TACHOSIL- thrombin human and fibrinogen patch
Baxalta US Inc.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATIONThese highlights do not include all the information needed to use TACHOSIL safely and effectively. See full prescribing information for TACHOSIL.
TACHOSIL® Fibrin Sealant Patch Absorbable Patch for Topical Use Initial U.S. Approval: 2010 RECENT MAJOR CHANGESINDICATIONS AND USAGETachoSil is a fibrin sealant patch indicated for use with manual compression in adult and pediatric patients as an adjunct to hemostasis in cardiovascular and hepatic surgery, when control of bleeding by standard surgical techniques (such as suture, ligature or cautery) is ineffective or impractical. (1) Limitations for TachoSil Use Not for use in place of sutures or other forms of mechanical ligation in treatment of major arterial or venous bleeding. (1) Not for use in children under one month of age. (8.4) DOSAGE AND ADMINISTRATIONDOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONSWARNINGS AND PRECAUTIONS
ADVERSE REACTIONSThe most common adverse reactions reported in >1% of patients during clinical trials were anemia, nausea and vomiting, fever, abdominal pain, increased white blood cell count, ascites, itching, atrial fibrillation, pleural effusion, gastrointestinal hemorrhage, wound infection, hypophosphatemia, urinary tract infection and post-procedural bile leakage in hepatic surgery. (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Baxter Healthcare Corporation at 1-866-888-2472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. USE IN SPECIFIC POPULATIONSPregnancy: No human or animal data. Use only if clearly needed. (8.1) Pediatric: Use in children under the age of one month may be unsafe or ineffective due to small size and limited ability to apply the patch as recommended. (8.4) See 17 for PATIENT COUNSELING INFORMATION. Revised: 7/2015 |
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
TachoSil is a fibrin sealant patch indicated for use with manual compression in adult and pediatric patients as an adjunct to hemostasis in cardiovascular and hepatic surgery when control of bleeding by standard surgical techniques (such as suture, ligature or cautery) is ineffective or impractical.
Limitations for Use
- •
- TachoSil cannot safely or effectively be used in place of sutures or other form of mechanical ligation for the treatment of major arterial or venous bleeding.
- •
- Not for use in children under one month of age.
2 DOSAGE AND ADMINISTRATION
For topical use on cardiovascular or hepatic tissue only
- •
- Determine the number of patches to be applied by the size of the bleeding area.
- •
- Apply the yellow, active side of the patch to the bleeding area.
- •
- When applying TachoSil, do not exceed the maximum number of patches shown in Table 1 [see Warnings and Precautions (5.6)].
|
Table 1. Amount of Fibrinogen and Thrombin per Total Patch Size
|
|||
|
TachoSil Patch Size |
Human Fibrinogen (mg) |
Human Thrombin (Units) |
Maximum Number of Patches to be Applied |
|
3.7 inch x 1.9 inch |
337.4 |
123.1 |
10 |
|
1.9 inch x 1.9 inch |
170.5 |
62.2 |
14 |
|
1.2 inch x 1.0 inch |
55.5 |
20.3 |
42 |
2.1 Preparation for Application
TachoSil comes ready to use in sterile packages and must be handled using sterile technique in aseptic conditions. Discard damaged packages as resterilization is not possible.
- •
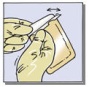
- When in the operating room, the outer aluminum foil pouch may be opened in a non-sterile environment (Fig. 1A). The inner sterile blister must be opened in a sterile environment (Fig. 1B).
- •
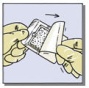
- Remove the TachoSil patch from the blister (Fig. 1C), which can be used as a container for pre-moistening of the patch, if needed.
- •
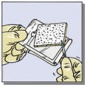
- Determine the size of patch(es) to be applied to the bleeding surface. Select the appropriate TachoSil patch so that it extends 1 to 2 cm beyond the margins of the wound. The patch can be cut to the correct size and shape if desired (Fig. 1D). If more than one patch is used, overlap patches by at least 1 cm.
- •
- Prior to application, cleanse the area to be treated to remove disinfectants and other fluids. The fibrinogen and thrombin proteins can be denatured by alcohol, iodine or heavy metal ions. If any of these substances have been used to clean the wound area, thoroughly irrigate the area before the application of TachoSil.
- •
- Apply TachoSil directly to the bleeding area either wet or dry. If applied wet, pre-moisten TachoSil in 0.9% saline solution for no more than one minute and then apply immediately. In the case of a wet tissue surface (e.g., oozing bleeding) TachoSil may be applied without pre-moistening.
Figure 1: Pictures illustrating steps for preparation for application of TachoSil
 |  |  | 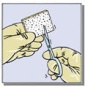 |
|
A |
B |
C |
D |
2.2 Method of Application
• Cleanse surgical instruments and gloves with saline solution to reduce the adherence to the TachoSil patch. The white, inactive side of TachoSil may also adhere to surgical instruments (e.g., forceps) or gloves covered with blood due to the affinity of collagen to blood.
• Apply the yellow, active side of the patch to the bleeding area (Fig. 2A) and hold in place with gentle pressure applied through moistened gloves or a moist pad for at least three minutes (Fig. 2B).
• To avoid pulling the patch loose, first place a clean surgical instrument at one end of the patch before relieving the pressure (Fig. 2C). Gentle irrigation may also aid in removing the pre-moistened pad or gloved hand without removing TachoSil from the bleeding area.
• Leave TachoSil in place once it adheres to organ tissue. Only remove unattached TachoSil patches (or part of) and replace with new patches.
• TachoSil cannot be resterilized once removed from inner pouch. Discard unused, opened packages of TachoSil at the end of the procedure.
Figure 2: Pictures illustrating steps for method of application of TachoSil
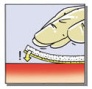 | 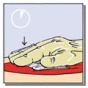 | 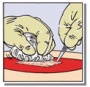 |
|
A |
B |
C |
Record patient name and TachoSil batch number every time that TachoSil is administered to a patient.
3 DOSAGE FORMS AND STRENGTHS
TachoSil is a topical fibrin sealant patch consisting of human fibrinogen and human thrombin coated onto an equine collagen sponge. The active side of the patch is yellow in color due to the presence of a colorant riboflavin (E101); and the non-active side is off-white in color.
Strength:
Each absorbable TachoSil patch contains:
Per square inch:
|
Human fibrinogen |
23.2 – 47.7 mg (35.5 mg) |
|
Human thrombin |
8.4 – 17.4 Units (12.9 U) |
Per square centimeter:
|
Human fibrinogen |
3.6 – 7.4 mg (5.5 mg) |
|
Human thrombin |
1.3 – 2.7 Units (2.0 U) |
4 CONTRAINDICATIONS
Do not use TachoSil for:
- •
- Intravascular application. Bleeding from large defects in visible arteries or veins where the injured vascular wall requires repair and maintenance of vessel patency or where there would be persistent exposure of TachoSil to blood flow during absorption of the product. This can result in life-threatening thromboembolic events [see Warnings and Precautions (5.1)].
- •
- Individuals known to have anaphylactic or severe systemic reaction to human blood products or horse proteins [see Warnings and Precautions (5.2)].
5 WARNINGS AND PRECAUTIONS
5.1 Thrombosis
Thrombosis can occur if TachoSil is applied intravascularly. Ensure that TachoSil is applied to the surface of cardiac, vascular, or hepatic tissue only.
5.2 Hypersensitivity Reactions
Hypersensitivity or allergic/anaphylactoid reactions may occur with TachoSil. Symptoms associated with allergic anaphylactic reactions include: flush, urticaria, pruritus, nausea, drop in blood pressure, tachycardia or bradycardia, dyspnea, severe hypotension and anaphylactic shock. These reactions may occur in patients receiving TachoSil for the first time or may increase with repetitive applications of TachoSil.
5.3 Infection
Avoid application to contaminated or infected areas of the body, or in the presence of active infection.
5.5 Compression
When placing TachoSil into cavities or closed spaces, avoid over-packing because this may cause compression of underlying tissue.
5.6 Dislodged Material
Use only minimum amount of TachoSil patches necessary to achieve hemostasis. Do not pack. Theoretically, excess patch material can become dislodged and migrate to other areas of the body. Remove unattached pieces of TachoSil; if medically necessary [see Dosage and Administration (2.2)].
5.7 Transmissible Infectious Agents
Because the biological components of this product are made from human blood, it may carry a risk of transmitting infectious agents (e.g., viruses), and theoretically, the variant Creutzfeldt-Jakob disease (vCJD) agent and the Creutzfeldt-Jakob disease (CJD) agent. The risk that TachoSil will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain virus infections, and by inactivating and removing, certain viruses [see Description (11)]. Despite these measures, such products can still potentially transmit disease. There is also the possibility that unknown infectious agents may be present in such products.
All infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Baxter Healthcare Corporation, at telephone number 1-800-423-2862. The physician should discuss the risks and benefits of this product with the patient.
Some viruses, such as parvovirus B19, are particularly difficult to remove or inactivate at this time. Parvovirus B19 most seriously affects pregnant women (fetal infection); immune-compromised individuals or individuals with an increased erythropoiesis (e.g., hemolytic anemia) [see Use in Specific Populations (8.1) and Patient Counseling Information (17)].
6 ADVERSE REACTIONS
The adverse reactions reported in more than one percent of patients during clinical trials were anemia, nausea and vomiting, fever, abdominal pain, increased white blood cell count, ascites, itching, atrial fibrillation, pleural effusion, gastrointestinal hemorrhage, wound infection, hypophosphatemia, urinary tract infection, and post-procedural bile leakage in hepatic surgery.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Cardiovascular Surgery
In the cardiovascular trial, the most frequently reported adverse reactions were atrial fibrillation and pleural effusion. Seventy-four percent (74%) of patients treated with TachoSil and 75% of comparator treated patients experienced one or more clinically relevant adverse reactions (see Table 2).
|
Table 2. Most Frequent Adverse Reactions (Cardiovascular Trial) |
||
|
Adverse Reaction |
TachoSil |
Comparator* |
|
N = 62†
|
N = 57†
|
|
|
Atrial fibrillation |
18 (29%) |
14 (25%) |
|
Pleural effusion |
14 (23%) |
11 (19%) |
|
Pyrexia |
4 (6%) |
3 (5%) |
Hepatic Surgery
In the hepatic surgery trial, the most frequently reported adverse reactions were nausea and anemia (see Table 3). Ninety-four percent (94%) of patients treated with TachoSil and 94% of comparator treated patients experienced one or more clinically relevant adverse reactions.
|
Table 3. Most Frequent Adverse Reactions (Hepatic Resection Trial) |
||
|
Adverse Reaction |
TachoSil |
Comparator* |
|
N = 114†
|
N = 109†
|
|
|
Nausea |
34 (30%) |
29 (27%) |
|
Anemia |
26 (23%) |
23 (21%) |
Post-operative bile leakage was observed in 8 (7%) of patients after treatment with TachoSil and 13 (12%) after treatment with comparator.¶
Immunogenicity
Antibodies against components of fibrin sealant/hemostatic products may occur rarely.
However in a clinical trial with human fibrinogen/human thrombin sponge (patch) in hepatic surgery, in which patients were investigated for the development of antibodies, about 26% of the 96 patients tested and treated with human fibrinogen/human thrombin sponge (patch) developed antibodies to equine collagen. The equine collagen antibodies that developed in some patients after human fibrinogen/human thrombin sponge (patch) use were not reactive with human collagen. One patient developed antibodies to human fibrinogen.
There were no adverse events attributable to the development of human fibrinogen or equine collagen antibodies.
There is very limited clinical data available regarding re-exposure of the human fibrinogen/human thrombin sponge (patch). Two subjects have been re-exposed in a clinical trial and have not reported any immune-mediated adverse events, however, their antibody status to collagen or fibrinogen is unknown.
Pediatric Clinical Trial Experience
In pediatric patients, the most frequently reported adverse reactions were diarrhea, hypertension and increased transaminases (see Table 4). Ninety-four percent (94%) of patients treated with TachoSil and 100% of comparator treated patients experienced one or more clinically relevant adverse reactions.
|
Table 4. Most Frequent Adverse Reactions in Pediatric Patients (All Trials) |
||
|
Adverse Reaction |
TachoSil |
Comparator* |
|
N = 36†
|
N = 9†
|
|
|
Diarrhea |
6 (17%) |
0 |
|
Hypertension |
6 (17%) |
1 (11%) |
|
Transaminases Increased |
4 (11%) |
0 |
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of TachoSil. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been reported in postmarketing experience with TachoSil:
General disorders and administration site conditions: drug ineffective, inflammation, granuloma, catheter-related complication, multi-organ failure
Injury, poisoning and procedural complications: foreign body trauma, post-procedural pulmonary embolism
Vascular disorders: thrombosis
Infections and infestations: hepatitis C
Respiratory, thoracic and mediastinal disorders: respiratory distress, laryngeal edema, hemothorax
Blood and lymphatic system disorders: splenic hemorrhage, eosinophilia
Renal and urinary disorders: renal artery thrombosis, renal failure
Endocrine disorders: parathyroid disorder
Eye disorders: mydriasis
Nervous system disorders: nerve compression
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Animal reproduction studies have not been conducted with TachoSil. There are no adequate and well-controlled studies in pregnant women. It is also not known whether TachoSil can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. TachoSil should be administered to pregnant women only if clearly needed.
8.3 Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when TachoSil is administered to nursing mothers.
8.4 Pediatric Use
The use of TachoSil has been studied in patients aged one month to 16 years; use in children under the age of one month may be unsafe or ineffective due to small size and limited ability to apply the patch as recommended. Use of TachoSil in the one month to 16 years age group is supported by evidence from adequate and well-controlled studies of TachoSil in adults with additional data from two clinical trials, which included 36 pediatric patients at the age of 16 years or younger. The data supports the use of TachoSil for hemostasis in pediatric patients undergoing cardiovascular and hepatic surgery [see Clinical Studies (14.3)].
8.5 Geriatric Use
Clinical trials to date included 326 patients older than 65 years of age receiving TachoSil. No overall differences in safety or effectiveness were observed between the elderly and younger patients, however, greater susceptibility of some older patients to adverse reactions cannot be ruled out.
11 DESCRIPTION
TachoSil Fibrin Sealant Patch is a sterile, bioabsorbable combination product comprised of two active substances (human plasma-derived fibrinogen and human plasma-derived thrombin) coated onto a collagen sponge of equine origin. The collagen sponge serves as a flexible and mechanically stable carrier for the active substances to facilitate application of the human fibrinogen and thrombin to the wound surface. The active side of the patch is yellow in color due to the presence of a colorant riboflavin (E101); and the non-active side is off-white in color. Each square inch of the patch contains approximately 35.5 mg of human fibrinogen and 12.9 units of human thrombin. Other inactive ingredients include equine collagen, human albumin, sodium chloride, sodium citrate, and L‑arginine hydrochloride.
TachoSil is sterilized by gamma irradiation after completion of inner and outer packaging, resulting in a sterile product in a sterile inner package.
Viral Clearance
The active biological substances of TachoSil (human fibrinogen and human thrombin) are manufactured from pooled human plasma collected in FDA-licensed facilities in the United States. Human plasma is tested by FDA-licensed Nucleic Acid Test (NAT) for hepatitis B virus (HBV), hepatitis C virus (HCV), and human immunodeficiency virus-1 (HIV-1). NAT testing for hepatitis A virus (HAV) and parvovirus B19 is also performed. Human plasma is also tested for the presence of hepatitis B surface antigen (HBsAg), and antibodies to hepatitis C virus (anti-HCV) and human immunodeficiency viruses types 1 and 2 (anti-HIV 1/2).
The manufacturing procedure for each TachoSil component and final product include processing steps designed to reduce the risk of viral transmission. In particular, the virus clearance steps in the manufacture of human fibrinogen and thrombin include pasteurization, precipitation and adsorption. The virus clearance step in the manufacture of the collagen sponge is the pH treatment.
The virus clearance capacity of these procedures in the manufacture of fibrinogen, thrombin and collagen sponge has been validated using viruses with a wide range of physicochemical characteristics. These in vitro validation studies were conducted using samples from manufacturing intermediates spiked with virus suspensions of known titers followed by further processing under conditions equivalent to those in the respective manufacturing steps. The cumulative virus reduction factors (expressed as log10) are shown in Table 5 for each virus tested.
|
|||||
|
Table 5. Cumulative Virus Reduction Factors for the Components of TachoSil |
|||||
|
Cumulative Reduction Factors for Virus Removal/Inactivation of Human Thrombin |
|||||
|
Reduction Factors [log10] of Virus* tested |
|||||
|
Enveloped Viruses |
Non-enveloped Viruses |
||||
|
Manufacturing step |
HIV-1 |
HSV |
BVDV |
CPV |
HAV |
|
Pasteurization, precipitation and adsorption steps |
≥19.6 |
≥21.4 |
≥13.4 |
6.6 |
8.7 |
|
Cumulative Reduction Factors for Virus Removal/Inactivation of Human Fibrinogen |
|||||
|
Reduction Factors [log10] of Virus* tested |
|||||
|
Enveloped Viruses |
Non-enveloped Viruses |
||||
|
Manufacturing step |
HIV-1 |
HSV† |
BVDV |
CPV |
HAV |
|
Pasteurization, precipitation and adsorption steps |
≥9.6 |
≥9.1 |
≥11.2 |
4.4 |
≥6.7 |
|
Reduction Factors for Virus Removal/Inactivation of the Collagen Sponge (equine) |
|||||
|
Reduction Factors [log10] of Virus* tested |
|||||
|
Enveloped Viruses |
Non-enveloped Viruses |
||||
|
Manufacturing step |
PRV |
PI-3 |
PPV |
Reo3 |
|
|
pH treatment |
≥5.7 |
≥5.9 |
--- |
--- |
|
A validation study was also conducted to evaluate the capacity for gamma irradiation to inactivate and/or remove viruses in the final TachoSil product. The virus reduction factors (expressed as log10) are shown in Table 6 for each virus tested.
|
||||
|
Table 6. Virus Reduction Factors for TachoSil Final Sterilization by Gamma Irradiation |
||||
|
Reduction Factor of Gamma Irradiation (Final Sterilization of TachoSil) |
||||
|
Reduction Factors [log10] of Virus* tested |
||||
|
Enveloped Viruses |
Non-enveloped Viruses |
|||
|
Manufacturing step |
PRV |
PI-3 |
PPV |
Reo3 |
|
Gamma Irradiation |
≥4.7 |
≥4.0 |
3.0 |
≥6.2 |
All infections considered by a physician possibly to have been transmitted by this product should be reported to Baxter [see Warnings and Precautions (5.7)].
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
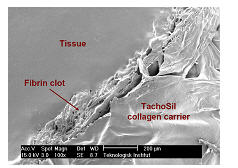
The mechanism of action of TachoSil is based on the interaction between the active biological substances (human fibrinogen and human thrombin) and the physiology of the fibrin clot formation (Fig. 3A). Upon contact with a bleeding wound surface, the active substances coated onto the equine collagen patch become dissolved and partly diffuse into the wound surface. The subsequent fibrinogen-thrombin reaction initiates the last step in the cascade of biochemical reactions-conversion of fibrinogen into fibrin monomers that further polymerize to form the fibrin clot.
Hemostasis is achieved when the formed fibrin clot adheres the collagen patch to the wound surface, thus providing a physical barrier to bleeding (Fig 3B). TachoSil exhibits flexibility to accommodate for the physiological movements of tissues and organs and can withstand pressures up to 61.4 hPa (46.1 mmHg).
Figure 3: Scanning Electron Microscopy Photos of TachoSil
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies to evaluate the carcinogenic potential of TachoSil or studies to determine the genotoxicity or the effect of TachoSil on fertility have not been performed. An assessment of the carcinogenic potential of TachoSil was completed to demonstrate minimal carcinogenic risk from product use.
13.2 Animal Toxicology and/or Pharmacology
In a study conducted in swine, TachoSil was applied to liver wounds and showed progressive degradation. However, remnants of the TachoSil patch may remain present for more than 12 months. Histologic examination at 26 and 52 weeks revealed that granulation tissue encapsulates TachoSil remnants and forms a firm capsule around them. Remnants were found in all treated animals after 26 weeks (12/12) and in most animals (6/8) after 52 weeks. No other local reactivity or toxicities were noted.
14 CLINICAL STUDIES
14.1 Cardiovascular
An open-label, multi-center, randomized, parallel-group study comparing TachoSil with comparator (hemostatic fleece without additional active coagulation stimulating compounds) treatment was conducted to evaluate TachoSil for control of bleeding in 119 patients undergoing cardiovascular surgery requiring cardiopulmonary bypass procedure. Of the 119 subjects, 88 (74%) were male and 31 (26%) female. Mean (range) age was 67 (23 to 86) years; subjects older than 65 years constituted 58% of the male and 74% of the female population. All subjects were White/Caucasian.
In the Intent-to-Treat (ITT) population, 59 patients were randomized to treatment with TachoSil and 60 patients were randomized to treatment with the comparator. A larger proportion of patients in the TachoSil treatment group (44/59; 75%) than in the comparator treatment group (20/60; 33%) achieved hemostasis within three minutes, which was a statistically significant difference (p<0.0001).
Fifty-six out of 59 (95%) patients in the TachoSil treatment group achieved hemostasis at six minutes compared to 43 out of 60 (72%) in the comparator treatment group, which also was statistically significant (p=0.0006), see Table 7.
|
Table 7. Efficacy Results in Cardiovascular Surgery, by Treatment, Intent-to-Treat Population |
||||
|
Treatment |
Total number
|
Percentage of
|
95% CI for
|
p-value† |
|
Hemostasis at 3 min |
||||
|
TachoSil (n=59) |
44 |
75% |
[0.635; 0.857] |
<0.0001 |
|
Comparator‡ (n=60) |
20 |
33% |
[0.214; 0.453] |
|
|
Hemostasis at 6 min |
||||
|
TachoSil (n=59) |
56 |
95% |
[0.893;1.000] |
0.0006 |
|
Comparator‡ (n=60) |
43 |
72% |
[0.603; 0.831] |
|
14.2 Hepatic
A randomized, open-label, parallel group, multi-center trial comparing TachoSil with comparator was conducted to evaluate TachoSil for the secondary treatment of local bleeding in patients undergoing hepatic surgery.
A total of 114 patients were randomized to treatment with TachoSil and 110 patients were randomized to treatment with comparator. A similar proportion of male patients and female patients were randomly assigned in the trial (53% and 47%, respectively). The mean (SD) age of patients was 58.1 (13.95) years and in both treatment groups approximately 30% of the patients were above 65 years. The majority of patients were White/Caucasian (80%) and the most common ethnicity was non-Hispanic/non-Latino (88%).¶
A larger proportion of patients in the TachoSil treatment group (81%) than in the comparator treatment group (50%) achieved hemostasis within the first three minutes after treatment application. The primary analysis of the proportion of patients who achieved hemostasis within three minutes showed a statistically significant difference between treatment groups in favor of TachoSil (p<0.001) and obtained a clinical relevant difference between the two groups defined from the estimated odds ratio, see Table 8.
With respect to one of the two secondary endpoints, 108 (95%) patients in the TachoSil group and 84 (76%) patients in the comparator group achieved hemostasis within five minutes in which the secondary analysis also showed a statistically significant difference between the group in favor of TachoSil (p<0.001, multiplicity adjusted), see Table 8.
|
Table 8. Efficacy Results in Hepatic Surgery, by Treatment, Intent-To-Treat Population |
||||
|
Treatment |
Total number of patients
|
Percentage of patients
|
Odds ratio
|
p-value* |
|
Hemostasis at 3 min |
||||
|
TachoSil (n=114) |
92 |
81% |
4.87 |
<0.001 |
|
Comparator† (n=110) |
55 |
50% |
||
|
Hemostasis at 5 min |
||||
|
TachoSil (n=114) |
108 |
95% |
6.24 |
<0.001 |
|
Comparator† (n=110) |
84 |
76% |
||
|
Time to hemostasis‡ |
<0.001 |
|||
|
TachoSil (n=114) | ||||
|
Comparator (n=110) | ||||
14.3 Pediatric
In a pediatric subset of a randomized, open-label, parallel group, multi-center trial comparing TachoSil with comparator the efficacy of TachoSil for the secondary treatment of local bleeding in pediatric patients undergoing hepatic surgery was evaluated.
In the randomized part of the study, eight patients were treated with TachoSil and nine patients were treated with the comparator. After the randomization phase, an additional 12 patients were treated with TachoSil in a single extension arm to reach 20 patients exposed to TachoSil. A similar proportion of male and female pediatric patients were treated overall in the trial (48% and 52%, respectively). The majority of patients were White/Caucasian (79%) and the most common ethnicity was non-Hispanic/non-Latino (69%). The mean age was slightly higher in the TachoSil group (4.58 years; range 0.4, 13.0 years) than in the comparator group (3.77 years; range 0.4, 16.0 years).
A larger proportion of the patients in the TachoSil group (7/8 [88%]) than in the comparator group (4/9 [44%]) achieved hemostasis within three minutes in the randomized part of the study. The results were similar considering the whole set of pediatric patients exposed to TachoSil (17/20 [85%]), see Table 9. No statistical testing was performed of the difference between the two groups due to the small sample size. Seven patients in the TachoSil group (88%) and seven patients in the comparator group (78%) achieved hemostasis within five minutes, see Table 9.
|
Table 9. Achievement of Hemostasis of Pediatric Patients Undergoing Hepatic Surgery, Intent-To-Treat Population/Safety Population |
|||
|
Treatment |
Total number of
|
Percentage
|
Exact
|
|
Hemostasis at 3 min |
|||
|
TachoSil (n=8) |
7 |
88% |
[47.3, 99.7] |
|
Comparator* (n=9) |
4 |
44% |
[13.7, 78.8] |
|
Hemostasis at 5 min |
|||
|
TachoSil (n=8) |
7 |
88% |
[47.3, 99.7] |
|
Comparator* (n=9) |
7 |
78% |
[40.0, 97.2] |
|
All TachoSil Patients† |
|||
|
Hemostasis at 3 min |
17 |
85% |
[62.1, 96.8] |
|
TachoSil (n=20) |
|||
|
Hemostasis at 5 min |
19 |
95% |
[75.1, 99.9] |
|
TachoSil (n=20) |
|||
In another clinical study, 16 pediatric patients were included in a prospective, multi-center, uncontrolled, study where TachoSil was used in connection with resection of the liver with or without segmental liver transplantation.
The analysis of the efficacy parameter, time to hemostasis, showed that 13 of 16 patients achieved hemostasis within three minutes after application of TachoSil. One patient obtained hemostasis eight minutes after treatment application and two subjects failed to achieve satisfactory hemostasis within 10 minutes, which necessitated alternative hemostatic measures.
The estimated proportion of patients achieving hemostasis within three minutes was 81%, which is considered similar to results seen in the randomized controlled study.
16 HOW SUPPLIED/STORAGE AND HANDLING
Each TachoSil patch is packaged in an appropriately sized blister pack of polystyrene formed foil and grid varnish coated medicinal paper and overwrapped with an aluminum laminate foil pack with a desiccant bag. Each patch is packaged individually.
Each TachoSil patch is supplied in the following pack sizes:
|
(NDC 0944-8701-01) |
|
(NDC 0944-8702-02) |
|
(NDC 0944-8703-01) |
|
(NDC 0944-8703-05) |
17 PATIENT COUNSELING INFORMATION
- •
- Advise patients that, because TachoSil is made from human blood, it may carry a risk of transmitting infections agents (e.g., viruses), and theoretically, the Creutzfeldt-Jakob (CJD) agent [see Warnings and Precautions (5.7)].
- •
- Because TachoSil may cause the formation of clots in blood vessels if exposed intravascularly, advise patients to consult their physician if they experience chest pain, shortness of breath or difficulty speaking or swallowing, or leg tenderness or swelling [see Warnings and Precautions (5.1)].
- •
- Instruct patients to consult their physician if symptoms of B19 virus infection appear (fever, drowsiness and chills) followed about two weeks later by a rash and joint pain. Parvovirus B19 most seriously affects pregnant women (fetal infection); immune-compromised individuals or individuals with an increased erythropoiesis (e.g., hemolytic anemia) [see Warnings and Precautions (5.7)].
Distributed by:
Baxter Healthcare Corporation
Westlake Village, CA 91362 USA
Manufactured by:
Takeda Austria GmbH
St. Peter Strasse 25
A-4020 Linz, Austria
U.S. License No. 1894
Revised: July 2015
TachoSil is a trademark of Takeda Nycomed AS registered with the U.S. Patent and Trademark Office.
All other trademarks are the property of their respective owners.
TAC272 R1
PRINCIPAL DISPLAY PANEL – 3.7 inch x 1.9 inch Patch Carton
NDC 0944-8701-01
Absorbable Fibrin
Sealant Patch
TachoSil®
1 patch of 3.7 inch x 1.9 inch
(9.5 cm × 4.8 cm)
Single use only
Topical use only
Do not use intravascularly
Store at 2°C to 25°C (36°F to 77°F)
Do not freeze
Directions for use: See package insert
Use immediately once the foil pouch is opened
Do not resterilize
Dispose of any unused product or waste material
in accordance with local requirements
Rx Only
Product Code 1144922
Baxter
PRINCIPAL DISPLAY PANEL – 1.9 inch x 1.9 inch Patch Carton
NDC 0944-8702-02
Absorbable Fibrin
Sealant Patch
TachoSil®
2 patches of 1.9 inch x 1.9 inch
(4.8 cm × 4.8 cm)
Single use only
Topical use only
Do not use intravascularly
Store at 2°C to 25°C (36°F to 77°F)
Do not freeze
Directions for use: See package insert
Use immediately once the foil pouch is opened
Do not resterilize
Dispose of any unused product or waste material
in accordance with local requirements
Rx Only
Product Code 1144923
Baxter
PRINCIPAL DISPLAY PANEL – 1.2 inch x 1.0 inch Patch Carton
NDC 0944-8703-01
Absorbable Fibrin
Sealant Patch
TachoSil®
1 patch of 1.2 inch x 1.0 inch
(3.0 cm × 2.5 cm)
Single use only
Topical use only
Do not use intravascularly
Store at 2°C to 25°C (36°F to 77°F)
Do not freeze
Directions for use: See package insert
Use immediately once the foil pouch is opened
Do not resterilize
Dispose of any unused product or waste material
in accordance with local requirements
Rx Only
Product Code 0000000
Baxter
| TACHOSIL
thrombin human and fibrinogen patch |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| TACHOSIL
thrombin human and fibrinogen patch |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| TACHOSIL
thrombin human and fibrinogen patch |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - Baxalta US Inc. (079887619) |