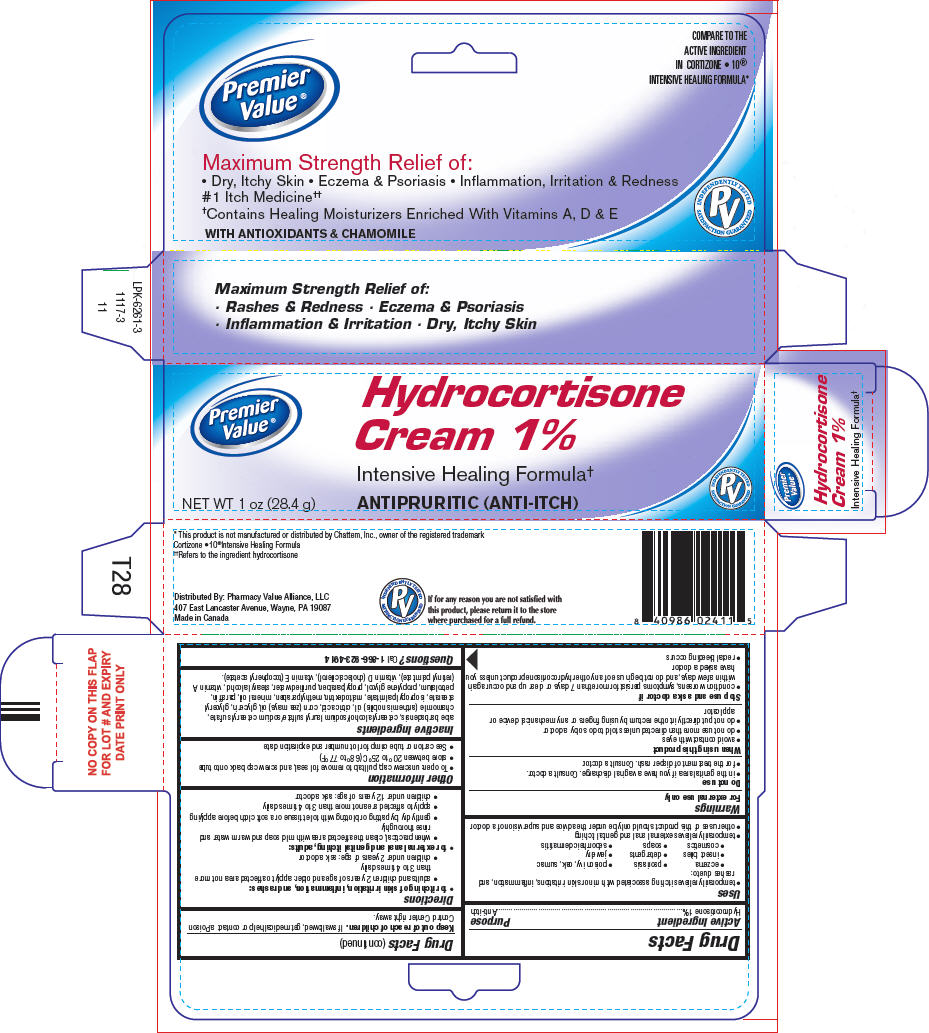
Uses
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
- eczema
- psoriasis
- poison ivy, oak, sumac
- insect bites
- detergents
- jewelry
- cosmetics
- soaps
- seborrheic dermatitis
- temporarily relieves external anal and genital itching
- other uses of this product should only be under the advice and supervision of a doctor
Warnings
For external use only
Do not use
- in the genital area if you have a vaginal discharge. Consult a doctor.
- for the treatment of diaper rash. Consult a doctor.
When using this product
- avoid contact with eyes
- do not use more than directed unless told to do so by a doctor
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Directions
-
for itching of skin irritation, inflammation, and rashes:
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
-
for external anal and genital itching, adults:
- when practical, clean the affected area with mild soap and warm water and rinse thoroughly
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
- apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
Other information
- To open: unscrew cap, pull tab to remove foil seal, and screw cap back onto tube
- store between 20° to 25°C (68° to 77°F)
- See carton or tube crimp for lot number and expiration date
Inactive Ingredients
aloe barbadensis, cetearyl alcohol/sodium lauryl sulfate/sodium cetearyl sulfate, chamomile (anthemis nobilis) oil, citric acid, corn (zea mays) oil, glycerin, glyceryl stearate, isopropyl palmitate, maltodextrin, methylparaben, mineral oil, paraffin, petrolatum, propylene glycol, propylparaben, purified water, stearyl alcohol, vitamin A (retinyl palmitate), vitamin D (cholecalciferol), vitamin E (tocopheryl acetate).