INTELENCE; however, concentrations of the active metabolite, 14-hydroxy-clarithromycin, were increased. Because 14-hydroxy-clarithromycin has reduced activity against Mycobacterium aviumcomplex (MAC), overall activity against this pathogen may be altered. Alternatives to clarithromycin, such as azithromycin, should be considered for the treatment of MAC. |
| Antimalarial:
artemether/lumefantrine * | ↔ etravirine
↓ artemether ↓ dihydroartemisinin ↓ lumefantrine | Caution is warranted when co-administering INTELENCE and artemether/lumefantrine as it is unknown whether the decrease in exposure of artemether or its active metabolite, dihydroartemisinin, could result in decreased antimalarial efficacy. No dose adjustment is needed for INTELENCE. |
| Antimycobacterials:
rifampin rifapentine | ↓ etravirine | Rifampin and rifapentine are potent inducers of CYP450 enzymes. INTELENCE should not be used with rifampin or rifapentine as co-administration may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of INTELENCE. |
| Antimycobacterial:
rifabutin * | ↓ etravirine
↓ rifabutin ↓ 25- O-desacetylrifabutin | If INTELENCE is NOT co-administered with a protease inhibitor/ritonavir, then rifabutin at a dose of 300 mg once daily is recommended.
If INTELENCE is co-administered with darunavir/ritonavir, lopinavir/ritonavir or saquinavir/ritonavir, then rifabutin should not be co-administered due to the potential for significant reductions in etravirine exposure. |
| Benzodiazepine:
diazepam | ↑ diazepam | Concomitant use of INTELENCE with diazepam may increase plasma concentrations of diazepam. A decrease in diazepam dose may be needed. |
| Corticosteroid:
dexamethasone (systemic) | ↓ etravirine | Systemic dexamethasone induces CYP3A and can decrease etravirine plasma concentrations. This may result in loss of therapeutic effect of INTELENCE. Systemic dexamethasone should be used with caution or alternatives should be considered, particularly for long-term use. |
| Herbal products:
St. John's wort ( Hypericum perforatum) | ↓ etravirine | Concomitant use of INTELENCE with products containing St. John's wort may cause significant decreases in etravirine plasma concentrations and loss of therapeutic effect of INTELENCE. INTELENCE and products containing St. John's wort should not be co-administered. |
| Hepatitis C virus (HCV) direct-acting antivirals: | ||
| daclatasvir | ↓ daclatasvir | Co-administration of INTELENCE with daclatasvir may decrease daclatasvir concentrations. Increase the daclatasvir dose to 90 mg once daily. |
| elbasvir/grazoprevir | ↓ elbasvir
↓ grazoprevir | Co-administration of INTELENCE with elbasvir/grazoprevir may decrease elbasvir and grazoprevir concentrations, leading to reduced therapeutic effect of elbasvir/grazoprevir. Co-administration is not recommended. |
| HMG-CoA reductase inhibitors:
atorvastatin * | ↔ etravirine
↓ atorvastatin ↑ 2-OH-atorvastatin | The combination of INTELENCE and atorvastatin can be given without dose adjustments, however, the dose of atorvastatin may need to be altered based on clinical response. |
| pravastatin
rosuvastatin | ↔ etravirine
↔ pravastatin ↔ rosuvastatin | No interaction between pravastatin, rosuvastatin and INTELENCE is expected. |
| lovastatin
simvastatin | ↓ lovastatin
↓ simvastatin | Lovastatin and simvastatin are CYP3A substrates and co-administration with INTELENCE may result in lower plasma concentrations of the HMG-CoA reductase inhibitor. |
| fluvastatin
pitavastatin | ↑ fluvastatin
↑ pitavastatin | Fluvastatin and pitavastatin are metabolized by CYP2C9 and co-administration with INTELENCE may result in higher plasma concentrations of the HMG-CoA reductase inhibitor. Dose adjustments for these HMG-CoA reductase inhibitors may be necessary. |
| Immunosuppressants:
cyclosporine sirolimus tacrolimus | ↓ immunosuppressant | INTELENCE and systemic immunosuppressants should be co-administered with caution because plasma concentrations of cyclosporine, sirolimus, or tacrolimus may be affected. |
| Narcotic analgesics/treatment of opioid dependence:
buprenorphine buprenorphine/naloxone * methadone * | ↔ etravirine
↓ buprenorphine ↔ norbuprenorphine ↔ methadone | INTELENCE and buprenorphine (or buprenorphine/naloxone) can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as buprenorphine (or buprenorphine/naloxone) maintenance therapy may need to be adjusted in some patients.
INTELENCE and methadone can be co-administered without dose adjustments, however, clinical monitoring for withdrawal symptoms is recommended as methadone maintenance therapy may need to be adjusted in some patients. |
| Phosphodiesterase type 5 (PDE-5) inhibitors:
sildenafil * tadalafil vardenafil | ↓ sildenafil
↓ N-desmethyl-sildenafil | INTELENCE and sildenafil can be co-administered without dose adjustments, however, the dose of sildenafil may need to be altered based on clinical effect. |
| Platelet aggregation inhibitors:
clopidogrel | ↓ clopidogrel (active) metabolite | Activation of clopidogrel to its active metabolite may be decreased when clopidogrel is co-administered with INTELENCE. Alternatives to clopidogrel should be considered. |
7.4 Drugs Without Clinically Significant Interactions with INTELENCE
In addition to the drugs included in Table 4, the interaction between INTELENCE and the following drugs were evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)] : didanosine, enfuvirtide (ENF), ethinylestradiol/norethindrone, omeprazole, paroxetine, raltegravir, ranitidine, and tenofovir disoproxil fumarate.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to INTELENCE during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Prospective pregnancy data from clinical trials and the APR are not sufficient to adequately assess the risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Etravirine use during pregnancy has been evaluated in a limited number of individuals as reported by the APR, and available data show 1 birth defect in 66 first trimester exposures to etravirine-containing regimens (see Data) .
The estimated background rate for major birth defects is 2.7% in the U.S. reference population of the Metropolitan Atlanta Congenital Defects Program (MACDP). The rate of miscarriage is not reported in the APR. The estimated background rate of miscarriage in clinically recognized pregnancies in the U.S. general population is 15–20%. The background risk of major birth defects and miscarriage for the indicated population is unknown.
In animal reproduction studies, no adverse developmental effects were observed with orally administered etravirine at exposures equivalent to those at the maximum recommended human dose (MRHD) of 400 mg daily (see Data) .
Data
Human Data
Based on prospective reports to the APR of 116 live births following exposure to etravirine-containing regimens during pregnancy (including 66 exposed in the first trimester and 38 exposed in the second/third trimester), the number of birth defects in live births for etravirine was 1 out of 66 with first trimester exposure and 0 out of 38 with second/third trimester exposure. Prospective reports from the APR of overall major birth defects in pregnancies exposed to INTELENCE is compared with a U.S. background major birth defect rate. Methodological limitations of the APR include the use of MACDP as the external comparator group. Limitations of using an external comparator include differences in methodology and populations, as well as confounding due to the underlying disease; these limitations preclude an accurate comparison of outcomes.
INTELENCE (200 mg twice daily) in combination with other antiretroviral agents was evaluated in a clinical trial enrolling 15 pregnant subjects during the second and third trimesters of pregnancy and postpartum. Thirteen subjects completed the trial through postpartum period (6–12 weeks after delivery). The pharmacokinetic data demonstrated that exposure to total etravirine was generally higher during pregnancy compared with postpartum [see Clinical Pharmacology (12.3)] .
Among subjects who were virologically suppressed (HIV-1 RNA less than 50 copies/mL) at baseline (9/13), virologic suppression was maintained through the third trimester and postpartum period. Among subjects with HIV-1 RNA greater than 50 copies/mL and less than 400 copies/mL at baseline (3/13), viral loads remained less than 400 copies/mL. In one subject with HIV-1 RNA greater than 1,000 copies/mL at baseline (1/13), HIV-1 RNA remained greater than 1,000 copies/mL during the study period. Thirteen infants were born to 13 HIV-infected pregnant individuals in this study. HIV-1 test results were not available for 2 infants. Among the eleven infants with HIV-1 test results available, who were born to 11 HIV-infected pregnant individuals who completed the study, all had test results that were negative for HIV-1 at the time of delivery. No unexpected safety findings were observed compared with the known safety profile of INTELENCE in non-pregnant adults.
Animal Data
Reproductive and developmental toxicity studies were performed in rats (at 250, 500 and 1,000 mg/kg/day) and rabbits (at 125, 250 and 375 mg/kg/day) administered etravirine on gestation days 6 through 16, and 6 through 19, respectively. In both species, no treatment-related embryo-fetal effects were observed. In addition, no treatment-related effects were observed in a pre- and postnatal development study performed in rats administered oral doses up to 500 mg/kg/day on gestation days 7 through lactation day 7. The systemic drug exposures achieved at the high dose in these animal studies were equivalent to those at the MRHD.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommend that HIV-1-infected mothers not breastfeed their infants to avoid risking postnatal transmission of HIV.
Based on limited data, etravirine has been shown to be present in human breast milk. There are no data on the effects of etravirine on the breastfed infant, or the effects of etravirine on milk production.
Because of the potential for (1) HIV-1 transmission (in HIV-negative infants), (2) developing viral resistance (in HIV-positive infants) and (3) adverse reactions in breastfed infants similar to those seen in adults, instruct mothers not to breastfeed if they are receiving INTELENCE.
8.4 Pediatric Use
The safety and effectiveness of INTELENCE have been established for the treatment of HIV-infected pediatric patients from 2 years of age to less than 18 years [see Indications and Usage (1)and Dosage and Administration (2.3)] . Use of INTELENCE in pediatric patients 2 years to less than 18 years of age is supported by evidence from adequate and well-controlled studies of INTELENCE in adults with additional data from two Phase 2 trials in treatment-experienced pediatric subjects, TMC125-C213, 6 years to less than 18 years of age (N=101) and TMC125-C234/IMPAACT P1090, 2 years to less than 6 years of age (N=20). Both studies were open-label, single arm trials of etravirine plus an optimized background regimen. In clinical trials, the safety, pharmacokinetics, and efficacy were comparable to that observed in adults except for rash (greater than or equal to Grade 2) which was observed more frequently in pediatric subjects [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] . Postmarketing reports of Stevens-Johnson syndrome in pediatric patients receiving INTELENCE have been reported [see Warnings and Precautions (5.1), and Adverse Reactions (6.2)] .
Treatment with INTELENCE is not recommended in pediatric patients less than 2 years of age [see Clinical Pharmacology (12.3)] . Five HIV-infected subjects from 1 year to < 2 years of age were enrolled in TMC125-C234/IMPAACT P1090. Etravirine exposure was lower than reported in HIV-infected adults (AUC 12hgeometric mean ratio [90% CI] was 0.59 [0.34, 1.01] for pediatric subjects from 1 year to < 2 years of age compared to adults). Virologic failure at Week 24 (confirmed HIV-RNA greater than or equal to 400 copies/mL) occurred in 3 of 4 evaluable subjects who discontinued before or had reached Week 24. Genotypic and phenotypic resistance to etravirine developed in 1 of the 3 subjects who experienced virologic failure.
8.5 Geriatric Use
Clinical studies of INTELENCE did not include sufficient numbers of subjects aged 65 years of age and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
8.6 Hepatic Impairment
No dose adjustment of INTELENCE is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. The pharmacokinetics of INTELENCE have not been evaluated in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)] .
8.7 Renal Impairment
Since the renal clearance of etravirine is negligible (less than 1.2%), a decrease in total body clearance is not expected in patients with renal impairment. No dose adjustments are required in patients with renal impairment. As etravirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
10 OVERDOSAGE
There is no specific antidote for overdose with INTELENCE. Human experience of overdose with INTELENCE is limited. The highest dose studied in healthy volunteers was 400 mg once daily. Treatment of overdose with INTELENCE consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Because etravirine is highly protein bound, dialysis is unlikely to result in significant removal of the active substance.
11 DESCRIPTION
INTELENCE ®(etravirine) is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
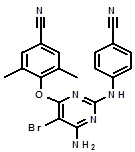
The chemical name for etravirine is 4-[[6-amino-5-bromo-2-[(4-cyanophenyl)amino]-4-pyrimidinyl]oxy]-3,5-dimethylbenzonitrile. Its molecular formula is C 20H 15BrN 6O and its molecular weight is 435.28. Etravirine has the following structural formula:
Etravirine is a white to slightly yellowish-brown powder. Etravirine is practically insoluble in water over a wide pH range. It is very slightly soluble in propylene glycol and slightly soluble in ethanol. Etravirine is soluble in polyethylene glycol (PEG)400 and freely soluble in some organic solvents (e.g., N,N-dimethylformamide and tetrahydrofuran).
INTELENCE ®25 mg tablets are available as white to off-white, oval scored tablets for oral administration. Each 25 mg tablet contains 25 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
INTELENCE ®100 mg tablets are available as white to off-white, oval tablets for oral administration. Each 100 mg tablet contains 100 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
INTELENCE ®200 mg tablets are available as white to off-white, biconvex, oblong tablets for oral administration. Each 200 mg tablet contains 200 mg of etravirine and the inactive ingredients colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose and silicified microcrystalline cellulose.
12 CLINICAL PHARMACOLOGY
12.3 Pharmacokinetics
The pharmacokinetic properties of INTELENCE were determined in healthy adult subjects and in treatment-experienced HIV-1-infected adult and pediatric subjects. The systemic exposures (AUC) to etravirine were lower in HIV-1-infected subjects (Table 5) than in healthy subjects.
| Parameter | Etravirine
N=575 |
|---|---|
|
|
| AUC 12h(ng∙h/mL) | |
| Geometric mean ± standard deviation | 4522 ± 4710 |
| Median (range) | 4380 (458–59084) |
| C 0h(ng/mL) | |
| Geometric mean ± standard deviation | 297 ± 391 |
| Median (range) | 298 (2–4852) |
Note: The median protein binding adjusted EC 50for MT4 cells infected with HIV-1/IIIB in vitroequals 4 ng/mL.
Absorption and Bioavailability
Following oral administration, etravirine was absorbed with a T maxof about 2.5 to 4 hours. The absolute oral bioavailability of INTELENCE is unknown.
In healthy subjects, the absorption of etravirine is not affected by co-administration of oral ranitidine or omeprazole, drugs that increase gastric pH.
Effects of Food on Oral Absorption
The systemic exposure (AUC) to etravirine was decreased by about 50% when INTELENCE was administered under fasting conditions, as compared to when INTELENCE was administered following a meal. Within the range of meals studied, the systemic exposures to etravirine were similar. The total caloric content of the various meals evaluated ranged from 345 kilocalories (17 grams fat) to 1160 kilocalories (70 grams fat).
Distribution
Etravirine is about 99.9% bound to plasma proteins, primarily to albumin (99.6%) and alpha 1-acid glycoprotein (97.66% to 99.02%) in vitro. The distribution of etravirine into compartments other than plasma (e.g., cerebrospinal fluid, genital tract secretions) has not been evaluated in humans.
Metabolism
In vitroexperiments with human liver microsomes (HLMs) indicate that etravirine primarily undergoes metabolism by CYP3A, CYP2C9, and CYP2C19 enzymes. The major metabolites, formed by methyl hydroxylation of the dimethylbenzonitrile moiety, were at least 90% less active than etravirine against wild-type HIV in cell culture.
Elimination
After single dose oral administration of 800 mg 14C-etravirine, 93.7% and 1.2% of the administered dose of 14C-etravirine was recovered in the feces and urine, respectively. Unchanged etravirine accounted for 81.2% to 86.4% of the administered dose in feces. Unchanged etravirine was not detected in urine. The mean (± standard deviation) terminal elimination half-life of etravirine was about 41 (± 20) hours.
Specific Populations
Geriatric Patients
Population pharmacokinetic analysis in HIV-infected subjects showed that etravirine pharmacokinetics are not considerably different within the age range (18 to 77 years) evaluated [see Use in Specific Populations (8.5)] .
Pediatric Patients
The pharmacokinetics of etravirine in 115 treatment-experienced HIV-1-infected pediatric subjects, 2 years to less than 18 years of age showed that the administered weight-based dosages resulted in etravirine exposure comparable to that in adults receiving INTELENCE 200 mg twice daily [see Dosage and Administration (2.3)] . The pharmacokinetic parameters for etravirine (AUC 12hand C 0h) are summarized in Table 6.
| Study | TMC125-C213 | TMC125-C234/IMPAACT P1090 |
|---|---|---|
| Age Range (years) | (6 years to less than 18 years) | (2 years to less than 6 years) |
| Parameter | N=101 | N=14 |
| AUC 12h(ng∙h/mL) | ||
| Geometric mean ± standard deviation | 3742 ± 4314 | 3504 ± 2923 |
| Median (range) | 4499 (62–28865) | 3579 (1221–11815) |
| C 0h(ng/mL) | ||
| Geometric mean ± standard deviation | 205 ± 342 | 183 ± 240 |
| Median (range) | 287 (2–2276) | 162 (54–908) |
The pharmacokinetics and dose of etravirine in pediatric subjects less than 2 years of age have not been established [see Use in Specific Populations (8.4)].
Male and Female Patients
No significant pharmacokinetic differences have been observed between males and females.
Racial or Ethnic Groups
Population pharmacokinetic analysis of etravirine in HIV-infected subjects did not show an effect of race on exposure to etravirine.
Patients with Renal Impairment
The pharmacokinetics of etravirine have not been studied in patients with renal impairment. The results from a mass balance study with 14C-etravirine showed that less than 1.2% of the administered dose of etravirine is excreted in the urine as metabolites. No unchanged drug was detected in the urine. As etravirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Use in Specific Populations (8.7)] .
Patients with Hepatic Impairment
Etravirine is primarily metabolized by the liver. The steady state pharmacokinetic parameters of etravirine were similar after multiple dose administration of INTELENCE to subjects with normal hepatic function (16 subjects), mild hepatic impairment (Child-Pugh Class A, 8 subjects), and moderate hepatic impairment (Child-Pugh Class B, 8 subjects). The effect of severe hepatic impairment on the pharmacokinetics of etravirine has not been evaluated [see Use in Specific Populations (8.6)] .
Pregnancy and Postpartum
After intake of INTELENCE 200 mg twice daily in combination with other antiretroviral agents (13 subjects with 2 NRTIs, 1 subject with 2 NRTIs + lopinavir + ritonavir, 1 subject with 2 NRTIs + raltegravir), based on intra-individual comparison, the C maxand AUC 12hof total etravirine were 23 to 42% higher during pregnancy compared with postpartum (6–12 weeks). The C minof total etravirine was 78 to 125% higher during pregnancy compared with postpartum (6–12 weeks), while two subjects had C min<10 ng/mL in the postpartum period (6–12 weeks) [C minof total etravirine was 11 to 16% higher when these 2 subjects are excluded] (see Table 7) [see Use in Specific Populations (8.1)] . Increased etravirine exposures during pregnancy are not considered clinically significant. The protein binding of etravirine was similar (>99%) during the second trimester, third trimester, and postpartum period.
| Parameter
Mean ± SD (median) | Postpartum
N=10 | 2
ndTrimester
N=13 | 3
rdTrimester
N=10 * |
|---|---|---|---|
| C min, ng/mL | 269 ± 182 (284) † | 383 ± 210 (346) | 349 ± 103 (371) |
| C max, ng/mL | 569 ± 261 (528) | 774 ± 300 (828) | 785 ± 238 (694) |
| AUC 12h, ng∙h/mL | 5004 ± 2521 (5246) | 6617 ± 2766 (6836) | 6846 ± 1482 (6028) |
Patients with Hepatitis B and/or Hepatitis C Virus Co-Infection
Population pharmacokinetic analysis of the TMC125-C206 and TMC125-C216 trials showed reduced clearance for etravirine in HIV-1-infected subjects with hepatitis B and/or C virus co-infection. Based upon the safety profile of INTELENCE [see Adverse Reactions (6)] , no dose adjustment is necessary in patients co-infected with hepatitis B and/or C virus.
Drug Interactions
Etravirine is a substrate of CYP3A, CYP2C9, and CYP2C19. Therefore, co-administration of INTELENCE with drugs that induce or inhibit CYP3A, CYP2C9, and CYP2C19 may alter the therapeutic effect or adverse reaction profile of INTELENCE.
Etravirine is an inducer of CYP3A and inhibitor of CYP2C9, CYP2C19 and P-gp. Therefore, co-administration of drugs that are substrates of CYP3A, CYP2C9 and CYP2C19 or are transported by P-gp with INTELENCE may alter the therapeutic effect or adverse reaction profile of the co-administered drug(s).
Drug interaction studies were performed with INTELENCE and other drugs likely to be co-administered and some drugs commonly used as probes for pharmacokinetic interactions. The effects of co-administration of other drugs on the AUC, C max, and C minvalues of etravirine are summarized in Table 8 (effect of other drugs on INTELENCE). The effect of co-administration of INTELENCE on the AUC, C max, and C minvalues of other drugs are summarized in Table 9 (effect of INTELENCE on other drugs). For information regarding clinical recommendations, [see Drug Interactions (7)] .
| Co-administered Drug | Dose/Schedule of Co-administered Drug | N | Exposure | Mean Ratio of
Etravirine
Pharmacokinetic Parameters 90% CI; No Effect = 1.00 |
||
|---|---|---|---|---|---|---|
| C max | AUC | C min | ||||
| CI = Confidence Interval; N = number of subjects with data; N.A. = not available; ↑ = increase; ↓ = decrease; ↔ = no change | ||||||
|
||||||
| Co-administration with HIV protease inhibitors (PIs) | ||||||
| Atazanavir | 400 mg once daily | 14 | ↑ | 1.47
(1.36–1.59) | 1.50
(1.41–1.59) | 1.58
(1.46–1.70) |
| Atazanavir/ritonavir * | 300/100 mg once daily | 14 | ↑ | 1.30
(1.17–1.44) | 1.30
(1.18–1.44) | 1.26
(1.12–1.42) |
| Darunavir/ritonavir | 600/100 mg twice daily | 14 | ↓ | 0.68
(0.57–0.82) | 0.63
(0.54–0.73) | 0.51
(0.44–0.61) |
| Lopinavir/ritonavir
(tablet) | 400/100 mg twice daily | 16 | ↓ | 0.70
(0.64–0.78) | 0.65
(0.59–0.71) | 0.55
(0.49–0.62) |
| Ritonavir | 600 mg twice daily | 11 | ↓ | 0.68
(0.55–0.85) | 0.54
(0.41–0.73) | N.A. |
| Saquinavir/ritonavir | 1000/100 mg twice daily | 14 | ↓ | 0.63
(0.53–0.75) | 0.67
(0.56–0.80) | 0.71
(0.58–0.87) |
| Tipranavir/ritonavir | 500/200 mg twice daily | 19 | ↓ | 0.29
(0.22–0.40) | 0.24
(0.18–0.33) | 0.18
(0.13–0.25) |
| Co-administration with nucleoside reverse transcriptase inhibitors (NRTIs) | ||||||
| Didanosine | 400 mg once daily | 15 | ↔ | 1.16
(1.02–1.32) | 1.11
(0.99–1.25) | 1.05
(0.93–1.18) |
| Tenofovir disoproxil fumarate | 300 mg once daily | 23 | ↓ | 0.81
(0.75–0.88) | 0.81
(0.75–0.88) | 0.82
(0.73–0.91) |
| Co-administration with CCR5 antagonists | ||||||
| Maraviroc | 300 mg twice daily | 14 | ↔ | 1.05
(0.95–1.17) | 1.06
(0.99–1.14) | 1.08
(0.98–1.19) |
| Maraviroc (when co-administered with darunavir/ritonavir) † | 150/600/100 mg twice daily | 10 | ↔ | 1.08
(0.98–1.20) | 1.00
(0.86–1.15) | 0.81
(0.65–1.01) |
| Co-administration with integrase strand transfer inhibitors | ||||||
| Raltegravir | 400 mg twice daily | 19 | ↔ | 1.04
(0.97–1.12) | 1.10
(1.03–1.16) | 1.17
(1.10–1.26) |
| Co-administration with other drugs | ||||||
| Artemether/lumefantrine | 80/480 mg, 6 doses at 0, 8, 24, 36, 48, and 60 hours | 14 | ↔ | 1.11
(1.06–1.17) | 1.10
(1.06–1.15) | 1.08
(1.04–1.14) |
| Atorvastatin | 40 mg once daily | 16 | ↔ | 0.97
(0.93–1.02) | 1.02
(0.97–1.07) | 1.10
(1.02–1.19) |
| Clarithromycin | 500 mg twice daily | 15 | ↑ | 1.46
(1.38–1.56) | 1.42
(1.34–1.50) | 1.46
(1.36–1.58) |
| Fluconazole | 200 mg once daily in the morning | 16 | ↑ | 1.75
(1.60–1.91) | 1.86
(1.73–2.00) | 2.09
(1.90–2.31) |
| Omeprazole | 40 mg once daily | 18 | ↑ | 1.17
(0.96–1.43) | 1.41
(1.22–1.62) | N.A. |
| Paroxetine | 20 mg once daily | 16 | ↔ | 1.05
(0.96–1.15) | 1.01
(0.93–1.10) | 1.07
(0.98–1.17) |
| Ranitidine | 150 mg twice daily | 18 | ↓ | 0.94
(0.75–1.17) | 0.86
(0.76–0.97) | N.A. |
| Rifabutin | 300 mg once daily | 12 | ↓ | 0.63
(0.53–0.74) | 0.63
(0.54–0.74) | 0.65
(0.56–0.74) |
| Voriconazole | 200 mg twice daily | 16 | ↑ | 1.26
(1.16–1.38) | 1.36
(1.25–1.47) | 1.52
(1.41–1.64) |
| Co-administered Drug | Dose/Schedule of Co-administered Drug | N | Exposure | Mean Ratio of
Co-administered Drug
Pharmacokinetic Parameters 90% CI; No effect = 1.00 |
||
|---|---|---|---|---|---|---|
| C max | AUC | C min | ||||
| CI = Confidence Interval; N = number of subjects with data; N.A. = not available; ↑ = increase; ↓ = decrease; ↔ = no change | ||||||
| Co-administration with HIV protease inhibitors (PIs) | ||||||
| Atazanavir | 400 mg once daily | 14 | ↓ | 0.97
(0.73–1.29) | 0.83
(0.63–1.09) | 0.53
(0.38–0.73) |
| Atazanavir/ritonavir | 300/100 mg once daily | 13 | ↓ | 0.97
(0.89–1.05) | 0.86
(0.79–0.93) | 0.62
(0.55–0.71) |
| Atazanavir/ritonavir * | 300/100 mg once daily | 20 | ↓ | 0.96
(0.80–1.16) | 0.96
(0.76–1.22) | 0.82
(0.55–1.22) |
| Darunavir/ritonavir | 600/100 mg twice daily | 15 | ↔ | 1.11
(1.01–1.22) | 1.15
(1.05–1.26) | 1.02
(0.90–1.17) |
| Fosamprenavir/ritonavir | 700/100 mg twice daily | 8 | ↑ | 1.62
(1.47–1.79) | 1.69
(1.53–1.86) | 1.77
(1.39–2.25) |
| Lopinavir/ritonavir
(tablet) | 400/100 mg twice daily | 16 | ↔ | 0.89
(0.82–0.96) | 0.87
(0.83–0.92) | 0.80
(0.73–0.88) |
| Saquinavir/ritonavir | 1000/100 mg twice daily | 15 | ↔ | 1.00
(0.70–1.42) | 0.95
(0.64–1.42) | 0.80
(0.46–1.38) |
| Tipranavir/ritonavir | 500/200 mg twice daily | 19 | ↑ | 1.14
(1.02–1.27) | 1.18
(1.03–1.36) | 1.24
(0.96–1.59) |
| Co-administration with nucleoside reverse transcriptase inhibitors (NRTIs) | ||||||
| Didanosine | 400 mg once daily | 14 | ↔ | 0.91
(0.58–1.42) | 0.99
(0.79–1.25) | N.A. |
| Tenofovir disoproxil fumarate | 300 mg once daily | 19 | ↔ | 1.15
(1.04–1.27) | 1.15
(1.09–1.21) | 1.19
(1.13–1.26) |
| Co-administration with CCR5 antagonists | ||||||
| Maraviroc | 300 mg twice daily | 14 | ↓ | 0.40
(0.28–0.57) | 0.47
(0.38–0.58) | 0.61
(0.53–0.71) |
| Maraviroc (when co-administered with darunavir/ritonavir) † | 150/600/100 mg twice daily | 10 | ↑ | 1.77
(1.20–2.60) | 3.10
(2.57–3.74) | 5.27
(4.51–6.15) |
| Co-administration with integrase strand transfer inhibitors | ||||||
| Dolutegravir | 50 mg once daily | 16 | ↓ | 0.48
(0.43 to 0.54) | 0.29
(0.26 to 0.34) | 0.12
(0.09 to 0.16) |
| Dolutegravir (when co-administered with darunavir/ritonavir) | 50 mg once daily + 600/100 mg twice daily | 9 | ↓ | 0.88
(0.78 to 1.00) | 0.75
(0.69 to 0.81) | 0.63
(0.52 to 0.76) |
| Dolutegravir (when co-administered with lopinavir/ritonavir | 50 mg once daily + 400/100 mg twice daily | 8 | ↔ | 1.07
(1.02 to 1.13) | 1.11
(1.02 to 1.20) | 1.28
(1.13 to 1.45) |
| Raltegravir | 400 mg twice daily | 19 | ↓ | 0.89
(0.68–1.15) | 0.90
(0.68–1.18) | 0.66
(0.34–1.26) |
| Co-administration with other drugs | ||||||
| Artemether | 80/480 mg, 6 doses at 0, 8, 24, 36, 48, and 60 hours | 15 | ↓ | 0.72
(0.55–0.94) | 0.62
(0.48–0.80) | 0.82
(0.67–1.01) |
| Dihydroartemisinin | 15 | ↓ | 0.84
(0.71–0.99) | 0.85
(0.75–0.97) | 0.83
(0.71–0.97) |
|
| Lumefantrine | ||||||
| 15 | ↓ | 1.07
(0.94–1.23) | 0.87
(0.77–0.98) | 0.97
(0.83–1.15) |
||
| Atorvastatin | 40 mg once daily | 16 | ↓ | 1.04
(0.84–1.30) | 0.63
(0.58–0.68) | N.A. |
| 2-hydroxy-atorvastatin | 16 | ↑ | 1.76
(1.60–1.94) | 1.27
(1.19–1.36) | N.A. | |
| Buprenorphine | Individual dose regimen ranging from 4/1 mg to 16/4 mg once daily | 16 | ↓ | 0.89
(0.76–1.05) | 0.75
(0.66–0.84) | 0.60
(0.52–0.68) |
| Norbuprenorphine | 16 | ↔ | 1.08
(0.95–1.23) | 0.88
(0.81–0.96) | 0.76
(0.67–0.87) |
|
| Clarithromycin | 500 mg twice daily | 15 | ↓ | 0.66
(0.57–0.77) | 0.61
(0.53–0.69) | 0.47
(0.38–0.57) |
| 14-hydroxy-clarithromycin | 15 | ↑ | 1.33
(1.13–1.56) | 1.21
(1.05–1.39) | 1.05
(0.90–1.22) |
|
| Digoxin | 0.5 mg single dose | 16 | ↑ | 1.19
(0.96–1.49) | 1.18
(0.90–1.56) | N.A. |
| Ethinylestradiol | 0.035 mg once daily | 16 | ↑ | 1.33
(1.21–1.46) | 1.22
(1.13–1.31) | 1.09
(1.01–1.18) |
| Norethindrone | 1 mg once daily | 16 | ↔ | 1.05
(0.98–1.12) | 0.95
(0.90–0.99) | 0.78
(0.68–0.90) |
| Fluconazole | 200 mg once daily in the morning | 15 | ↔ | 0.92
(0.85–1.00) | 0.94
(0.88–1.01) | 0.91
(0.84–0.98) |
| R(-) Methadone | Individual dose regimen ranging from 60 to 130 mg/day | 16 | ↔ | 1.02
(0.96–1.09) | 1.06
(0.99–1.13) | 1.10
(1.02–1.19) |
| S(+) Methadone | 16 | ↔ | 0.89
(0.83–0.97) | 0.89
(0.82–0.96) | 0.89
(0.81–0.98) |
|
| Paroxetine | 20 mg once daily | 16 | ↔ | 1.06
(0.95–1.20) | 1.03
(0.90–1.18) | 0.87
(0.75–1.02) |
| Rifabutin | 300 mg once daily | 12 | ↓ | 0.90
(0.78–1.03) | 0.83
(0.75–0.94) | 0.76
(0.66–0.87) |
| 25- O-desacetylrifabutin | 300 mg once daily | 12 | ↓ | 0.85
(0.72–1.00) | 0.83
(0.74–0.92) | 0.78
(0.70–0.87) |
| Sildenafil | 50 mg single dose | 15 | ↓ | 0.55
(0.40–0.75) | 0.43
(0.36–0.51) | N.A. |
| N-desmethyl-sildenafil | 15 | ↓ | 0.75
(0.59–0.96) | 0.59
(0.52–0.68) | N.A. | |
| Voriconazole | 200 mg twice daily | 14 | ↑ | 0.95
(0.75–1.21) | 1.14
(0.88–1.47) | 1.23
(0.87–1.75) |
12.4 Microbiology
Mechanism of Action
Etravirine is an NNRTI of HIV-1. Etravirine binds directly to reverse transcriptase (RT) and blocks the RNA-dependent and DNA-dependent DNA polymerase activities by causing a disruption of the enzyme's catalytic site. Etravirine does not inhibit the human DNA polymerases α, β, and γ.
Antiviral Activity in Cell Culture
Etravirine exhibited activity against laboratory strains and clinical isolates of wild-type HIV-1 in acutely infected T-cell lines, human peripheral blood mononuclear cells, and human monocytes/macrophages with median EC 50values ranging from 0.9 to 5.5 nM (i.e., 0.4 to 2.4 ng/mL). Etravirine demonstrated antiviral activity in cell culture against a broad panel of HIV-1 group M isolates (subtype A, B, C, D, E, F, G) with EC 50values ranging from 0.29 to 1.65 nM and EC 50values ranging from 11.5 to 21.7 nM against group O primary isolates. Etravirine did not show antagonism when studied in combination with the following antiretroviral drugs—the NNRTIs delavirdine, efavirenz, and nevirapine; the N(t)RTIs abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir, and zidovudine; the PIs amprenavir, atazanavir, darunavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, and tipranavir; the gp41 fusion inhibitor ENF; the integrase strand transfer inhibitor raltegravir and the CCR5 co-receptor antagonist maraviroc.
Resistance
In Cell Culture
Etravirine-resistant strains were selected in cell culture originating from wild-type HIV-1 of different origins and subtypes, as well as NNRTI resistant HIV-1. Development of reduced susceptibility to etravirine typically required more than one substitution in reverse transcriptase of which the following were observed most frequently: L100I, E138K, E138G, V179I, Y181C, and M230I.
In Treatment-Experienced Subjects
In the Phase 3 trials TMC125-C206 and TMC125-C216, substitutions that developed most commonly in subjects with virologic failure at Week 48 to the INTELENCE-containing regimen were V179F, V179I, and Y181C which usually emerged in a background of multiple other NNRTI resistance-associated substitutions. In all the trials conducted with INTELENCE in HIV-1 infected subjects, the following substitutions emerged most commonly: L100I, E138G, V179F, V179I, Y181C and H221Y. Other NNRTI-resistance-associated substitutions which emerged on etravirine treatment in less than 10% of the virologic failure isolates included K101E/H/P, K103N/R, V106I/M, V108I, Y181I, Y188L, V189I, G190S/C, N348I and R356K. The emergence of NNRTI substitutions on etravirine treatment contributed to decreased susceptibility to etravirine with a median fold-change in etravirine susceptibility of 40-fold from reference and a median fold-change of 6-fold from baseline.
Cross-Resistance
Cross-resistance among NNRTIs has been observed. Cross-resistance to delavirdine, efavirenz, and/or nevirapine is expected after virologic failure with an etravirine-containing regimen. Virologic failure on a rilpivirine-containing regimen with development of rilpivirine resistance is likely to result in cross-resistance to etravirine (see Treatment-Naïve HIV-1-Infected Subjects in the Phase 3 Trials for EDURANT (rilpivirine)below). Cross-resistance to etravirine has been observed after virologic failure on a doravirine-containing regimen with development of doravirine resistance. Some NNRTI-resistant viruses are susceptible to etravirine, but genotypic and phenotypic testing should guide the use of etravirine (see Baseline Genotype/Phenotype and Virologic Outcome Analysesbelow).
Site-Directed NNRTI Mutant Virus
Etravirine showed antiviral activity against 55 of 65 HIV-1 strains (85%) with single amino acid substitutions at RT positions associated with NNRTI resistance, including the most commonly found K103N. The single amino acid substitutions associated with an etravirine reduction in susceptibility greater than 3-fold were K101A, K101P, K101Q, E138G, E138Q, Y181C, Y181I, Y181T, Y181V, and M230L, and of these, the greatest reductions were Y181I (13-fold change in EC 50value) and Y181V (17-fold change in EC 50value). Mutant strains containing a single NNRTI resistance-associated substitution (K101P, K101Q, E138Q, or M230L) had cross-resistance between etravirine and efavirenz. The majority (39 of 61; 64%) of the NNRTI mutant viruses with 2 or 3 amino acid substitutions associated with NNRTI resistance had decreased susceptibility to etravirine (fold-change greater than 3). The highest levels of resistance to etravirine were observed for HIV-1 harboring a combination of substitutions V179F + Y181C (187 fold-change), V179F + Y181I (123 fold-change), or V179F + Y181C + F227C (888 fold-change).
Clinical Isolates
Etravirine retained a fold-change less than or equal to 3 against 60% of 6171 NNRTI-resistant clinical isolates. In the same panel, the proportion of clinical isolates resistant to delavirdine, efavirenz and/or nevirapine (defined as a fold-change above their respective biological cutoff values in the assay) was 79%, 87%, and 95%, respectively. In TMC125-C206 and TMC125-C216, 34% of the baseline isolates had decreased susceptibility to etravirine (fold-change greater than 3) and 60%, 69%, and 78% of all baseline isolates were resistant to delavirdine, efavirenz, and nevirapine, respectively. Of subjects who received etravirine and were virologic failures in TMC125-C206 and TMC125-C216, 90%, 84%, and 96% of viral isolates obtained at the time of treatment failure were resistant to delavirdine, efavirenz, and nevirapine, respectively.
Treatment-Naïve HIV-1-Infected Subjects in the Phase 3 Trials for EDURANT (Rilpivirine)
There are currently no clinical data available on the use of etravirine in subjects who experienced virologic failure on a rilpivirine-containing regimen. However, in the rilpivirine adult clinical development program, there was evidence of phenotypic cross-resistance between rilpivirine and etravirine. In the pooled analyses of the Phase 3 clinical trials for rilpivirine, 38 rilpivirine virologic failure subjects had evidence of HIV-1 strains with genotypic and phenotypic resistance to rilpivirine. Of these subjects, 89% (34 subjects) of virologic failure isolates were cross-resistant to etravirine based on phenotype data. Consequently, it can be inferred that cross-resistance to etravirine is likely after virologic failure and development of rilpivirine resistance. Refer to the prescribing information for EDURANT (rilpivirine) for further information.
Baseline Genotype/Phenotype and Virologic Outcome Analyses
In TMC125-C206 and TMC125-C216, the presence at baseline of the substitutions L100I, E138A, I167V, V179D, V179F, Y181I, Y181V, or G190S was associated with a decreased virologic response to etravirine. Additional substitutions associated with a decreased virologic response to etravirine when in the presence of 3 or more additional 2008 IAS-USA defined NNRTI substitutions include A98G, K101H, K103R, V106I, V179T, and Y181C. The presence of K103N, which was the most prevalent NNRTI substitution in TMC125-C206 and TMC125-C216 at baseline, did not affect the response in the INTELENCE arm. Overall, response rates to etravirine decreased as the number of baseline NNRTI substitutions increased (shown as the proportion of subjects achieving viral load less than 50 plasma HIV RNA copies/mL at Week 48) (Table 10).
| ENF: enfuvirtide | ||
|
||
| # IAS-USA-Defined NNRTI substitutions * | Etravirine
N=561 |
|
| Re-used/not used ENF | de novoENF | |
| All ranges | 61% (254/418) | 76% (109/143) |
| 0 | 68% (52/76) | 95% (20/21) |
| 1 | 67% (72/107) | 77% (24/31) |
| 2 | 64% (75/118) | 86% (38/44) |
| 3 | 55% (36/65) | 62% (16/26) |
| ≥ 4 | 37% (19/52) | 52% (11/21) |
| Placebo
N=592 |
||
| All ranges | 34% (147/435) | 59% (93/157) |
Response rates assessed by baseline etravirine phenotype are shown in Table 11. These baseline phenotype groups are based on the select subject populations in TMC125-C206 and TMC125-C216 and are not meant to represent definitive clinical susceptibility breakpoints for INTELENCE. The data are provided to give clinicians information on the likelihood of virologic success based on pre-treatment susceptibility to etravirine in treatment-experienced patients.
| ENF: enfuvirtide | |||
|
|||
| Fold Change | Etravirine
N=559 |
||
| Re-used/not used ENF | de novoENF | Clinical response range | |
| All ranges | 61% (253/416) | 76% (109/143) | Overall Response |
| 0–3 | 69% (188/274) | 83% (75/90) | Higher than Overall Response |
| > 3–13 | 50% (39/78) | 66% (25/38) | Lower than Overall Response |
| > 13 | 41% (26/64) | 60% (9/15) | Lower than Overall Response |
| Placebo
N=583 |
|||
| All ranges | 34% (145/429) | 60% (92/154) | |
The proportion of virologic responders (viral load less than 50 HIV-1 RNA copies/mL) by the phenotypic susceptibility score (PSS) of the background therapy, including ENF, is shown in Table 12.
| PSS * | INTELENCE + BR
N=559 | Placebo + BR
N=586 |
|---|---|---|
|
||
| 0 | 43% (40/93) | 5% (5/95) |
| 1 | 61% (125/206) | 28% (64/226) |
| 2 | 77% (114/149) | 59% (97/165) |
| ≥ 3 | 75% (83/111) | 72% (72/100) |
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Etravirine was evaluated for carcinogenic potential by oral gavage administration to mice and rats for up to approximately 104 weeks. Daily doses of 50, 200 and 400 mg/kg were administered to mice and doses of 70, 200 and 600 mg/kg were administered to rats in the initial period of approximately 41 to 52 weeks. The high and middle doses were subsequently adjusted due to tolerability and reduced by 50% in mice and by 50 to 66% in rats to allow for completion of the studies. In the mouse study, statistically significant increases in the incidences of hepatocellular carcinoma and incidences of hepatocellular adenomas or carcinomas combined were observed in treated females. In the rat study, no statistically significant increases in tumor findings were observed in either sex. The relevance of these liver tumor findings in mice to humans is not known. Because of tolerability of the formulation in these rodent studies, maximum systemic drug exposures achieved at the doses tested were lower than those in humans at the clinical dose (400 mg/day), with animal vs. human AUC ratios being 0.6-fold (mice) and 0.2-to-0.7-fold (rats).
Mutagenesis
Etravirine tested negative in the in vitroAmes reverse mutation assay, in vitrochromosomal aberration assay in human lymphocyte, and in vitroclastogenicity mouse lymphoma assay, tested in the absence and presence of a metabolic activation system. Etravirine did not induce chromosomal damage in the in vivomicronucleus test in mice.
14 CLINICAL STUDIES
14.1 Treatment-Experienced Adult Subjects
The clinical efficacy of INTELENCE is derived from the analyses of 48-week data from 2 ongoing, randomized, double-blinded, placebo-controlled, Phase 3 trials, TMC125-C206 and TMC125-C216 (DUET-1 and DUET-2) in subjects with 1 or more NNRTI resistance-associated substitutions. These trials are identical in design and the results below are pooled data from the two trials.
TMC125-C206 and TMC125-C216 are Phase 3 studies designed to evaluate the safety and antiretroviral activity of INTELENCE in combination with a background regimen (BR) as compared to placebo in combination with a BR. Eligible subjects were treatment-experienced HIV-1-infected subjects with plasma HIV-1 RNA greater than 5000 copies/mL while on an antiretroviral regimen for at least 8 weeks. In addition, subjects had 1 or more NNRTI resistance-associated substitutions at screening or from prior genotypic analysis, and 3 or more of the following primary PI substitutions at screening: D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, V82A/F/L/S/T, I84V, N88S, or L90M. Randomization was stratified by the intended use of ENF in the BR, previous use of darunavir/ritonavir, and screening viral load. Virologic response was defined as HIV-1 RNA less than 50 copies/mL at Week 48.
All study subjects received darunavir/ritonavir as part of their BR, and at least 2 other investigator-selected antiretroviral drugs (N[t]RTIs with or without ENF). Of INTELENCE-treated subjects, 25.5% used ENF for the first time ( de novo) and 20.0% re-used ENF. Of placebo-treated subjects, 26.5% used de novoENF and 20.4% re-used ENF.
In the pooled analysis for TMC125-C206 and TMC125-C216, demographics and baseline characteristics were balanced between the INTELENCE arm and the placebo arm (Table 13). Table 13 displays selected demographic and baseline disease characteristics of the subjects in the INTELENCE and placebo arms.
| INTELENCE + BR
N=599 | Placebo + BR
N=604 |
|
|---|---|---|
| RASs = Resistance-Associated Substitutions, BR=background regimen, FC = fold change in EC 50 | ||
|
||
| Demographic characteristics | ||
| Median age, years (range) | 46
(18–77) | 45
(18–72) |
| Sex | ||
| Male | 90.0% | 88.6% |
| Female | 10.0% | 11.4% |
| Race | ||
| White | 70.1% | 69.8% |
| Black | 13.2% | 13.0% |
| Hispanic | 11.3% | 12.2% |
| Asian | 1.3% | 0.6% |
| Other | 4.1% | 4.5% |
| Baseline disease characteristics | ||
| Median baseline plasma HIV-1 RNA (range), log 10copies/mL | 4.8
(2.7–6.8) | 4.8
(2.2–6.5) |
| Percentage of subjects with baseline viral load: | ||
| < 30,000 copies/mL | 27.5% | 28.8% |
| ≥ 30,000 copies/mL and < 100,000 copies/mL | 34.4% | 35.3% |
| ≥ 100,000 copies/mL | 38.1% | 35.9% |
| Median baseline CD4+ cell count (range), cells/mm 3 | 99
(1–789) | 109
(0–912) |
| Percentage of subjects with baseline CD4+ cell count: | ||
| < 50 cells/mm 3 | 35.6% | 34.7% |
| ≥ 50 cells/mm 3and < 200 cells/mm 3 | 34.8% | 34.5% |
| ≥ 200 cells/mm 3 | 29.6% | 30.8% |
| Median (range) number of primary PI substitutions * | 4
(0–7) | 4
(0–8) |
| Percentage of subjects with previous use of NNRTIs: | ||
| 0 | 8.2% | 7.9% |
| 1 | 46.9% | 46.7% |
| > 1 | 44.9% | 45.4% |
| Percentage of subjects with previous use of the following NNRTIs: | ||
| Efavirenz | 70.3% | 72.5% |
| Nevirapine | 57.1% | 58.6% |
| Delavirdine | 13.7% | 12.6% |
| Median (range) number of NNRTI RASs † | 2
(0–8) | 2
(0–7) |
| Median fold change of the virus for the following NNRTIs: | ||
| Delavirdine | 27.3 | 26.1 |
| Efavirenz | 63.9 | 45.4 |
| Etravirine | 1.6 | 1.5 |
| Nevirapine | 74.3 | 74.0 |
| Percentage of subjects with previous use of a fusion inhibitor | 39.6% | 42.2% |
| Percentage of subjects with a Phenotypic Sensitivity Score (PSS) for the background therapy ‡of: | ||
| 0 | 17.0% | 16.2% |
| 1 | 36.5% | 38.7% |
| 2 | 26.9% | 27.8% |
| ≥ 3 | 19.7% | 17.3% |
Efficacy at Week 48 for subjects in the INTELENCE and placebo arms for the pooled TMC125-C206 and TMC125-C216 study populations are shown in Table 14.
| INTELENCE + BR
N=599 | Placebo + BR
N=604 |
|
|---|---|---|
| BR=background regimen | ||
| Virologic responders at Week 48
Viral Load < 50 HIV-1 RNA copies/mL | 359 (60%) | 232 (38%) |
| Virologic failures at Week 48
Viral Load ≥ 50 HIV-1 RNA copies/mL | 123 (21%) | 201 (33%) |
| Death | 11 (2%) | 19 (3%) |
| Discontinuations before Week 48: | ||
| due to virologic failures | 58 (10%) | 110 (18%) |
| due to adverse events | 31 (5%) | 14 (2%) |
| due to other reasons | 17 (3%) | 28 (5%) |
At Week 48, 70.8% of INTELENCE-treated subjects achieved HIV-1 RNA less than 400 copies/mL as compared to 46.4% of placebo-treated subjects. The mean decrease in plasma HIV-1 RNA from baseline to Week 48 was -2.23 log 10copies/mL for INTELENCE-treated subjects and -1.46 log 10copies/mL for placebo-treated subjects. The mean CD4+ cell count increase from baseline for INTELENCE-treated subjects was 96 cells/mm 3and 68 cells/mm 3for placebo-treated subjects.
Of the study population who either re-used or did not use ENF, 57.4% of INTELENCE-treated subjects and 31.7% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL. Of the study population using ENF de novo,67.3% of INTELENCE-treated subjects and 57.2% of placebo-treated subjects achieved HIV-1 RNA less than 50 copies/mL.
Treatment-emergent CDC category C events occurred in 4% of INTELENCE-treated subjects and 8.4% of placebo-treated subjects.
Study TMC125-C227 was a randomized, exploratory, active-controlled, open-label, Phase 2b trial. Eligible subjects were treatment-experienced, PI-naïve HIV-1-infected subjects with genotypic evidence of NNRTI resistance at screening or from prior genotypic analysis. The virologic response was evaluated in 116 subjects who were randomized to INTELENCE (59 subjects) or an investigator-selected PI (57 subjects), each given with 2 investigator-selected N(t)RTIs. INTELENCE-treated subjects had lower antiviral responses associated with reduced susceptibility to the N(t)RTIs and to INTELENCE as compared to the control PI-treated subjects.
14.2 Treatment-Experienced Pediatric Subjects (2 Years to Less Than 18 Years of Age)
The efficacy of INTELENCE for treatment-experienced pediatric subjects is based on two Phase 2 trials, TMC125-C213 and TMC125-C234/IMPAACT P1090.
Pediatric Subjects (6 Years to Less Than 18 Years of Age [TMC125-C213])
TMC125-C213, a single-arm, Phase 2 trial evaluating the pharmacokinetics, safety, tolerability, and efficacy of INTELENCE enrolled 101 antiretroviral treatment-experienced HIV-1 infected pediatric subjects 6 years to less than 18 years of age and weighing at least 16 kg. Subjects eligible for this trial were on an antiretroviral regimen with confirmed plasma HIV-1 RNA of at least 500 copies/mL and viral susceptibility to INTELENCE at screening.
The median baseline plasma HIV-1 RNA was 3.9 log 10copies/mL, and the median baseline CD4+ cell count was 385 × 10 6cells/mm 3.
At Week 24, 52% of subjects had HIV-1 RNA less than 50 copies per mL. The proportion of subjects with HIV-1 RNA less than 400 copies/mL was 67%. The mean CD4+ cell count increase from baseline was 112 × 10 6cells/mm 3.
Pediatric Subjects (2 Years to Less Than 6 Years of Age [TMC125-C234/IMPAACT P1090])
TMC125-C234/IMPAACT P1090 is a Phase 1/2 trial evaluating the pharmacokinetics, safety, tolerability, and efficacy of INTELENCE in 20 antiretroviral treatment-experienced HIV-1 infected pediatric subjects 2 years to less than 6 years of age. The study enrolled subjects who had virologic failure on an antiretroviral treatment regimen after at least 8 weeks of treatment, or who had interrupted treatment for at least 4 weeks. Enrolled subjects had a history of virologic failure while on an antiretroviral regimen, with a confirmed HIV-1 RNA plasma viral load greater than 1,000 copies/mL and with no evidence of phenotypic resistance to etravirine at screening.
The median baseline plasma HIV-1 RNA was 4.4 log 10copies/mL, the median baseline CD4+ cell count was 817.5 × 10 6cells/mm 3, and the median baseline CD4+ percentage was 28%.
Virologic response, defined as achieving plasma viral load less than 400 HIV-1 RNA copies/mL, was evaluated.
Study treatment included etravirine plus an optimized background regimen of antiretroviral drugs. In addition to etravirine, all 20 subjects received a ritonavir-boosted protease inhibitor in combination with 1 or 2 NRTIs (n=14) and/or in combination with an integrase inhibitor (n=7).
At the time of the Week 24 analysis, seventeen subjects had completed at least 24 weeks of treatment or discontinued earlier. At Week 24, the proportion of subjects with less than 400 HIV-1 RNA copies/mL was 88% (15/17), and the proportion of subjects with less than 50 HIV-1 RNA copies/mL was 50% (7/14), for those with available data. The median change in plasma HIV-1 RNA from baseline to Week 24 was -2.14 log 10copies/mL. The median CD4+ cell count increase and the median CD4+ percentage increase from baseline was 298 × 10 6cells/mm 3and 5%, respectively.
16 HOW SUPPLIED/STORAGE AND HANDLING
INTELENCE ®(etravirine) 25 mg tablets are supplied as white to off-white, oval, scored tablets containing 25 mg of etravirine. Each tablet is debossed with "TMC" on one side.
INTELENCE ®(etravirine) 100 mg tablets are supplied as white to off-white, oval tablets containing 100 mg of etravirine. Each tablet is debossed with "TMC125" on one side and "100" on the other side.
INTELENCE ®(etravirine) 200 mg tablets are supplied as white to off-white, biconvex, oblong tablets containing 200 mg of etravirine. Each tablet is debossed with "T200" on one side.
INTELENCE tablets are packaged in bottles in the following configuration:
- 25 mg tablets—bottles of 120 (NDC 59676-572-01). Each bottle contains 2 desiccant pouches.
- 100 mg tablets—bottles of 120 (NDC 59676-570-01). Each bottle contains 3 desiccant pouches.
- 200 mg tablets—bottles of 60 (NDC 59676-571-01). Each bottle contains 3 desiccant pouches.
Store INTELENCE tablets at 25°C (77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature]. Store in the original bottle. Keep the bottle tightly closed in order to protect from moisture. Do not remove the desiccant pouches.
Keep INTELENCE and all medicines out of the reach of children.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Administration
Advise patients to take INTELENCE following a meal twice a day on a regular dosing schedule, as missed doses can result in development of resistance. The type of food does not affect the exposure to etravirine. Inform patients not to take more or less than the prescribed dose of INTELENCE or discontinue therapy with INTELENCE without consulting their physician. INTELENCE must always be used in combination with other antiretroviral drugs [see Dosage and Administration (2.4)].
Advise patients to swallow the INTELENCE tablet(s) whole with a liquid such as water. Instruct patients not to chew the tablets. Patients who are unable to swallow the INTELENCE tablet(s) whole may disperse the tablet(s) in water. The patient should be instructed to do the following:
- place the tablet(s) in 5 mL (1 teaspoon) of water, or at least enough liquid to cover the medication,
- stir well until the water looks milky,
- add approximately 15 mL (1 tablespoon) of liquid. Water may be used, but orange juice or milk may improve taste. Patients should not place the tablets in orange juice or milk without first adding water. The use of warm (temperature greater than 104°F [greater than 40°C]) or carbonated beverages should be avoided.
- drink the mixture immediately,
- rinse the glass several times with orange juice, milk or water and completely swallow the rinse each time to make sure the patient takes the entire dose.
Severe Skin Reactions
Inform patients that severe and potentially life-threatening rash has been reported with INTELENCE. Rash has been reported most commonly in the first 6 weeks of therapy. Advise patients to immediately contact their healthcare provider if they develop rash. Instruct patients to immediately stop taking INTELENCE and seek medical attention if they develop a rash associated with any of the following symptoms as it may be a sign of a more serious reaction such as Stevens-Johnson syndrome, toxic epidermal necrolysis or severe hypersensitivity: fever, generally ill feeling, extreme tiredness, muscle or joint aches, blisters, oral lesions, eye inflammation, facial swelling, swelling of the eyes, lips, mouth, breathing difficulty, and/or signs and symptoms of liver problems (e.g., yellowing of your skin or whites of your eyes, dark or tea colored urine, pale colored stools/bowel movements, nausea, vomiting, loss of appetite, or pain, aching or sensitivity on your right side below your ribs). Patients should understand that if severe rash occurs, they will be closely monitored, laboratory tests will be ordered and appropriate therapy will be initiated [see Warnings and Precautions (5.1)] .
Drug Interactions
INTELENCE may interact with many drugs; therefore, advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, including St. John's wort [see Warnings and Precautions (5.2)] .
Immune Reconstitution Syndrome
Advise patients to inform their healthcare provider immediately of any symptoms of infection, as in some patients with advanced HIV infection (AIDS), signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started [see Warnings and Precautions (5.3)].
Fat Redistribution
Inform patients that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy, including INTELENCE, and that the cause and long-term health effects of these conditions are not known at this time [see Warnings and Precautions (5.4)] .
Pregnancy Registry
Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant individuals exposed to INTELENCE [see Use in Specific Populations (8.1)] .
Lactation
Instruct mothers with HIV-1 infection not to breastfeed because HIV-1 can be passed to the baby in breast milk [see Use in Specific Populations (8.2)] .
Manufactured for:
Janssen Products, LP, Horsham, PA 19044
For patent information: www.janssenpatents.com
© 2008, 2018 Janssen Pharmaceutical Companies
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised March 2023 | |||
|
PATIENT INFORMATION
|
||||
|
Important: Ask your healthcare provider or pharmacist about medicines that should not be taken with INTELENCE. For more information, see the section " What should I tell my healthcare provider before taking INTELENCE?" |
||||
|
What is INTELENCE? INTELENCE is a prescription medicine that is used to treat human immunodeficiency virus-1 (HIV-1) infection in combination with other HIV-1 medicines, in adults and children 2 years of age and older who have taken HIV-1 medicines in the past. HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). INTELENCE is not recommended for use in children less than 2 years of age. |
||||
|
What should I tell my healthcare provider before taking INTELENCE? Before taking INTELENCE tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines may interact with INTELENCE. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.
|
||||
|
How should I take INTELENCE?
|
||||
|
What are the possible side effects of INTELENCE? INTELENCE can cause serious side effects including:
If you get a rash with any of the following symptoms, stop taking INTELENCE and call your healthcare provider or get medical help right away: |
||||
|
|
|
||
|
Sometimes allergic reactions can affect body organs, such as your liver. Call your healthcare provider right away if you have any of the following signs or symptoms of liver problems: |
||||
|
|
|||
The most common side effects of INTELENCE in adults include rash as well as numbness, tingling or pain in the hands or feet. The most common side effects of INTELENCE in children include rash and diarrhea. These are not all the possible side effects of INTELENCE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
|
How should I store INTELENCE?
Keep INTELENCE and all medicines out of the reach of children. |
||||
|
General information about the safe and effective use of INTELENCE Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use INTELENCE for a condition for which it was not prescribed. Do not give INTELENCE to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about INTELENCE that is written for health professionals. |
||||
|
What are the ingredients in INTELENCE? Active ingredient:etravirine. 25 mg and 100 mg INTELENCE tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate and microcrystalline cellulose. 200 mg INTELENCE tablets contain the following inactive ingredients:colloidal silicon dioxide, croscarmellose sodium, hypromellose, magnesium stearate, microcrystalline cellulose, and silicified microcrystalline cellulose. Manufactured for:
For more information, call Janssen Products, LP at 1-800-526-7736. |
||||
PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label
120 Tablets
NDC59676-570-01
INTELENCE
®
(etravirine) tablets
100 mg
Each tablet contains
100 mg of etravirine.
Rx only
janssen
ALERT: Find out about medicines that
should NOT be taken with INTELENCE
®
from your healthcare provider.