CERENIA- maropitant citrate injection, solution
Pfizer Animal Health
----------
Cerenia® (maropitant citrate)
Injectable Solution
DESCRIPTION
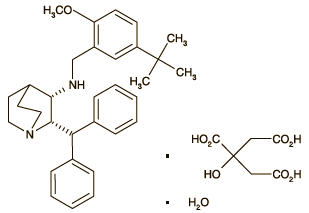
Maropitant is a neurokinin (NK1) receptor antagonist that blocks the pharmacological action of substance P in the central nervous system (CNS). Maropitant is the non-proprietary designation for a substituted quinuclidine. The empirical formula is C32H40N2O C6H8O7 H2O and the molecular weight 678.81. The chemical name is (2S,3S)-2- benzhydryl-N-(5-tert-butyl-2-methoxybenzyl) quinuclidin-3- amine citrate monohydrate. Each mL of CERENIA Injectable Solution contains 10 mg maropitant, 63 mg sulphobutylether-beta-cyclodextrin, 3.3 mg meta-cresol and water for injection.
The chemical structure of maropitant citrate is:
INDICATIONS
CERENIA (maropitant citrate) Injectable Solution is indicated for the prevention and treatment of acute vomiting in dogs and for the treatment of vomiting in cats.
DOSAGE AND ADMINISTRATION
Administer CERENIA Injectable Solution subcutaneously at 1 mg/kg (0.45 mg/lb) equal to 1 mL/10 kg (1 mL/22 lb) of body weight once daily for up to 5 consecutive days. Use of refrigerated product may reduce the pain response associated with the injection.
CERENIA Injectable Solution is recommended for use in dogs 8 weeks and older and cats 16 weeks and older.
INFORMATION FOR USE
If vomiting persists despite treatment, the case should be re-evaluated. CERENIA is most effective in preventing vomiting associated with chemotherapy if administered prior to the chemotherapeutic agent.
For dogs, CERENIA Injectable Solution may be used interchangeably with CERENIA Tablets for once daily dosing for the prevention of acute vomiting.
WARNINGS
Not for use in humans. Keep out of reach of children. In case of accidental injection or exposure, seek medical advice. Topical exposure may elicit localized allergic skin reactions in some individuals. Repeated or prolonged exposure may lead to skin sensitization. In case of accidental skin exposure, wash with soap and water. CERENIA is also an ocular irritant. In case of accidental eye exposure, flush with water for 15 minutes and seek medical attention.
In puppies younger than 11 weeks of age, histological evidence of bone marrow hypocellularity was observed at higher frequency and greater severity in puppies treated with CERENIA compared to control puppies. In puppies 16 weeks and older, bone marrow hypocellularity was not observed (see ANIMAL SAFETY).
PRECAUTIONS
The safe use of CERENIA has not been evaluated in dogs or cats used for breeding, or in pregnant or lactating bitches or queens. The safe use of CERENIA has not been evaluated in dogs or cats with gastrointestinal obstruction, or dogs or cats that have ingested toxins.
Use with caution in patients with hepatic dysfunction because CERENIA is metabolized by CYP3A enzymes (see Pharmacokinetics). Use with caution with other medications that are highly protein bound. The concomitant use of CERENIA with other protein bound drugs has not been studied in dogs or cats. Commonly used protein bound drugs include NSAIDs, cardiac, anticonvulsant, and behavioral medications. The influence of concomitant drugs that may inhibit the metabolism of CERENIA has not been evaluated. Drug compatibility should be monitored in patients requiring adjunctive therapy.
CERENIA causes dose related decreases in appetite and body weight (see ANIMAL SAFETY). To maximize therapeutic potential of CERENIA, the underlying cause of vomiting should be identified and addressed in dogs receiving CERENIA.
ADVERSE REACTIONS
For a complete listing of adverse reactions for CERENIA Injectable Solution reported to CVM see: http://www.fda.gov/AnimalVeterinary/SafetyHealth/ ProductSafetyInformation/ucm055369.htm
For a copy of the Material Safety Data Sheet (MSDS) or to report adverse reactions call Pfizer Animal Health at 1-800-366-5288.
DOGS
In a US field study for the prevention and treatment of vomiting associated with administration of cisplatin for cancer chemotherapy, the following adverse reactions were reported in 77 dogs treated with CERENIA Injectable Solution at 1 mg/kg subcutaneously or 41 dogs treated with placebo:
| Adverse Reaction | Placebo (n=41) | CERENIA (n=77) | ||
|---|---|---|---|---|
| # dogs | % occur | # dogs | % occur | |
| Diarrhea | 1 | 2.4 | 6 | 7.8 |
| Anorexia | 0 | 0 | 4 | 5.2 |
| Injection site reaction (swelling, pain upon injection) | 0 | 0 | 3 | 4 |
| Lethargy | 1 | 2.4 | 2 | 2.6 |
The following adverse reactions were reported during the course of a US field study for the prevention and treatment of acute vomiting in dogs treated with 1 mg/kg CERENIA Injectable Solution subcutaneously and/or CERENIA Tablets at a minimum of 2 mg/kg orally once daily for up to 5 consecutive days:
| Adverse Reaction | Placebo (n=69) | CERENIA (n=206) | ||
|---|---|---|---|---|
| # dogs | % occur | # dogs | % occur | |
| Death during study | 4 | 5.8 | 10 | 4.9 |
| Euthanized during study | 0 | 0 | 2 | 1 |
| Diarrhea | 6 | 8.7 | 8 | 3.9 |
| Hematochezia/bloody stool | 5 | 7.2 | 4 | 1.9 |
| Anorexia | 2 | 2.9 | 3 | 1.5 |
| Otitis/Otorrhea | 0 | 0 | 3 | 1.5 |
| Endotoxic Shock | 1 | 1.4 | 2 | 1 |
| Hematuria | 0 | 0 | 2 | 1 |
| Excoriation | 0 | 0 | 2 | 1 |
Other clinical signs were reported but were <0.5% of dogs.
Adverse reactions seen in a European field study included ataxia, lethargy and injection site soreness in one dog treated with CERENIA Injectable Solution.
Post-Approval Experience
The following adverse events are based on post-approval adverse drug experience reporting. Not all adverse events are reported to FDA CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data.
The following adverse events are listed in decreasing order of reporting frequency in dogs: Pain/vocalization upon injection, depression/lethargy, anorexia, anaphylaxis/anaphylactoid reactions (including swelling of the head/face), ataxia, convulsions, and hypersalivation.
Cases of death (including euthanasia) have been reported.
CATS
The following adverse reactions were reported during the course of a US field study for the treatment of vomiting in cats treated with 1 mg/kg CERENIA Injectable Solution subcutaneously once daily for up to five consecutive days:
| Adverse Reaction | Placebo (n=62) | CERENIA (n=133) | ||
|---|---|---|---|---|
| # cats | % occur | # cats | % occur | |
| Moderate Response to Injection*,† | 1 | 1.6 | 30 | 22.6 |
| Significant Response to Injection*,‡ | 1 | 1.6 | 15 | 11.3 |
| Fever/Pyrexia | 2 | 3.2 | 2 | 1.5 |
| Dehydration | 0 | 0 | 3 | 2.3 |
| Lethargy | 0 | 0 | 2 | 1.5 |
| Anorexia | 0 | 0 | 1 | 0.8 |
| Hematuria | 0 | 0 | 1 | 0.8 |
| Hypersalivation | 0 | 0 | 1 | 0.8 |
| Injection site swelling | 1 | 1.6 | 0 | 0 |
The FDA Center for Veterinary Medicine has received reports of adverse events associated with the use of CERENIA in cats since its approval for dogs in 2007. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data. The following adverse events are listed in decreasing order of reporting frequency in cats: depression/lethargy, anorexia, injection site pain, and hypersalivation.
CLINICAL EXPERIENCE
The pain or vocalization upon injection resolves within minutes without treatment. Administration of CERENIA Injectable Solution at refrigerated temperature may mitigate this response (see DOSAGE AND ADMINISTRATION).
Allergic reactions typically resolve with treatment within 48 hours after discontinuing CERENIA administration.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Vomiting is a complex process coordinated centrally by the emetic center which consists of several brainstem nuclei (area postrema, nucleus tractus solitarius, dorsal motor nucleus of the vagus) that receive and integrate sensory stimuli from central and peripheral sources and chemical stimuli from the circulation and the cerebrospinal fluid. Maropitant is a neurokinin 1 (NK1) receptor antagonist which acts by inhibiting the binding of substance P, a neuropeptide of the tachykinin family. Substance P is found in significant concentrations in the nuclei comprising the emetic center and is considered the key neurotransmitter involved in emesis.1 By inhibiting the binding of substance P within the emetic center, maropitant provides broad-spectrum effectiveness against neural (central) and humoral (peripheral) causes of vomiting. In vivo model studies in dogs have shown that maropitant has antiemetic effectiveness against both central and peripheral emetogens including apomorphine, cisplatin, and syrup of ipecac.
- 1
- Diemunsch P, Grelot L. Potential of substance P antagonists as antiemetics. [Review] [60 refs]. Drugs. 2000;60:533-46.
Pharmacokinetics
CERENIA is formulated using sulphobutylether-β-cyclodextrin (SBECD), which exhibits enhanced binding to maropitant at refrigerated temperatures. The enhanced binding affinity reverses rapidly upon warming.
DOGS
The pharmacokinetic characterization associated with maropitant after oral (PO) or subcutaneous (SC) administration in adult Beagle dogs is provided in the table below.
| SC at 1 mg/kg (n=6) | PO at 2 mg/kg (n=8) | PO at 8 mg/kg (n=8) | |
|---|---|---|---|
| AUC0-inf (hr*ng/mL) | 860±137 | 561±322 | 7840±5600 |
| Cmax (ng/mL) | 92±34 | 81±32 | 776±604 |
| T1/2 (hr) | 8.84 (6.07–17.7) | 4.03 (2.48–7.09) | 5.46 (3.39–7.65) |
| Tmax (hr) | 0.75±1.11 | 1.9±0.5 | 1.7±0.7 |
The absolute bioavailability of maropitant was much higher following SC injection (91% at 1 mg/kg) than after PO administration (24% at 2 mg/kg). Oral bioavailability may be underestimated due to the presence of nonlinear kinetics and the resulting longer T1/2 seen after intravenous (IV) administration. Although hepatic first-pass metabolism contributed to the relatively low bioavailability after an oral dose, prandial status does not significantly affect the extent of oral bioavailability. Greater than dose-proportional drug exposure can be expected with an increase in dose (1–16 mg/kg PO). Systemic clearance of maropitant following IV administration was 970, 995, and 533 mL/hr/kg at doses of 1, 2 and 8 mg/kg, respectively. An accumulation ratio of 1.5 was observed following once-daily use of maropitant for five consecutive days at 1 (SC) or 2 mg/kg (PO). Urinary recovery of maropitant and its major metabolite was minimal (<1% each). The hepatic metabolism of maropitant involves two cytochrome P-450 isoenzymes: CYP2D15 and CYP3A12. Based on in vitro enzyme kinetics data, it is believed that the non-linear kinetics may be partially associated with saturation of the low capacity enzyme (CYP2D15). However as doses increase (20–50 mg/kg PO), dose proportionality is re-established. Based upon in vitro enzyme kinetics, involvement of a high capacity enzyme (CYP3A12) may contribute to this return to dose linearity. Plasma protein binding of maropitant was high (99.5%).
Based on differences in plasma trough concentrations from a single study, the exposure of 10 week old puppies to maropitant may be lower than that observed in adult dogs, particularly after doses of 1 or 2 mg/kg.
CATS
The pharmacokinetic profile of maropitant when administered as a single subcutaneous dose of 1 mg/kg body weight to 8 cats was characterized by a mean (range) maximum concentration (Cmax) in plasma of approximately 165 (108-332) ng/mL. Cmax was achieved on average 0.32 (0.25-0.5) hours post-dosing (Tmax). Peak concentrations were followed by a decline in systemic exposure with a mean apparent elimination half-life (t1/2) of 16.8 (10.3-32.8) hours and mean area under the curve (AUC0-∞) of 3490 (1440-6760) hr*ng/mL. There appears to be an age-related effect on the pharmacokinetics of maropitant in cats with kittens (16 wks) having faster clearance than adults. The mean bioavailability of maropitant after subcutaneous administration in cats was 91.3%. The mean total body clearance (CL) and volume of distribution at steady-state (Vss) determined after intravenous administration at 0.25 mg/kg to 8 cats was 0.27 (0.14-0.59) L/h/kg and 3.04 (2.27 to 3.80) L/kg, respectively. Maropitant displays linear kinetics when administered subcutaneously within the 0.25 – 3 mg/kg dose range. Following repeated subcutaneous administration of once-daily doses of 1 mg/kg body weight for 5 consecutive days, accumulation was 250%. Maropitant undergoes cytochrome P450 (CYP) metabolism in the liver. CYP1A and CYP3A-related enzymes were identified as the feline isoforms involved in the hepatic biotransformation of maropitant. Renal and fecal clearances are minor routes of elimination for maropitant, with less than 1% of a 1 mg/ kg subcutaneous dose appearing in the urine or feces as maropitant. For the major metabolite, 10.4% of the maropitant dose was recovered in urine and 9.3% in feces. Plasma protein binding of maropitant in cats was estimated to be 99.1%.
EFFECTIVENESS
DOGS
In laboratory model studies, CERENIA Injectable Solution administered at 1 mg/kg in Beagle dogs reduced the number of emetic events associated with established neural (central) and humoral (peripheral) stimuli. Following administration of apomorphine (central emetic stimuli), vomiting was observed in 16.7% (2 of 12) of dogs treated with CERENIA Injectable Solution and 83.3% (10 of 12) of placebo-treated dogs. Following administration of syrup of ipecac (peripheral emetic stimuli) vomiting was observed in 25% (3 of 12) of dogs treated with CERENIA Injectable Solution and in 100% (12 of 12) of dogs treated with placebo.
In a study of veterinary cancer patients, dogs were treated with CERENIA Injectable Solution or placebo either 1 hour prior to cisplatin (prevention) or after the first vomiting episode following cisplatin (treatment) and monitored for 5 hours. In the groups evaluated for prevention of vomiting, 94.9% (37/39) of the dogs administered CERENIA Injectable Solution and 4.9% (2/41) of the dogs administered placebo did not vomit. In the groups evaluated for treatment, 21% (8/38) of the dogs administered CERENIA Injectable Solution and 5.1% (2/39) of the dogs administered placebo had no further episodes of vomiting following treatment.
| Number of Vomiting Episodes | Dogs with Vomiting Episodes* (% of Dogs) | |||
|---|---|---|---|---|
| Treatment of Vomiting | Prevention of Vomiting | |||
| Placebo (n=39†) | CERENIA (n=38†) | Placebo (n=41) | CERENIA (n=39) | |
|
||||
| 0 | 2 (5.1) | 8 (21.1) | 2 (4.9) | 37 (94.9) |
| 1 | 3 (7.7) | 7 (18.4) | 2 (4.9) | 1 (2.6) |
| 2 | 4 (10.3) | 6 (15.8) | 3 (7.3) | 1 (2.6) |
| 3 | 3 (7.7) | 6 (15.8) | 4 (9.8) | 0 (0) |
| 4 | 4 (10.3) | 4 (10.5) | 3 (7.3) | 0 (0) |
| 5 | 2 (5.1) | 5 (13.2) | 4 (9.8) | 0 (0) |
| 6 | 14 (35.9) | 1 (2.6) | 1 (2.4) | 0 (0) |
| 7 | 2 (5.1) | 1 (2.6) | 12 (29.3) | 0 (0) |
| 8 | 2 (5.1) | 0 (0) | 5 (12.2) | 0 (0) |
| 9 | 2 (5.1) | 0 (0) | 2 (4.9) | 0 (0) |
| 10 | 0 (0) | 0 (0) | 2 (4.9) | 0 (0) |
| 11 | 1 (2.6) | 0 (0) | 0 (0) | 0 (0) |
| 12 | NA | NA | 1 (2.4) | 0 (0) |
In a study of 275 canine patients presented to veterinary hospitals with a history of acute vomiting, dogs were initially administered CERENIA Injectable Solution or placebo on Day 0. Following the initial dose, dogs allocated to the CERENIA group were treated with either CERENIA Tablets at a minimum of 2 mg/kg orally or Injectable Solution at 1 mg/kg subcutaneously once daily at the discretion of the clinician. Dogs allocated to the placebo group were treated using either an injectable placebo solution or placebo tablets once daily at the discretion of the clinician. Of the 199 dogs included in the analysis for effectiveness, 27 of 54 dogs (50%) in the placebo group displayed vomiting at some time during the study and 31 of 145 dogs (21.4%) in the CERENIA-treated group displayed vomiting during the study period.
| Days | Treatment | Route | # dogs | # vomited | % vomited |
|---|---|---|---|---|---|
|
|||||
| Day 0 | Placebo (54) | SC | 54 | 15 | 28% |
| CERENIA (145) | SC | 145 (143*) | 14 | 10% | |
| Day 1 | Placebo (45) | PO | 22 | 3 | 14% |
| SC | 23 | 16 | 70% | ||
| CERENIA (108) | PO | 67 | 2 | 3% | |
| SC | 41 | 16 | 39% | ||
| Day 2 | Placebo (16) | PO | 7 | 2 | 29% |
| SC | 9 | 6 | 67% | ||
| CERENIA (37) | PO | 24 | 0 | 0% | |
| SC | 13 | 8 | 62% | ||
| Day 3 | Placebo (6) | PO | 2 | 0 | 0% |
| SC | 4 | 1 | 25% | ||
| CERENIA (21) | PO | 14 | 0 | 0% | |
| SC | 7 | 5 | 71% | ||
| Day 4 | Placebo (2) | PO | 1 | 0 | 0% |
| SC | 1 | 1 | 100% | ||
| CERENIA (7) | PO | 5 | 0 | 0% | |
| SC | 2 | 1 | 50% | ||
| Day 5 | CERENIA (1) | SC | 1 | 0 | 0% |
In US field studies in veterinary patients, CERENIA Injectable Solution and Tablets were well tolerated in dogs presenting with various clinical conditions including parvovirus, gastroenteritis, and renal disease. There were no notable differences in mean laboratory values between CERENIAtreated and placebo-treated patients.
CERENIA Injectable Solution was used safely in dogs receiving other frequently used veterinary products such as fluid and electrolyte replacement solutions, antimicrobial agents, vaccines, antacids, and antiparasitic agents.
CATS
In a field study, 195 cats were presented to veterinary hospitals with a history of vomiting associated with various clinical conditions including gastroenteritis, gastritis, pancreatitis, inflammatory bowel disease, neoplasia, and hepatic lipidosis. Cats were treated with CERENIA Injectable Solution or placebo (in a ratio of 2:1) and observed in the veterinary hospital for 24 hours for the presence of an emetic event(s) defined as the observation of the act of vomiting or the presence of vomitus. Cats could continue antiemetic treatment every 24 hours for up to 5 consecutive days at the discretion of the clinician. Of 165 cats included in the analysis for effectiveness, 2 CERENIAtreated cats (1.8%) vomited 1 time each and 10 placebotreated cats (18.5%) vomited a total of 15 times in the first 24 hours post treatment.
| Study Day | Treatment | # cats | # vomited | % vomited |
|---|---|---|---|---|
| Day 0 | Placebo | 54 | 10 | 18.5 |
| CERENIA | 111 | 2 | 1.8 | |
| Day 1 | Placebo | 20 | 4 | 20 |
| CERENIA | 34 | 1 | 2.9 | |
| Day 2 | Placebo | 9 | 2 | 22.2 |
| CERENIA | 8 | 0 | 0 | |
| Day 3 | Placebo | 5 | 0 | 0 |
| CERENIA | 5 | 0 | 0 | |
| Day 4 | Placebo | 3 | 0 | 0 |
| CERENIA | 1 | 0 | 0 |
ANIMAL SAFETY
DOGS
Laboratory and field studies have demonstrated that CERENIA Injectable Solution is well tolerated in dogs after subcutaneous administration.
Fifty six Beagle dogs (28 males and 28 females) approximately 16 weeks of age were administered CERENIA Injectable Solution subcutaneously once daily for 15 days at 0, 1, 3, and 5 mg/kg. There were 8 dogs (4 males and 4 females) in the 1 mg/kg group and 16 dogs (8 males and 8 females) in all other groups. The primary treatment-related findings were injection site reactions. Swelling, thickened skin, or pain at one or more of the injection sites on one or more days of the study were observed in 6 of 16 animals treated with 3 mg/kg/day and 5 of 16 animals treated with 5 mg/kg/day. Additionally, the activated partial thromboplastin time (APTT) was prolonged (67.5 seconds, reference range 9-15 seconds) in one male dog in the 1 mg/kg group on study day 15. Relationship of the prolonged APTT to drug administration could not be determined.
Beagle dogs approximately 8 weeks of age were administered CERENIA Injectable Solution subcutaneously once daily for 15 days at 0, 1, 3, and 5 mg/kg using a protocol similar to the previous study. A dose dependent increase in frequency and severity of bone marrow hypoplasia was observed histologically. One placebo-treated dog died on day 14 of the study and was diagnosed with suppurative pancreatitis and esophagitis. Interpretation of the study results is complicated by the health status of study animals. Dogs used in the study were weaned early, minimally acclimated to the test facility, and many of the dogs in the study tested positive for coccidia.
Beagle dogs approximately 10 weeks of age were administered either placebo tablets for 2 days, CERENIA Tablets at 8 mg/kg for 2 days, placebo (saline) subcutaneously (SC) for 5 days, CERENIA Injectable Solution at 1 mg/kg SC for 5 days, or CERENIA Tablets at 2 mg/kg for 5 days (8 dogs in each dose group). Mild pain associated with injection was noted in more dogs and lasted longer in dogs that received maropitant injections compared to saline. Males administered CERENIA at 8 mg/kg orally for 2 days had a decrease in food consumption. Body weight and food consumption were variable throughout the 4 week acclimatization period. Two dogs that received 8 mg/kg maropitant orally for 2 days were below the reference range for reticulocyte counts. Decreases in reticulocyte counts were also seen in 4 (of 8) placebo treated dogs (SC saline for 5 days). Hypocellular femoral bone marrow described as "minimal" was seen in 1 male that received 1 mg/kg maropitant SC for 5 days; reticulocyte counts were not available for this dog.
CATS
Thirty-two domestic short hair cats (16 males and 16 females) approximately 16 weeks of age were administered CERENIA Injectable Solution subcutaneously once daily for 15 days at 0, 1, 3, and 5 mg/kg. There were 8 cats (4 males and 4 females) in each group. Treatment-related, dose dependent findings included pain associated with injection of CERENIA and injection site heat, pain, redness, and firmness. Pain on injection was observed in 5% of cats at 0 mg/kg, 50% of cats at 1 mg/kg, and 75% of cats at 3 and 5 mg/kg. Injection site firmness >10 mm in diameter was observed at one or more of the injection sites, on one or more days of the study, in 1 of 8 cats at 1 mg/kg, 7 of 8 cats at 3 mg/kg, and 7 of 8 cats at 5 mg/kg. There was a statistically significant reduction (p=0.0171) in food intake at 5 mg/kg compared to cats at 0 mg/kg. One cat at 5 mg/kg was lethargic on Days 12, 13, and 14 of the study. Increased skin turgor was observed in 1 cat at 3 mg/kg on Days 10 and 11, 1 cat at 3 mg/kg on Day 12, and 1 cat at 5 mg/kg on Day 12. At gross necropsy, there were no treatment-related findings. Histopathologic evaluation of injection sites revealed a dose dependent inflammatory response.
STORAGE CONDITIONS
CERENIA Injectable Solution should be stored at controlled room temperature 20-25°C (68-77°F) with excursions between 15-30°C (59-86°F). After first vial puncture, CERENIA Injectable Solution should be stored at refrigerated temperature 2-8°C (36-46°F). Use within 90 days of first vial puncture. Stopper may be punctured a maximum of 25 times.
Distributed by:
Pfizer Animal Health
Div. of Pfizer Inc, NY, NY 10017
Revised: February 2012
Made in France
8811852
PRINCIPAL DISPLAY PANEL - 20 mL Vial Label
Cerenia®
(maropitant citrate)
Injectable Solution
Antiemetic
Each mL contains: 10 mg maropitant, 63 mg
sulphobutylether-beta-cyclodextrin, 3.3 mg meta-cresol
and water for injection.
For subcutaneous injection in dogs and cats
CAUTION: Federal (USA) law restricts this drug to use by
or on the order of a licensed veterinarian.
NET CONTENTS: 20 mL
NADA #141-263, Approved by FDA
Pfizer

| CERENIA
maropitant citrate injection, solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Pfizer Animal Health (039055157) |