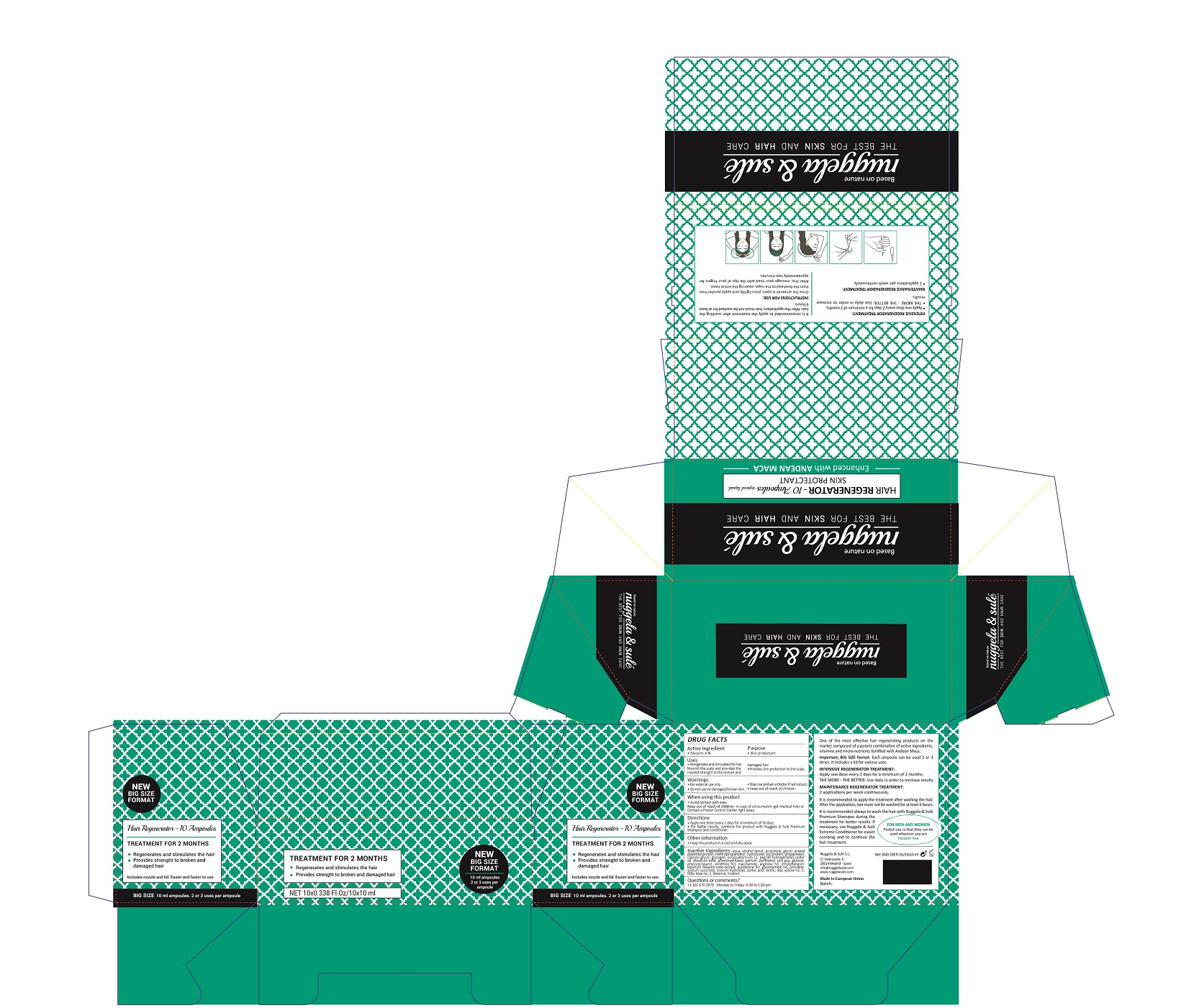
Warnings
For external use only.
Do not use on damaged or broken skin.
Stop use and ask a doctor if rash occurs.
Keep out of reach of children.
Whhen using this product do not get into eyes.
Children under 6 months: as a doctor.
Directions
- apply one dose every 2 days for a minimun of 30 days
- for better results, combine this product with Nuggela & Sule Premium shampoo and Conditioner
Uses
- regenerates and stimulates the hair
- nourish the scalp and provides the needed strenght to the broken and damaged hair
- provides skin protection to the scalp
Inactive Ingredients
AQUA, ALCOHOL DENAT, PROPYLENE GLYCOL, PLACENTAL PROTEIN, METHYLPROPANEDIOL, HYDROLYZED SOY PROTEIN, PROPANEDIOL, CAPRYLYL GLYCOL, GLYCOGEN, POLYQUATERNIUM-11, PEG-40 HYDROGENATED CASTOR OIL
DISODIUM EDTA, PHENOXYETHANOL, PARFUM, PANTHENOL, ZINC PCA, GLUCOSE, PHENYLPROPANOL, ORNITHINE HCL, NIACINAMIDE, ARGININE HCL, CHLORPHENESIN, LEPIDIUM MEYENII ROOT EXTRACT, PYRIDOXINE HCL, GLUCOSAMINE HCL, CITRULLINE, SODIUM SUCCINATE, SODIUM HYDROXIDE,SORBIC ACID, BIOTIN, D&C Yellow No. 5, FD&C Blue No.1, LIMONENE, LINALOOL