AURAX OTIC- antipyrine, benzocaine solution
Acella Pharmaceuticals, LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Aurax Otic Solution
(Antipyrine and Benzocaine)
DESCRIPTION: Each mL contains:
Antipyrine.............................................................55.0 mg (5.5%)
Benzocaine.............................................................14.0 mg (1.4%)
(also contains glycerin and oxyquinoline sulfate)
Aurax Otic Solution is an otic solution containing antipyrine, benzocaine, oxyquinoline sulfate and anhydrous glycerin for use in the ear. The solution congeals at 0°C (32°F) but returns to normal consistency, unchanged at room temperature.
Antipyrine is an analgesic with local anesthetic action, it is chemically 2,3-dimethyl-1-phenyl-3-pyrazolin-5-one. Antipyrine occurs as colorless crystals or white powder, has a slightly bitter taste and is soluble in water and alcohol.
Benzocaine is a local anesthetic. it is chemically ethyl- p-aminobenzoate or Benzoic Acid, 4-amino, ethyl ester. The active ingredients are represented by the following structural formulas:
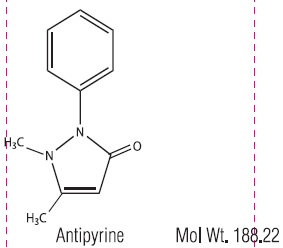
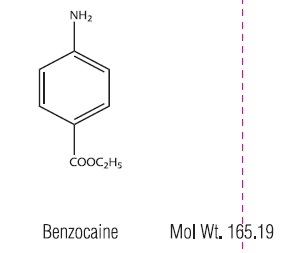
CLINICAL PHARMACOLOGY: Aurax Otic Solution combines the hygroscopic property of anhydrous glycerin with the analgesic action of antipyrine and benzocaine to relieve pressure, reduce inflammation and congestion, and alleviate pain and discomfort in acute otitis media. Aurax Otic Solution does not blanch the tympanic membrane or mask the landmarks and, therefore, does not distort the otoscopic picture.
INDICATIONS AND USAGE: Acute Otitis Media of Various Etiologies
- Prompt relief of pain and reduction of inflammation in the congestive and serous stages.
- Adjuvant therapy during systemic antibiotic administration for resolution of the infection.
Because of the close anatomical relationship of the eustachian tube to the nasal cavity, otitis media is a frequent problem, especially in children in whom the tube is shorter, wider and more horizontal than in adults.
CONTRAINDICATIONS: Aurax Otic Solution is contraindicated in any person with hypersensitivity to any of the components or substances related to them. This product is contraindicated in the presence of spontaneous perforation of the tympanic membrane or discharge.
WARNINGS: FOR USE IN EARS ONLY, NOT FOR USE IN EYES. KEEP OUT OF REACH OF CHILDREN
Discontinue promptly if sensitization or irritation occurs.
PRECAUTIONS: INFORMATION FOR PATIENTS: Avoid contaminating the dropper tip with material from the ear, fingers or other source.
CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY: No long-term studies in animals or humans have been conducted.
PREGNANCY CATEGORY C: Animal reproduction studies have not been conducted with Aurax Otic Solution. It is also not known whether Aurax Otic Solution can cause fetal harm when administered to a pregnant woman, or can affect reproduction capacity. Aurax Otic Solution should be given to a pregnant woman only if clearly needed.
NURSING MOTHERS: It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Aurax Otic Solution is administered to a nursing woman.
DOSAGE AND ADMINISTRATION: ACUTE OTITIS MEDIA: Instill Aurax Otic Solution permitting the solution to run along the wall of the canal until it is filled. Avoid touching the ear with dropper. Then moisten a cotton pledget with Aurax Otic Solution and insert into the meatus. Repeat every one to two hours until pain and congestion are relieved.
REMOVAL OF CERUMEN: Before: Instill Aurax Otic Solution three times daily for two to three days to help detach cerumen from wall of canal and facilitate removal.
After: Aurax Otic Solution is useful for drying out the canal or relieving discomfort.
Before and after removal of cerumen, a cotton pledget moistened with Aurax Otic Solution should be inserted into the meatus following installation.
Note: After use, do not rinse dropper tip, replace cap and close tightly
Protect solution from light and heat, and do not use if it is brown or contains a precipitate.
HOW SUPPLIED: Aurax Otic Solution is supplied in a 14 mL plastic bottle with a dropper tip under cap (NDC 42192-712-14). Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) see USP Controlled Room Temperature). DISCARD THIS PRODUCT SIX MONTHS AFTER OPENING.
All prescriptions and/or recommendations using this product shall be made subject to state and federal statutes as applicable. Note: This is not an Orange Book product. No representation is made as to generic status or bioequivalency. Each person recommending a prescription substitution using this product shall make such recommendations based on each such person's professional opinion and knowledge, upon evaluating the active ingredients, excipients, inactive ingredients and chemical information provided herein.
MANUFACTURED FOR: Acella Pharmaceuticals, LLC
11675 Great Oak Way, Suite 144, Alpharetta, GA 30022
| AURAX OTIC
antipyrine, benzocaine solution |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Acella Pharmaceuticals, LLC (825380939) |
| Registrant - Acella Pharmaceuticals, LLC (825380939) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Acella Pharmaceuticals, LLC | 825380939 | manufacture(42192-712) | |
